 |
 |
 |
| |
Heart Disease in HIV: Carotid IMT, measuring plaque in arteries
|
| |
| |
Reported by Jules Levin
Several studies were reported at CROI (Feb 2006, Denver) using Carotid IMT as an evaluation of risk for cardiovascular disease.
Long Term HIV Infection as an Independent Risk Factor for Early Atherosclerosis
..... In HIV positive individuals, internal carotid artery plaques were significantly more common ......The data from this study suggests long-term HIV infection may be an independent risk factor for early atherosclerosis as measured by ultrasonic IMT assessment.
Christoph Stephan*1, A Carlebach1, A Haberl1, M Bickel1, M Lorenz2, A Harmjantz2, A Buehler2, M Sitzer2, and S Staszewski1
1HIV Treatment and Res Unit and 2JW Goethe Univ Hosp, Frankfurt, Germany
Background: The impact of HIV infection on atherosclerosis has not yet been fully investigated. We investigated the relationship between HIV status and early ultrasonic atherosclerosis signs in a 1:4 case-control design in the context of conventional vascular risk factors.
Methods: We assessed the individual risk factors for atherosclerosis, blood pressure (BP), cholesterol levels and measured intima media thickness (IMT) of the carotid artery system by B-mode ultrasound in 292 consecutive HIV patients. The findings were compared to randomly selected 1168 control individuals from the Carotid Atherosclerosis Progression Study (CAPS), who were matched for age and sex.
Results:
The first positive HIV test among the positive patients was in median 10.6 years ago from IMT assessment (95%CI. +/- 5.2y), CDC-class B (40%) was more frequent than C (28%), mean CD4 cell count was 439/µL (95%CI: +/- 243) and most patients were virologically suppressed (74.7% <400copies/mL or 49.3% <20copies/mL).
Controls were significantly more obese (BMI 27.5 vs. 25.1), had higher cholesterol levels (5.87 vs. 5.41mmol/L), systolic (131.7 vs. 122.8mmHg) and diastolic (81.1 vs. 75.0mmHg) BP. HIV patients were taking significantly more antihypertensive (14.4% vs. 10.6%) and lipid lowering (18.5% vs. 5.1%) drugs, as was cigarette smoking (27.0 vs. 14.2 pack years).
In HIV positive individuals, internal carotid artery plaques were significantly more common (12.3% vs. 7.8%, p=0.0312) in unadjusted models.
The common carotid IMT was 5.0% (95% confidence interval: [2.3, 7.8%], p=0.0002) or 0.036mm [0.012, 0.060mm] (p=0.0034) higher in HIV positives when adjusted for multiple risk factors.
In the carotid bifurcation, the IMT values were 19% [14, 24%] or 0.180mm [0.123, 0.238mm] higher in HIV patients (p<0.0001), respectively. This effect was not significant for internal carotid IMT, but showed a trend (IMT 4.3% [-0.7, 9.5] or 0.037mm [-0.016, 0.089] higher, p=0.0905 or 0.1721).
Conclusions: The data from this study suggests long-term HIV infection may be an independent risk factor for early atherosclerosis as measured by ultrasonic IMT assessment. Assuming a similar risk-ratio would be found in large population-based trials, the observed IMT elevation suggests that vascular risk is 4-14% and the 'vascular age' is 4-5 years higher in HIV-positive subjects. Further studies are warranted to find an approach to specific pathophysiology and to evaluate the role of HIV-specific medication.
Increased Carotid Intima-media Thickness in HIV-infected Patients Is Associated with Increased C-reactive Protein Levels and Increased Cytomegalovirus Specific T Cell Activation
... HIV-infected subjects had thicker carotid IMT as compared to HIV-negative controls..... HIV infection may accelerate atherosclerosis through CMV-induced immune responses and/or inflammation....
Priscilla Hsue*1,2, P Hunt1,2, D Waters1,2, E Sinclair3, J Martin1,2, A Franklin2, R Hoh1,2, and S Deeks1,2
1Univ of California, San Francisco, US; 2San Francisco Gen Hosp Med Ctr, CA, US; and 3Gladstone Inst of Virology and Immunology, Univ of California, San Francisco, US
Background: HIV patients are at increased risk for myocardial infarction. However, the mechanism accounting for this increased risk remains unclear. Since cytomegalovirus (CMV) infection has been associated with accelerated atherosclerosis in the heart transplant population and CMV-specific immunity is altered by HIV disease, we hypothesized that CMV would be associated with increased atherosclerosis in HIV patients.
Methods: We measured high sensitivity C-reactive protein (hs-CRP), T cell activation, and carotid artery intima-media thickness (IMT) by ultrasound in 93 HIV-infected patients and in 37 uninfected controls. HIV-infected subjects were required to be on a stable ART regimen or no ART for 1 year. CMV-specific immune responses were defined as the proportion of CD4+ and CD8+ T cells which produced interferon-gamma in response to CMV pp65 protein. T cell activation was based on the mean density of CD38 on T cell subsets.
