 |
 |
 |
| |
What's Wrong With ddI Plus Tenofovir?
|
| |
| |
Report 3 from the 14th ICAAC, October 30-November 2, 2004, Washington, DC
Written for NATAP by Mark Mascolini
Four studies—two of them reported at the 2004 ICAAC meeting—now expose serious shortcomings of regimens combining the nucleoside analog ddI and the nucleotide analog tenofovir disoproxil fumarate (TDF):
• A high risk of early failure in people with lofty viral loads and low CD4 counts
• A paradoxical drop in CD4 cells among people who control HIV replication
What's going on? And, more importantly, are there people who shouldn't take these two once-daily drugs together? Here's what's known so far.
Risk of early virologic failure
First-line regimens that combine the nonnucleoside efavirenz with two nucleoside or nucleotide reverse transcriptase inhibitors (NRTIs) have won favor with many clinicians for everyone except pregnant women, women who plan to become pregnant, or people who can't cope with the early neurologic side effects of efavirenz—insomnia, high-definition dreams, and so on.
Efavirenz plus two NRTIs looks as strong as lopinavir/ritonavir with two NRTIs, and lighter in long-term toxicities. Although people still debate the virologic response nuances of the 2NN study comparing efavirenz with the nonnuke nevirapine, efavirenz looked safer in that trial (1).
But earlier this year a small study detailed in a poster at the Resistance Workshop spotlighted one efavirenz regimen that didn't work well—once-daily efavirenz, ddI, and TDF (2). Daniel Podzamczer and Barcelona colleagues planned to compare this threesome with the same drugs plus lopinavir/ritonavir in people starting treatment with a high viral load. But the randomized plan came undone when Podzamczer noted a surprisingly high failure rate among people taking once-daily efavirenz, TDF, and ddI (at 200 or 250 mg daily depending on weight).
An unplanned analysis of 17 people in the three-drug group and 19 in the four-drug group who finished 3 months of therapy turned up no big differences in pretreatment viral load (median 142,740 copies/mL in the three-drug arm and 145,674 copies/mL in the four-drug arm) or CD4 count (respective medians of 195 and 162 cells/µL). But 14 people (43%) taking ddI/TDF/efavirenz had already met the study definition of virologic failure—less than a 2-log (100-fold) drop in viral load by month 3 or a 1-log (10-fold) rebound from the lowest load reached. Nobody in the four-drug group had a virologic failure.
Levels of ddI and TDF inside cells were similar in people with good and bad virologic responses to ddI/TDF/efavirenz. But compared with good responders, poor responders had significantly more advanced HIV disease:
• Median starting CD4 count: 73.5 versus 217 cells/µL (P = 0.01)
• Proportion with starting CD4 count below 200 cells/µL: 100% 37.5% (P = 0.031)
• Median starting viral load: 317,474 versus 118,611 copies/mL (P = 0.044)
• Proportion with AIDS or stage B3 disease: 100% versus 0% (P < 0.001)
Although these findings engendered a bout of head scratching at the Resistance Workshop, attendees avoided dire pronouncements about ddI/TDF, preferring to see the results confirmed—or contradicted—in a bigger study.
A bigger study at ICAAC confirmed them. Once more the trial had to stop early when researchers glimpsed a surprisingly sluggish virologic response in treatment-naive people starting ddI and TDF with efavirenz (3). And once more the poor responders started this triple combo with a high viral load.
Graeme Moyle and coworkers at London's Chelsea and Westminster Hospital randomized 44 people to start TDF with ddI/efavirenz and 36 to start 3TC with the other two drugs. People in the TDF group weighing more than 60 kg took 250 mg of ddI daily with food. The 3TC group took 400 mg of ddI daily without food. Pretreatment viral loads averaged about 100,000 copies/mL in both groups. People taking TDF started with a median 174 cells/µL, compared with 158 cells/µL in the 3TC arm, a nonsignificant difference.
The researchers disbanded the ddI/TDF group when an unplanned interim analysis showed a substantially worse response in people randomized to TDF than in those randomized to 3TC. As in Podzamczer's trial, all poor responders had a high pretreatment viral load and low pretreatment CD4 count.
