 |
 |
 |
| |
Transmission of Drug Resistance in Newly Infected
Reported by Jules Levin
|
| |
| |
At the resistance Workshop similar rates for transmited HIV drug resistance were reported by a study in the US and in Europe: a CDC report on transmission of drug resistance in recently infected individuals reported a rate of 11.5% while the rate reported by European researchers was 10%. When comparing any studies on the rate of drug resistance and transmission in newly infected individuals, it is important to consider that their may be significant differences between the studies in the patient population studies and in the geographic areas studied. For example, the study by Susan Little published in the New England Journal of Medicine looked primarily at men who have sex with men. While the CDC study reported at this conference and discussed below examined IDUs, MSM, and heterosexuals. The two studies also examined different cities. Drug prescription trends and lifetsyle differences may be present in different cities. As well, other =important differences may be present. In these studies it appears as if the increasing trends we saw in the transmission of drug resistance may be leveling off but in discussion with some of the researchers they feel we may see increases in near future years.
Transmitted Drug-Resistance Persists
Susan Little (Univ Cal at SD) reported in an oral presentation today on the persistance of transmitted drug-resistant virus among individuals with primary HIV-infection who deferred antiretroviral therapy. Ten subjects were idtentified an average of 55 days after their estimated date of HIV-infection (range 23-65 days). The average follow-up on these patients is 242 days (range 64-1019 days). The average HIV viral load was a high 340,000 copies/ml. Average CD4 count was 489. The average replication capacity was 87% (range 40-172) relative to NL4-3 reference strain. 9 of the patients had NNRTI mutations, and 7 patients had the 103N mutation. 3 patients had NRTI mutations, they had a number of key NRTI mutations. 3 patients had protease inhibitor mutations, and 2/3 of these patients had numerous PI mutations. Only 1 patient lost their mutation. Little concluded that transmitted drug resistant mutations in all ARV classes persist for more than a year after HIV-infection. These mutations may last for a long time, perhaps 3 years or longer. But after they are no longer detectable by commercial resistance tests they are likely to remain but undetected. In addition, the mutations may remain longer in genital secretions which may promote transmission of drug-resistant virus.
The relatively high replication capacity for the transmitted viruses suggests this may favor transmission.
These findings have implications for treating patients and for the use of resistance testing. Patients who are treatment-naive may not respond well to treatment if they have HIV-drug resistance. If you wait several years after becoming infected with drug-resistant virus before treating, running a resistance test may not show any resistance because it may have faded into background but might emerge after starting therapy. This raises the issue of whether resistance testing should be recommended for persons with primary HIV-infection and patients who are treatment-naive. Currently Guidelines only recommend resistance testing for treatment-experienced patients.
Prevalence of Transmitted Drug-Resistance in Europe
Annemarie Wensing reported an oral presentation on transmission of drug resistance in 16 countries in Europe in the CATCH Study (Combined Alanysis of resistance Transmission over time of Chronically and acute infected HIV patients in Europe). She looked at 1400 newly diagnosed patients and assessed the prevalence of resistance over the period of 1996-2002. None of the patients were exposed to antiretriviral drugs. Average age was 35, 72% male, 69% subtype B. Average viral load was 65,000 copies/ml, and average CD4 count was 409.
The prevalence of any resistance mutations was 9.9%, NRTI resistance 7.1%, NNRTI resistance, 2.7%, protease 2.3%, and 2 or more classes of drugs 1.8%. For patients with subtype B virus 11.4% had drug resistance vs 3.7% with non-B who had drug resistance. So, if you exclude non-B subtypes the rate is higher. The higher rate in non-B subtypes is due to a longer history of treatment of patients carrying these viruses. She concluded that overall prevalence of drug resistance is 10% in Europe.