Results:
The median age of the HIV-infected patients was 46 years and 75 (81%) were male. The median CD4 count was 354 cells/mm3, and 57% of patients had a viral load <75 copies/ml. Most patients (92%) were on antiretroviral treatment.
Compared to HIV-negative controls, HIV-infected subjects had higher median baseline carotid IMT (0.95 mm vs 0.68 mm, p <0.001).
This difference remained significant after controlling for all traditional risk factors.
Compared to controls, HIV-infected patients had higher median levels of hs-CRP (1.1 mg/L vs 0.8 mg/L, p = 0.05), higher levels of T cell activation (745 vs 282 CD38 molecules/CD8+ T cell, P <0.0001), and higher CMV-specific T cell responses (2.09% vs 0.056% CMV-specific CD8+ T cells, p <0.0001). hs-CRP levels and CMV-specific T cell responses, but not T cell activation were independently associated with higher carotid IMT.
Although HIV infection was independently associated with carotid IMT after adjustment for CRP (p <0.001), this effect was no longer significant after adjustment for CMV-specific T cell responses (p = 0.11).
Conclusions: HIV-infected subjects had thicker carotid IMT as compared to HIV-negative controls. Their hs-CRP levels and CMV-specific T cells responses were also higher, and both of these measures were independently associated with carotid IMT. HIV infection may accelerate atherosclerosis through CMV-induced immune responses and/or inflammation.
3-year Follow-up of Carotid Intima-media Thickness in HIV-infected and Uninfected Adults: ACTG 5078
Judith Currier*1, M Kendall2, K Henry3, F Torriani4, J Conley5, B Alston-Smith6, M Basar7, K Mickelberg8, Y Li9, H Howard9, and ACTG 5078 Study Team
1Univ of California CARE Ctr, Los Angeles, US; 2Harvard Sch of Publ Hlth, Boston, MA, US; 3Univ of Minnesota, Minneapolis, US; 4Univ of California, San Diego, US; 5Univ of Washington, Seattle, US; 6Div of AIDS, NIAID,, NIH, DHHS, Rockville, MD, US; 7Frontier Sci & Tech Res Fndn, Buffalo, NY, US; 8Univ of Pennsylvania, Philadelphia, US; and 9Univ of Southern California, Los Angeles, US
CURRIER'S SUMMARY:
-- Neither HIV+ status or PI use resulted in a significant difference between the baseline or three year longitudinal caroitid IMT.
-- fasting blood sugar and insulin values did appear to be important predictors of IMT change in all models.
--for HIV+ subjects there was a suggestion that use of ritonavir, longer duration of PI use, fasting glucose, and higher HS-CRP were associated with increased rates of IMT progression in univariate & multivariate models of baseline variables. But Currier added in her talk that while statistically significant the magnitude of the changes are very small. For example, each additional 48 weeks of PI exposure was associated with a 0.0004mm increase in IMT, which may or may not be clinically significant.
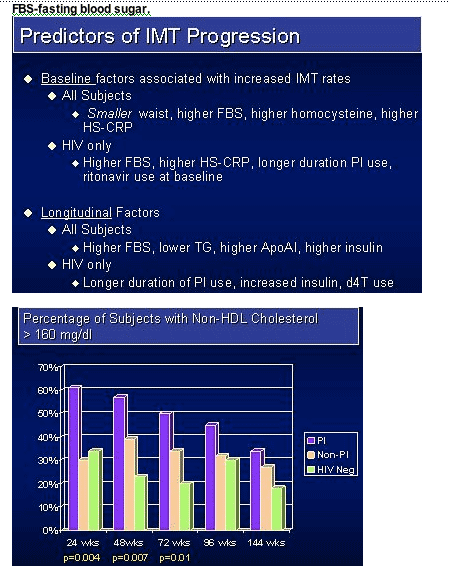
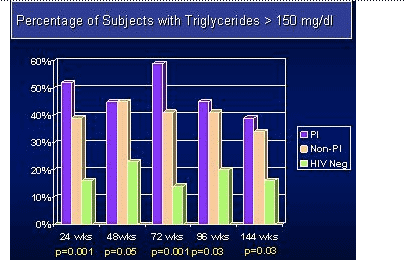
-- TG and non-HDL cholesterol levels, while higher at baseline in the PI groups appeared to decrease over time.
CONCLUSIONS BY CURRIER:
-- our study design, with closely matched groups, tried to isolate the impact of PI use and HIV infection on the risk of artherosclerosis.
-- while rates of IMT progression in the HIV+/PI hroup were greater than the HIV+/PI naive group this difference did not reach statistical significance in our small study.
-- our study was not designed to identify differences between drugs within the PI class, however it suggests that ritonavir containing PI regimens may differ in their impact on carotid IMT.
