 |
 |
 |
| |
Safety of Double Dose Pegasys (REPEAT Study)
|
| |
| |
Reported by Jules Levin
"Planned interim safety analysis of patients enrolled in the randomized, international REtreatment with PEgasys in pATients not responding to prior peginterferon alfa-2b/ribavirin combination therapy (REPEAT) study"
P. Marcellin,1 B. Freilich,2 P. Andreone,3 C. Brandao-Mello,4 A.M. Di Bisceglie,5 R. Rai,6 D. Jensen7
1Hôpital Beaujon, Clichy, France; 2Baptist Medical Center, Kansas City, MO, USA; 3University of Bologna, Bologna, Italy; 4University of Rio de Janeiro, Rio de Janeiro, Brazil; 5St Louis University, St Louis, MO, USA;
6Johns Hopkins University School of Medicine, Baltimore, MD, USA; 7Rush University Medical Center, Chicago, IL, USA
Presented at the 56th Annual Meeting of the American Association for the Study of Liver Diseases: 2005 November 11–15: San Francisco, CA, USA.
This research was funded by Roche, Basel, Switzerland.
Patrick Marcellin reported these data in a poster at AASLD Nov 2005 in SF.
Study Objective:
We report outcomes from a protocol-defined week 12 interim safety analysis of the REPEAT study in order to evaluate the safety profile of the high-dose induction regimen (peginterferon alfa-2a [40KD] 360 _g/week plus ribavirin) compared with the standard-dose regimen (peginterferon alfa-2a [40KD]
180 _g/week plus ribavirin). Patients were recruited between September 2003 and March 2005.
All patients were previous non-responders to treatment with standard doses of pegylated interferon alfa-2b (12KD) (PegIntron) plus ribavirin, given for >12 weeks. Treatment with pegylated interferon alfa-2b (12KD) plus ribavirin must have been discontinued for >12 weeks prior to enrollment.
A prospectively defined analysis of efficacy and safety outcomes was planned after all randomized patients had completed 12 weeks’ treatment. The incidences of adverse events, serious adverse events and deaths, along with laboratory abnormalities and dose reductions, were recorded for each treatment arm.
REPEAT is an ongoing randomized, multinational, phase III, open-label, parallel group trial.
REPEAT Study Design
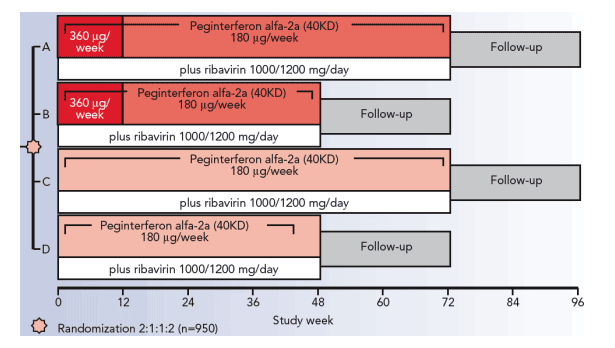
- Arm A: peginterferon alfa-2a (40KD) 360 _g/week during weeks 1–12 followed by peginterferon alfa-2a (40KD) 180 _g/week during weeks 13–72 (total treatment duration 72 weeks)
- Arm B: peginterferon alfa-2a (40KD) 360 _g/week during weeks 1–12 followed by peginterferon alfa-2a (40KD) 180 _g/week during weeks 13–48 (total treatment duration 48 weeks)
- Arm C: peginterferon alfa-2a (40KD) 180 _g/week during weeks 1–72 (total treatment duration 72 weeks)
- Arm D: peginterferon alfa-2a (40KD) 180 _g/week during weeks 1–48 (total treatment duration 48 weeks).
Patients were randomized according to geographic region, cirrhotic state and HCV genotype (1 vs non-1) to one of the four treatment regimens below. All patients received a weight-adjusted dose of ribavirin (1000/1200 mg/day).
As the primary aim of this analysis was to evaluate the safety of high-dose induction peginterferon alfa-2a (40KD) (360 _g/week) compared with the standard dose (180 _g/week), data from patients in Arms A and B (high-dose induction) were combined and compared with data from patients in Arms
C and D (standard dose) combined.