In patients with duration of infection <1 year 10.5% had resistance vs 7.2% with duration of infection for >1 year. For MSM, 15.5% had primary resistance, for IDUs 13.7% had resistance, and for heterosexuals 11.6% had primary resistance. 21-47% of patients with NRTI resistance had decreased drug susceptibilty, for with NNRTI resistance 22-26% had reduced drug susceptibility, and for patients with resistance to PIs 1-23% had decreased susceptibility. In other words, patients with drug-resistance had less susceptibility to HIV therapy and are less likely to respond well to HAART.
She reported the trend over time in the prevalence of transmitted drug resistance in patients infected for <1 year. In 1996-98, 14.5% had resistance, in 1999-2000 73% had resistance, and in 2001-02 9.5% had resistance. Wensing suggested that the small difference observed between acute and chronic infections confirms that mutations may persist over time. She recommended that the high prevalence of resistance indicates that resistance testing should be considered in newly diagnosed patients.
Drug Resistance in the USA
Prevalence of Mutations Associated with Resistance to Antiretroviral Drugs among Treatment-Naive Men and Women Newly Diagnosed with HIV in Ten Cities in the USA, 1997-2001: 11.5% of recent infections were resistant to an HIV drug
The Susan Little study published in the New England Journal of Medicine looked at rates of transmission of drug resistant virus among men who have sex with men. This study conducted by the CDC and presented in a poster by DE Bennett looked at a more diverse group of 1082 individuals: 46% were Black, 27% White, and 22% Hispanic. 44% were heterosexual, 46% MSM, and 10% IDU. 19% had recent HIV-infection. The study examined 10 cities: San Francisco, San Diego, Denver, New Orleans, Miami, Grand Rapids (Mich), Detroit, NYC, and Newark. In addition to the different patient population from Little's study, these cities are different than Susan Little examined in her study. These patients were newly diagnosed within the past 12 months. Recent HIV infection was defined as occurring within 4-6 months.
The prevalence of mutations associated with resistance was 8.3% to any drug; 6.4% in NRTIs; 1.7% to NNRTIs; 1.9% to protease inhibitors; and 1.3% to two or more drug classes.
The prevalence of drug resistance among those tested for recent infection: 11.5% of recently infected were resistant to any drug, 8.8% to NRTI, 3.9% to NNRTI, 2.8% to PI, and 3.9% to 2 or more classes of drugs. Among those tested for recent infection but were not recently infected the rates of resistance were lower: 7.4% were resistant to any drug; 6.1% were resistant to NRTI, 0.9% to NNRTI, 1.8% to PI, and 1.0% to 2 or more classes of drugs.
Bennett reported trends of drug resistance over years. In 1997-98, 7.7% (n=39) of recent infections had drug mutations associated with resistance; in 1999 the rate was 12.5%, and in 2000-2001 it was 12.7%. In discussion with Bennett and Little both feel these rates are likely to increase as more individuals exposed to drug resistant virus are captured in these studies.
Among men with drug resistance the rate increased from 4.4% in 1997-98 to 11% in 1999 and 10.2% in 2000-2001 (p=0.04). Among women 6% had drug resistance in 1997-98, 6.3% in 1999, and 5.3% in 2000-2001. These changes were not significant. MSM were more likely to have drug resistance (11%) than IDUs (8%), and for heterosexuals (5%). Whites were more likely to have drug resistance than Blacks, and Hispanics. These differences might reflect less access to health care and appropriate drug treatment, access to support for adherence, and access to support for risk reduction.
Amongst women, IDUs were more likely to have resistance than heterosexuals, and Whites were more likely to have resistance than Blacks and Hispanics; but these differences were not significant.
Drug Resistance in San Francisco: prevalence of primary drug resistance remains high in San Francisco
Bob Grant (UCSF) reported a poster on trends in HIV drug resistance in SF. He reported declining NRTI primary resistance.
