 |
 |
 |
| |
"VALOPICITABINE (NM283) ALONE & IN COMBINATION WITH PEG-INTERFERON IN PATIENTS WITH GENOTYPE-1 CHRONIC HEPATITIS C:
Preliminary results from an ongoing phase II, multicenter study"
|
| |
| |
Reported by Jules Levin
DDW, Chicago, May 2005
Several new drugs for HCV are in clinical trials in HCV+ patients and appear promising including NM283, a polymerase inhibitor administered orally, and VX-950, an HCV protease inhibitor. NM283 plus peg-IFN may provide a treatment option for previous pegIFN+RBV partial responders. The protease inhibitor is still in a preliminary liquid formulation, as the initial study in patients was first just reported, and the company Vertex, is working on a tablet formulation. In addition, for the first time data was publicly reported, at DDW, for a new HCV drug called CPG 10101, or Actilon. Actilon is a member of a new class of investigational medicines known as TLR Therapeutics(TM) being developed by Coley Pharmacueticals for the treatment of major medical conditions including cancers, infectious diseases, allergy and asthma. TLR Therapeutics target Toll-like receptors (TLRs) which act as immune system sentinels that recognize the distinct molecular patterns characteristic of foreign pathogens. Coley & the presenter at DDW, Dr Bruce Bacon, believe that Actilon stimulates TLR9, targeting dendritic cells and B cells, to induce both early and long term immune responses. The short-term innate immune response is thought to drive rapid reductions in viral load in the blood. Longer term, Actilon is thought to promote virus-specific adaptive immunity, including strong T cell responses, to provide sustained anti-viral effects. In a phase Ib study in 42 adult patients with chronic HCV who previously failed IFN/RBV & most had genotype 1, 20mg of Actilon administered twice weekly by subcutaneous injection HCV viral load achieved a maximum log decrease of 1.4 within 5 weeks. Actilon appeared safe & tolerable. I will report more extensive data from this study. These drugs are in relatively early stages of clinical development although NM283 development is ahead of these other two drugs. But, it will be several years before NM283 or these others may reach availability to patients. And of course, that is if development runs smoothly without safety & activity problems.
NM283 Study
At DDW on May 17, 2005 in Chicago, Maribell Rodriguez reported an update with new data on ALT from the study of NM283.
AUTHOR SUMMARY:
NM283 showed marked antiviral activity:
--8/16 patients receiving NM283+PegIFN were PCR-negative at week 24. 3/16 patients are still pending between weeks 2 & 24.
--Mean HCV RNA (viral load) declined -4.5 log IU/mL (range: -2.33 to -6.2 log by week 24.
--all 9 patients completing week 24 had >=2 log reductions in viral load
--8/9 patients were below the LLOQ for Amplicor PCR (<600 IU/mL)
--6/9 patients were below LLOD for Taqman PCR (<10 IU/mL)
--4/5 patients had detectable PCR at week 12 but had multi-log reductions after week 12.
--Rodriguez said the kinetics of response to NM283+PegIFN may be different from the kinetics of response to pegIFN+RBV, perhaps synergistic.
--no HCV RNA "breakthroughs" to date, with data to 24 weeks; viral genotyping underway
--good tolerability: negligible hemotologic side effects—possible safety advantage without ribavirin
--large phase III studies are planned for prior treatment failures & treatment-naïve patients
Here is some background information provided by Dr Rodriguez. In phase I/II study, NM283 reduced HCV RNA, viral load, by a mean 1.2 log in 2 weeks at optimal dosing. 87% of patients had previously failed IFN therapies. In vitro, NM283 & IFNa show synergistic antiviral effects in BVDV model. This data supports early clinical investigation of NM283 plus peg-IFNa, to maximize antiviral efficacy & minimize resistance.
This study was designed initially to take a short-term look at safety, antiviral activity & pharmacokinetics for 28 days. Due to encouraging early data, treatment was extended to 12, then 24, and finally to 48 weeks via protocol ammedments, with FDA & investigator agreement. The study extension provides exploratory longer-term efficacy & safety data for the combination of NM283 plus Peg-IFN, prior to controlled phase IIb-III trials. Treatment extension required continued tolerance & virologic response:
>=2 log reduction from baseline at week 12
PCR negative at week 24.
The study is ongoing to one year, 12 & 24 week data were presented at DDW.
The trial is an open-label PK interaction & safety study enrolling 30 treatment-naïve adults with compensated chronic hepatitis C with HCV RNA >5 log & alt <5 times the upper limit of normal (ULN). Eligible patients received NM283 (n=12) vs NM283 plus peg-IFNa-2b (n=18). NM283 was administered initially at 400 mg/day and then increased to 600mg/day & then to 800mg/day to day 8, then 800mg/day to day 28.
The majority of patients were Latino because Dr Rodriguez enrolled many patients in San Juan. Baseline mean HCV RNA was 6 log & ALT was 63-70 IU/mL.