Only data from patients who received 1 or more dose of study medication and had 1 or more post-baseline safety assessment were included in the analysis (safety population).
BACKGROUND
Since the introduction of pegylated interferon combination therapy, sustained virological response (SVR) rates for patients with chronic hepatitis C have increased substantially compared with conventional interferon plus ribavirin combination therapy.
Despite this improved response rate, data indicate that almost half of all patients treated with pegylated interferon alfa-2b (12KD) plus ribavirin fail to achieve an SVR.
At present, treatment options for patients who have failed an initial course of pegylated interferon plus ribavirin combination therapy are limited.
Data from a small cohort study (n=31) have indicated that a proportion of patients who failed to respond to pegylated interferon alfa-2b (12KD) plus ribavirin can achieve an SVR when retreated with peginterferon alfa-2a (40KD) (PEGASYS) 180 _g/week plus ribavirin (COPEGUS®) 1000/1200 mg/day.
Retreatment studies among previous non-responders to conventional interferon-based regimens have previously suggested that the use of induction doses and/or a prolonged duration of treatment may be effective in improving response rates seen during retreatment.
The REPEAT (REtreatment with PEgasys in pATients not responding to prior pegylated interferon alfa-2b/ribavirin combination therapy) study has been initiated to compare the efficacy and safety of peginterferon alfa-2a (40KD) plus ribavirin given for 48 or 72 weeks, including a high-fixed dose induction and extended treatment regimens, in patients who were non-responders to previous
treatment with pegylated interferon alfa-2b (12KD) plus ribavirin.
RESULTS (see tables & graphs following text)
Author Conclusions:
When complete, data from the REPEAT study will provide valuable information on the potential benefits of using a fixed induction dose of peginterferon alfa-2a (40KD) (PEGASYS) and an extended duration of treatment among non-responders to treatment with pegylated interferon alfa-2b (12KD) plus ribavirin.
The results of this prospectively defined, protocol-planned interim analysis demonstrate that the safety profile of peginterferon alfa-2a (40KD) combined with ribavirin (COPEGUS) does not appear to be compromised in patients receiving a high fixed induction dose (360 ug/week), compared with the standard treatment regimen (180 ug/week plus ribavirin).
The data revealed no substantial increase in the incidences of adverse events, serious adverse events or laboratory abnormalities among patients treated with the high-dose induction of peginterferon alfa-2a (40KD).
The incidence of adverse events and serious adverse events in patients with cirrhosis was similar to that seen in the overall population. Specifically, the overall frequency of adverse events and serious adverse events was similar in cirrhotic patients in the standard-dose and the high-dose induction treatment groups.
Results from the week-12 protocol-planned efficacy analysis, also presented at this congress indicate that high induction doses of peginterferon alfa-2a (40KD) appear to be more effective than standard doses in delivering an early virological response. SVR outcomes, the primary study end-point, are awaited with interest.
SAFETY
Premature withdrawals
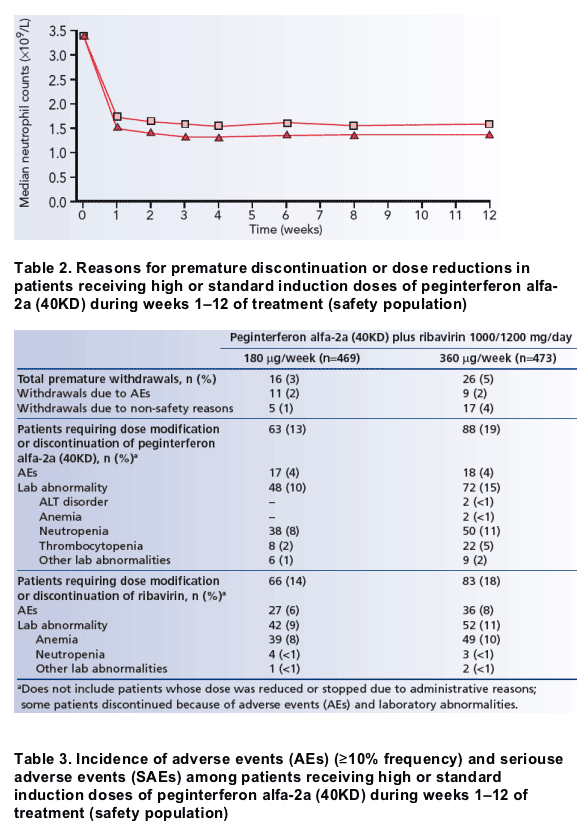
During the first 12 weeks of the study, a slightly higher proportion of patients receiving high-dose compared with standard-dose peginterferon alfa-2a (40KD) discontinued treatment prematurely (Table 2).