Abstract:
BACKGROUND: Increasing prevalence of primary, or transmitted, drug resistance has been reported in San Francisco, other North American cities and the UK, while decreasing trends in primary resistance have been observed in other settings. Interpreting divergent trends in primary drug resistance requires additional contemporaneous epidemiological information from the same geographical region.
|
|
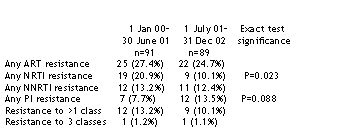
|
| |
METHODS: The Options project identifies persons in the San Francisco Bay Area with acute and early HIV-1 infection based on evolving serology. Drug resistance is assessed genotypically and phenotypically (TRUGENE, PhenoSense). Genotypic resistance was defined as any major protease inhibitor (PI) resistance-associated mutation and any nucleoside reverse transcriptase inhibitor (NRTI)- or non-NRTI (NNRTI)-associated mutation, including variants of
T215. RT V118I, a common polymorphism, was not considered in this analysis.
RESULTS: During an 18-month period between 1 July 2001 and 31 December 2002, 89 previously unreported recently infected and drug-naive persons were identified, and compared with 91 recently infected subjects identified during the previous 18 month period in the same geographical region. Among 314 genotypes from recently infected persons from 1996 to 2002, 9 of 25
(36%) of viruses with PI resistance and 16 of 28 (57%) of viruses with NNRTI resistance had no evidence of resistance to other drug classes. For comparison, isolated PI or NNRTI resistance was much less common among treated persons referred for clinical testing: 19 of 269 (7%) PI-resistant mutants and 17 of 69 (25%) of NNRTI-resistant mutants had no evidence of resistance to other classes. Among recently infected persons, seven of nine viruses with isolated PI resistance were PR L90M mutants and 11 of 16 viruses with isolated NNRTI resistance were RT K103N mutants.
CONCLUSIONS: The prevalence of primary drug resistance remains high in San Francisco, although transmission of viruses with three-class multidrug
resistance is rare. Although NRTIs are prescribed nearly universally in combination regimens, we observe declining proportions of transmitted viruses with NRTI resistance, and isolated PI or NNRTI resistance without NRTI resistance was common. Persons with NRTI resistance may have diminished infectiousness due to partial viral suppression, which is associated with continued NRTI activity and reductions in pro/pol replication capacity. In contrast, persons infected with RT K103N or PR L90M viral mutants
may have relatively preserved infectiousness if NRTI resistance is absent, as might occur with use of NRTI-sparing regimens arising due to partial adherence or to avoid toxicity.
The UK HIV Drug Resistance Database: development and use for
national surveillance
Deenan Pillay reported in a poster on the first output from the UK database on the prevalence of resistance in treatment-naive patients, patients with acute/recent infection (seroconverters), and treatment-experienced patients. A large HIV- drug resistance clinical database containing nearly 10,000 results has been established in UK. About 15% of drug naive patients who have had a resistance test are infected with virus with at least one key resistance associated mutation.
Abstract
Determining the clinical correlates of HIV drug resistance and estimation of resistance surveillance parameters require large databases. We have therefore
established a UK-wide non-proprietary collaboration of all virology laboratories and major clinical centres to pool resistance data and interpretation patterns,
together with pre- and post-test virological, clinical and therapeutic information. This clinical interface includes patients, represented within the UK Collaborative HIV Cohort study. Resistance test data is generated by Ôin house', VGI (Bayer) and ABI (Abbott) sequencing methodologies within UK diag-nostic laboratories, as well as by VIRCO.
As of March 2003, 9808 test results obtained since 1996 from 6692 patients have been entered. key (primary) resistance-associated mutations are based on
IAS-USA (2002). Prevalence data has been ascribed to the time periods 1996-1998, 1999-2000 and 2001-2003. Provisional findings are as follows. Of all
antretroviral-experienced patients, nucleoside analogue resistance (defined as at least one key mutation) remained stable (66, 71, 69%, respectively), whereas protease inhibitor (PI) resistance peaked in the 1999-2000 period (26, 32, 27%, respectively) and non-nucleoside reverse transcriptase inhibitor
(NNRTI) resistance has continued to rise (20, 40, 48%). The time trends for PI and NNRTI resistance are statistically significant (P<0.001). Resistance in
drug-naive individuals (n=1968), defined as any key mutation, appears to be levelling out after an initial rise (10, 16, 17%). When analysing the small subset
reported as acute/recent infections (n=157), no clear differences with non-acute drug-naive infections were noted.