Adherence measured by electronic bottle caps exceeded 99% in both groups. But after 4 weeks 4 people taking ddI/TDF/efavirenz (9%) did not lower their viral load at least 10-fold. After 12 weeks, 22 of 36 people (61%) taking that regimen had a viral load below 50 copies/mL, compared with 24 of 34 (71%) taking ddI/3TC/ efavirenz. At that point a last-observation-carried-forward analysis reckoned no virologic failures in the 3TC group and 5 among 41 people (12%) in the TDF group, a significant difference (P < 0.05). All 5 people with a virologic failure had a CD4 count below 200 cells/µL and a viral load above 100,000 copies/mL before treatment.
The Chelsea and Westminster team offered the "working hypothesis" that "a lower barrier to the development of resistance" may explain the hefty early failure rate in people who start ddI/TDF/efavirenz with a high viral load. Genotypic analysis spotted nonnucleoside mutations before nucleoside mutations in the 5 people with virologic failures.
In Podzamczer's study 5 of 6 people in whom this regimen flopped had the nonnucleoside G190S/E mutation with or without the more infamous K103N. This trend inspired speculation that G190S/E may be particularly treacherous for people also taking ddI, because research years ago found that G190S paves the way for L74V/I mutations, commonly linked to ddI (4). Changes at L74 did emerge in both Podzamcer's and Moyle's cohorts, but only 2 of 5 people in Moyle's group had G190S or E when ddI/TDF/efavirenz fizzled.
Perhaps the most one can venture at this point is that ddI/TDF/EFV doesn't bat down high viral loads fast enough to prevent speedy evolution of nonnucleoside mutations. Why, precisely, remains unclear. But the consistent correlation between high loads and early failure of the combo in two studies marks ddI/TDF/efavirenz as a poor choice for anyone with more than 100,000 copies/mL.
Paradoxical CD4 drops with ddI/TDF
Spanish researchers first noticed surprising CD4-cell swoons in people who switched to a ddI/TDF regimen with good viral control (5). This 302-person retrospective analysis charted slips in CD4 counts regardless of whether people took ddI/TDF with nevirapine, efavirenz, or lopinavir/ritonavir. More than half of those taking ddI/TDF lost more than 100 CD4 cells/µL during 48 weeks of treatment.
Most people in this study took 400/300 mg of ddI/TDF once daily, a ddI dose now recognized as too high because TDF drives up ddI levels and so raises the risk of side effects. Today's recommended ddI/TDF dose is 250/300 mg once daily for people weighing more than 60 kg.
But why should CD4 counts fall with a potent (even though side-effect-prone) 400/300 mg combination of ddI/TDF plus a nonnuke or boosted PI? Analysis of another Spanish cohort offered at ICAAC by Vincent Soriano (Carlos III Hospital, Madrid) shed more light on this conundrum (6).
Soriano and colleagues at other Spanish centers retrospectively studied 570 people taking a non-PI regimen from September 2002 to June 2003. Everyone kept viral replication under wraps for a year or more during that period. The study group included 298 people taking ddI/TDF, 18 as part of their first regimen and 280 to simplify a more complex combination. For comparison, Soriano also included 88 people taking only ddI, 44 taking only TDF, and 140 taking neither of those nukes.
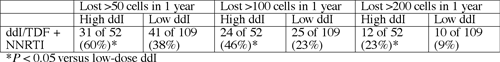
Among 173 people who took ddI/TDF as part of a simplification regimen with a nonnucleoside, 78 (45%) lost more than 50 cells/µL after 12 months of therapy, a significant difference from people taking non-ddI/TDF regimens (P < 0.001). Among 11 people taking ddI/TDF for simplification with one or more other nucleosides, 10 (91%) lost at least 100 cells/µL in 12 months, and 7 (64%) lost at least 200 cells/µL (P < 0.001 for both compared with non-ddI/TDF combos).
Partway through the study period, news came that the ddI dose should be trimmed from 400 to 250 mg daily when given with TDF. The Spanish clinicians followed that advice, and Soriano spelled out the results. Among people who switched to ddI/TDF and a nonnucleoside for simplification, the higher dose consistently caused bigger CD4 drops.
|
|
| |
| |
 |
|
| |
| |
In a multivariate analysis a longer time on high-dose ddI raised the risk of a CD4-cell drop during 12 months of treatment (P = 0.002). Soriano proffered these conclusions:
• People taking ddI/TDF tend to lose CD4 cells (measured in absolute number or percentage) despite complete virologic suppression.
• These drops appear about 6 months after treatment begins and worsen with time.
• CD4 declines are worse and come earlier when ddI/TDF is taken with another nucleoside rather than a nonnucleoside.