Overall, a similar proportion of patients in the high-dose induction group and the standard-dose group discontinued because of adverse events (Table 2). The proportion of patients with cirrhosis who withdrew from treatment because of adverse events was similar to that in the overall patient population (3% in the high-dose induction group, 2% in the standard-dose group).
Withdrawals due to non-safety reasons in the overall patient population were more common in the high-dose induction group than in the standard-dose group (Table 2).
Dose reductions and discontinuation of study medication
A greater proportion of patients in the high-dose induction group required dose reductions or discontinuation of peginterferon alfa-2a (40KD) due to adverse events and/or laboratory abnormalities (Table 2). The frequency of dose reductions or discontinuation of peginterferon alfa-2a (40KD) due to neutropenia was slightly higher in the high-dose induction vs the standard-dose group
(11% vs 8%).
Ribavirin dose modifications or discontinuations occurred in a similar proportion of patients receiving high-dose and standard-dose peginterferon alfa-2a (40KD) (18% vs 14%) (Table 2).
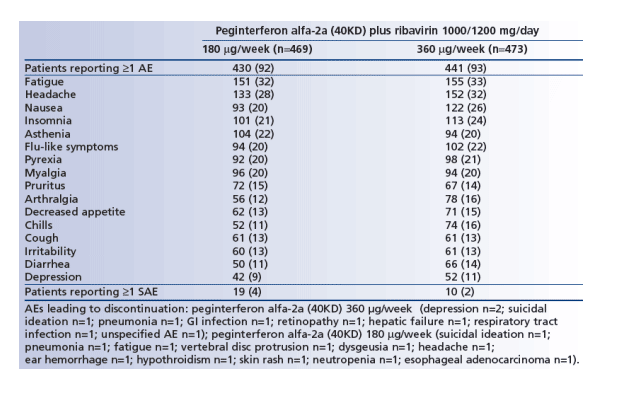
Adverse events, serious adverse events and deaths
The overall frequency of adverse events was similar between the two groups (Table 3). Fatigue, headache and nausea were the most common individual adverse events reported, occurring in a similar proportion of patients in both groups analyzed.
Serious adverse events were uncommon, occurring in 2% of patients in the high-dose induction group and 4% of those treated with standard-dose peginterferon alfa-2a (40KD) (Table 3).
The incidence of adverse events and serious adverse events in patients with cirrhosis was similar to that seen in the overall patient population; 94% and 91% of cirrhotic patients in the high-dose induction and standard-dose groups experienced ≥1 adverse event, while 3% and 5% of cirrhotic patients, respectively, experienced ≥1 serious adverse event.
No deaths were reported during the first 12 weeks of the study.
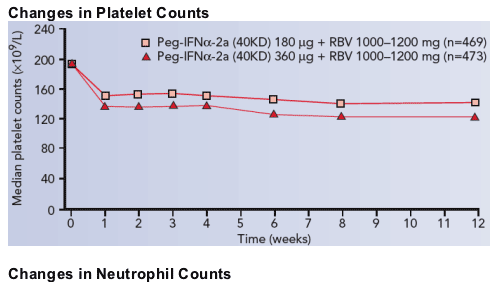
Changes in hematological parameters
Mean platelet and neutrophil counts decreased during treatment in both groups (Figure 2). Only one patient in the standard-dose group discontinued treatment due to neutropenia (Table 3).
In the overall patient population, 3% of patients in the standard-dose group and 6% of patients in the high-dose induction group had a lowest platelet count of 20 to <50 - 109 cells/L. This was compared with 8% and 11%, respectively, of patients with cirrhosis. No patient, cirrhotic or non-cirrhotic, withdrew from treatment due to thrombocytopenia.
|
|
| |
| |
|
 |
 |
|
|