-- future studies that attempt to quantify the relationship between HIV infection, specific ARV agents and artherosclerosis will need to carefully control for a myriad of factors, all of which are likely to vary over time.
During the Q&A Currier was I think asked if a subgroup of patients on PI with elevated lipids could have had faster progression of CIMT but not picked up in this study and I think she responded or alluded that that this study was not powered to pick up differences between PIs or PI-induced effect on lipids or artherosclerosis.
Background: Cross-sectional studies reveal a high prevalence of atherosclerosis in patients on ART. Uncontrolled studies suggest that the progression of atherosclerosis is accelerated in HIV-infected adults. We followed individuals for 3 years using carotid intima-media (IMT), a technique that correlates with coronary artery atherosclerosis and clinical cardiovascular events.
Progression of carotid IMT is predictive of clinical cardiovadculat events in epidemiological studies. Results from several CIMT studies have shown mixed results between rates & risk factors for progression. Hsue reports a 10-fold increase in rates of progression between HIV+ patients compared to controls and lower nadir CD4 was associated with an increased risk. Recent study by Mercie (HIV Medicine 2005) reported lower rate of progression in HIV+ than had previously been seen. Mercie reported higher CD4 at baseline & smoking were associated with greater risk for progression, but no association with ARV & progression.
Methods: Groups of 3 subjects (triads) who were matched on the following characteristics: age, sex, race/ethnicity, smoking status, blood pressure status, and menopausal status were enrolled.


Group I: HIV+ subjects with continuous use of PI therapy for 2 years;
Group II: HIV+ subjects without prior protease inhibitor (PI) use;
Group III: HIV-neg.
Subjects were excluded if they had or had a family history of coronary artery disease, diabetes mellitus, uncontrolled hypertension, or a body mass index >30. Ultrasonographers at 6 sites sent standardized IMT images at weeks 0, 24, 48, 72, 96, and 144 to a central reading site for measurement. Yearly change in carotid IMT was compared within the HIV-infected groups and between the HIV+ and uninfected groups in a matched analysis. The study had 80% power to detect a clinically relevant difference of 0.02 mm/y change in carotid IMT.
Previously reported baseline results where no difference was seen between groups after an average of 217 weeks of exposure in the PI group.
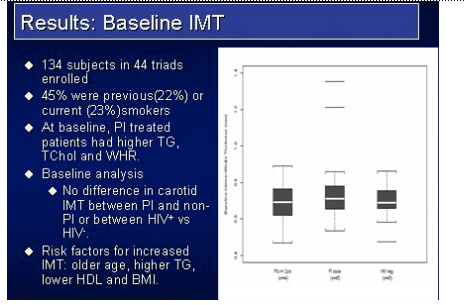
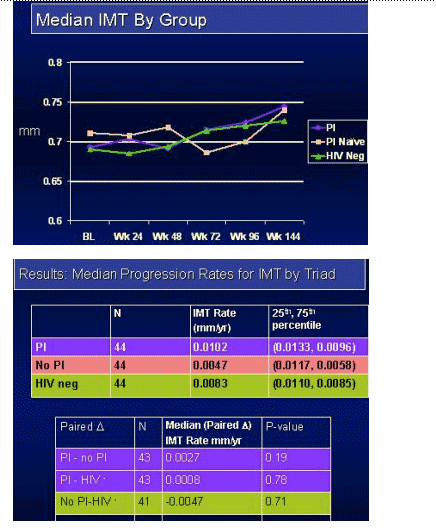
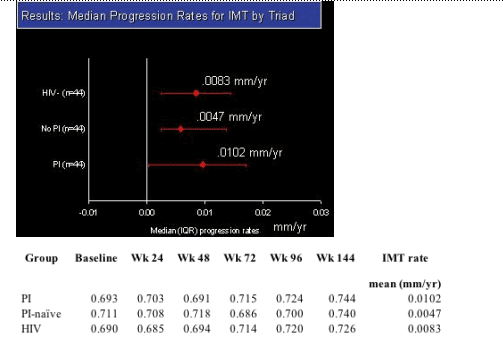
Results: The study accrued 134 subjects in 45 triads. At baseline, more PI-treated subjects had triglycerides > 300 mg/dL (23%), total cholesterol >200 mg/dL (64%) and met the Natl Cholesterol Education Program definition of metabolic syndrome (32%) compared to the HIV non-PI & HIV-neg subjects. Within the PI group 34% received ritonavir. Median follow-up was 152 weeks. Median IMT values (mm) and mean progression rates are shown in the table. Within-triad matched analysis of the IMT yearly rates showed no statistically significant differences between the PI-treated & PI-naive groups (p=0.19), between the PI-naive and HIV-neg group (p=0.78), or between the combined HIV groups and the HIV controls (p=0.71).

|
|
| |
| |
|
 |
 |
|
|