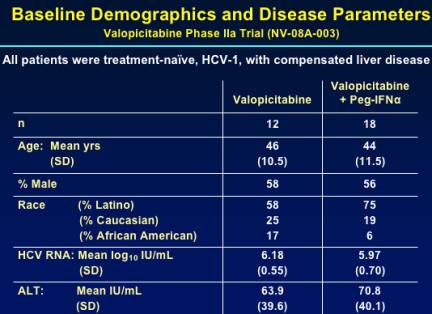
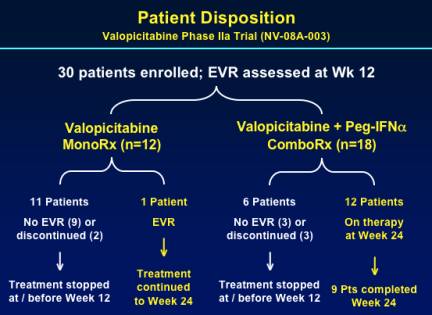
30 patients were enrolled; EVR was assessed at week 12. Only 1 patient on NM283 monotherapy achieved EVR. 9 of 18 patients receiving combination therapy completed 24 weeks.
NM283 appears safe & tolerable in the study. There were no serious adverse events or dose-limiting toxicities.
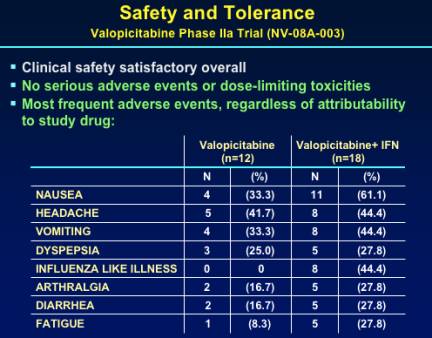
SAFETY & TOLERABILITY
Typical IFN side effects, no unexpected side effects.
Nausea, vomiting common in both NM283 & combination treatment arms, reported to be transient, probably related to NM283 & IFN. Onkly 1 patient discontinued for AE (decreased libido due to IFN). Only 1 grade _ lab abnormality observed during treatment:
--grade 3 ANC (absolute neutrophil count) reduction (620/mm3) at day 11 in a patient receiving combination therapy
--ANC returned to baseline levels at day 15, remained stable for the duration of treatment
--no dose interruption or treatment modification required
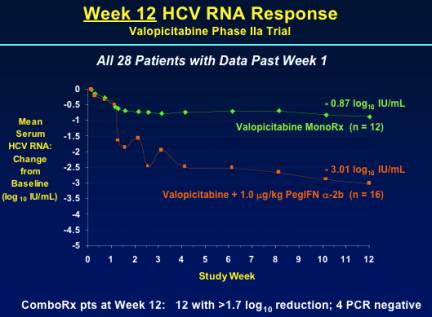
WEEK 12 HCV RNA RESPONSE
Mean HCV RNA reduction was -3.01 log IU/mL in the combination arm (n=16) & -0.87 IU/mL in the NM283 monotherapy arm (n=12). 12 patients in the combination arm had >1.7 log reduction; 4 were PCR negative.
WEEK 24 HCV RNA RESPONSE
Viral load was reduced by 1.9 log IU/mL from baseline in the patient receiving monotherapy (n=1). In the combination arm the mean HCV RNA reduction was -4.5 log IU/mL.As you can see from the graph the mean viral load was below Amplicor LLOD of 50 IU/mL.
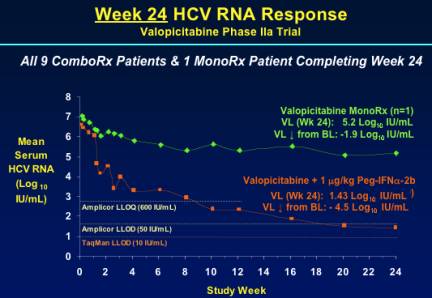
As you can see from the graph below several patients had viral load reduced to low levels within 12-16 weeks after beginning therapy but several patients became responders between weeks 12 to 24. At week 24, 8/9 patients had <600 IU/mL, 7/9 had <50 IU/mL, and 6 of 9 patients had <10 IU/mL using the sensitive TaqMan assay.
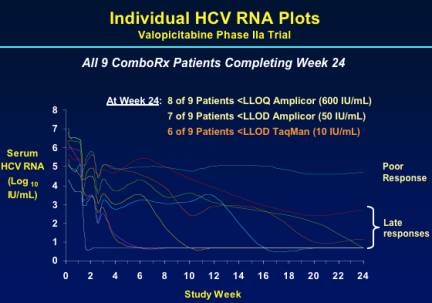
In the 5patients receiving combination treatment who were still PCR+ at week 12, 4 continued to have responses after week 12.
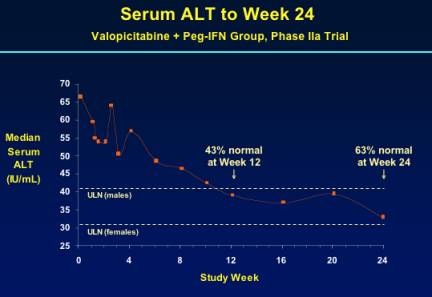
Median ALT was 65-70 at baseline & 43% of patients normalized ALT at week 12 & 63% were normal at week 24.
|
|
| |
| |
|
 |
 |
|
|