• People taking higher ddI doses lose more CD4 cells than those taking lower doses.
• These CD4 skids are not seen in people taking either ddI or TDF without the other drug.
Putting the pieces together (or trying to)
What explains these peculiar goings on? Theories have emerged, but they're not simple. For starters, Soriano explained, both ddI and TDF are adenosine analogs. (AZT and d4T are thymidine analogs, while 3TC and FTC are cytidine analogs.) The metabolites of ddI and TDF may exert a synergistic effect on mitochondria, the cells' powerpacks. That power drain may be particularly problematic for cells with normally high turnover rates, like CD4 T cells. Slower turnover would lead to lower CD4 counts.
Three nucleoside experts—Thomas Kakuda (Roche), Peter Anderson (University of Colorado), and Stephen Becker (University of California, San Francisco)—take this thinking a step farther in an article slotted for publication in the journal AIDS. They suggest a mechanism that explains not only CD4 plummets with ddI/TDF but also why triple-nuke regimens including these drugs fail virologically.
Kakuda and colleagues observe that TDF inhibits production of purine nucleoside phosphorylase (PNP), the compound that breaks down ddI for clearance (7). As a result, TDF boosts ddI concentrations. Scant PNP can boost levels of deoxyadenosine triphosphate (dATP) while decimating T-cell populations and function. Too much dATP and too little PNP may explain the T-cell toxicity (and lower CD4 counts) seen with ddI/TDF.
At the same time, PNP inhibition may upset pools dideoxynucleoside triphosphates (ddNTPs, the active inside-the-cell form of nucleoside or nucleotide analogs like ddI and TDF) and deoxynucleoside triphosphates (dNTPs, the natural nucleoside triphosphates inside cells that ddNTPs try to displace). Does this imbalance in dNTP/ddNTP pools, Kakuda and coauthors wonder, explain the virologic failure to ddI/TDF/3TC and ddI/TDF/abacavir?
Whatever the explanation, these T-cell findings in people taking ddI/TDF should prompt careful CD4 monitoring of anyone using those drugs.
Mark Mascolini writes about HIV infection (mailmark@ptd.net).
References
1. van Leth F, Phanuphak P, Ruxrungtham, et al. Comparison of first-line antiretroviral therapy with regimens including nevirapine, efavirenz, or both drugs, plus stavudine and lamivudine: a randomized open-label trial, the 2NN study. Lancet 2004:363:1253-1263.
2. Podzamczer D, Ferrer E, Gatell JM, et al. Early virologic failure and occurrence of resistance in naive patients receiving tenofovir, didanosine and efavirenz. XIII International HIV Drug Resistance Workshop. June 8-12, 2004. Tenerife. Abstract 156.
3. Moyle G, Maitland D, Hand J, et al. Early virological failure in persons with viral loads >100,000 cps/ml and CD4 counts <200/mm3 receiving ddI/tenofovir/efavirenz as initial therapy: results from a randomised comparative trial. 44th Interscience Conference on Antimicrobial Agents and Chemotherapy (ICAAC). October 30-November 2, 2004. Washington, DC. Abstract H-566.
4. Kleim JP, Rosner M, Winkler I, et al. Selective pressure of a quinoxaline nonnucleoside inhibitor of human immunodeficiency virus type 1 (HIV-1) reverse transcriptase (RT) on HIV-1 replication results in the emergence of nucleoside RT-inhibitor-specific (RT Leu-74-->Val or Ile and Val-75-->Leu or Ile) HIV-1 mutants. Proc Natl Acad Sci USA 1996;93:34-38.
5. Negredo E, Molto J, Burger D, et al. Unexpected CD4 cell count decline in patients receiving didanosine and tenofovir-based regimens despite undetectable viral load. AIDS 2004;18:459-563.
6. Barrios A, Negredo E, Rendon A, et al. Paradoxical CD4+ T-cell decline in patients with complete virus suppression under tenofovir plus didanosine combinations. 44th Interscience Conference on Antimicrobial Agents and Chemotherapy (ICAAC). October 30-November 2, 2004. Washington, DC. Abstract H-1132.
7. Ray AS, Olson L, Fridland A, et al. Role of purine nucleoside phosphorylase in interactions between 2',3'-dideoxyinosine and allopurinol, ganciclovir, or tenofovir. Antimicrob Agents Chemother 2004;48:1089-1095.
|
| |
|
 |
 |
|
|