We describe the first surveillance outputs from this UK database. Changes in the prevalence of PI and NNRTI resistance over time in treated patients mirror the changing use of these classes of drugs. In untreated patients we demonstrate continuing transmission of resistant viruses, the prevalence of which is not diminishing. Further, the similarity in prevalence between
acute and non-acute drug-naive patients suggests that testing in drug-naive patients/new diagnoses may be sufficient for monitoring transmission of drug resistance. Future studies from this database will include the resistance correlates of treatment response, the impact of HIV-1 subtypes on emergence of resistance and the identification of novel resistance-associated
mutations.
French Drug Resistance
French National Sentinel Survey of antiretroviral resistance in patients
with HIV-1 primary infection and in antiretroviral-naive chronically
infected patients in 2001-2002
ML Chaix reported in a poster on drug resistance in France. The objective of the study is to survey the frequency of genotypic antiretroviral resistance and the spreading of non-B subtypes in primary infected patients (2001-2002) and in antiretroviral-naive chronically infected patients (2001), and to compare it to results of previous years.
Plasma samples of 284 patients with acute HIV-1 infection and of 379 naive patients with chronic infection were tested for genotypic resistance.
Patients with acute infection were recruited within the French network on HIV-1 primary infection national survey, the PRIMO Cohort study (n=125), the different laboratories of the AC11 Resistance group (n=128), the INTERPRIM study (n=20) and the PRIMSTOP study (n=11). Patients with chronic infec-tion
were recruited in 23 AIDS care centres. Mutations were identified from the IAS-USA resistance panel (2002) and resistant virus was defined according to the
ANRS algorithm (www.hivfrenchresistance.org). For chronic infection, weighted statistical analyses were used to derived estimates of the percentage of patients with resistance mutations.
At inclusion, median CD4 cell counts and plasma HIV-1 RNA were 500 cells/mm 3 (38-1516) and 5.3 log copies/ml (1.78-7.57) for patients with primary infection and 385 cells/mm 3 (2-1280) and 4.5 log copies/ml (<1.70-6.94) for patients with chronic infection, respectively. Among the 284 primary infected patients, 32 (11%) (95% CI: 7.5-15.1) had baseline genotypic resistance to at least one antiretroviral drug. For patients with resistance mutations to one class of antiretroviral drug, the distribution was as follows:
to nucleoside reverse transcriptase inhibitors (NRTIs) in 20/284 [7%, (95% CI: 4.4-10.7)], to non-NRTIs (NNRTIs) in 7/284 [2.5%, (95% CI: 1.0-5)] and to protease inhibitors (PIs) in 12/284 [4.2%, (95% CI: 2.2-7.3)].
Five patients [2%, (95% CI: 1-4)] presented with virus mutations associated with multidrug resistance to two or three classes of antiretroviral drugs.
Among the 363 chronically infected patients with a median duration of known seropositivity of 6 months, the prevalence of virus with resistance to NRTI was 4/363 (1.1%; CI 95%: 0.0-2.1), resistance to NNRTI 1/363 (0.4%; CI95% 0.0-1.0), resistance to PI 4/363 (1.1%; CI95% 0.0-2.1), and resistance to at least 1 drug 6/363 (1.7%, CI 95% 0.2-3.2). In the chronically infected 52% of women were infected with non-B subtype compared to 232% of men. Among the individuals with primary infection 13% infected through homosexual or heterosexual transmission had a non-B subtype.
In France during the years 2001-2002, there was no significant progression of the frequency of resistant virus in these studied populations compared to previous surveys while the prevalence of non-B subtype was increasing.
|
|
| |
|
 |
 |
|
|