 |
 |
 |
| |
|
Phase II Proof of Concept Study of celgosivir in combination woth peginterferon alfa-2a and ribavirin in chronic hepatitis C genotype-1 non responder patients
|
| |
| |
Reported by Jules Levin
EASL, April 11-15, 2007, Barcelona
Kelly Kaita from University of Manitoba reported in the very last session today Sunday at EASL on using chestnut to cure HCV.
Celgosivir: a new agent for the treatment of chronic HCV infection.
--first in class oral antiviral compound that targets host enzyme.
--safety profile established in over 600 subjects.
--in vitro synergy demonstrated with interferon, ribavirin and other anti-HCV drugs in development.
Celgosivir is a 6-0-butanoyl ester derivative of castanosperine; it's derived from the Australian chestnut. It's a potent inhibitor of a-glucosidase I, which is a host enzyme required for viral assembly, release and infectivity.
Pharmacology: Celgosivir rapidly converts to castanospermine in the body. It can be detected in serum within 5 minutes; T-max is about 1 hour; terminal half-life about 19-23 hours; there are dose proportional (10-400 mg range) kinetics; this drug is excreted predominantly unchanged exclusively in the urine; to date, no other metabolites have been detected.
STUDY DESIGN
This was a randomized, double-blind, multi-center study. Treatment period is 12 weeks. Patients are genotype 1 treatment-experienced with prior non-response or partial response to a combination of peginterferon and ribavirin.
Patients were randomized to one of three treatment groups:
--PRC: peginterferon alfa-2b + ribavirin + celgosivir 400 mg
--PC: peginterferon alfa-2b + celgosivir 400 mg
--PR: peginterferon alfa-2b + ribavirin (active control) It will however be years before interferon or ribavirin may be eliminated from a regimen, and perhaps it will not be possible to eliminate one or both of these drugs
57 patients were enrolled. 36 were non-responders (prior response <2 log IU/mL decrease in HCV RNA, n=36). Within this group was a subgroup of null responders, the most difficult to treat patients with current therapy (prior decrease <0.4 log IU/mL decrease in HCV RNA, n=11). There was a group, n=21, of partial responders (prior response of >2 log IU/mL decrease in HCV RNA, achieved an Early Viral Response, but did not become negative and were positive at week 24). This group included 2 relapsers.
DEMOGRAPHICS
36 non-responders were enrolled: 15 in the PRC group; 11 in PC, 10 in PR.
These patients were comparable with respect to race, age, weight and baseline ALT.
Mean HCV RNA of about 6.7 log IU/mL in each treatment group.
The mean response to prior therapy was 0.71, 1.14 and 0.82 log IU/mL for PRC, PC and PR treatment groups, respectively.
Of the 11 null responders included in the non-responder group: 7, 2, and 2 patients in the PRC, PC and PR treatment arms, respectively.
21 patients were included in the Partial Responders cohort (PRC: 3, PC: 9, PR: 9). Baseline characteristics were comparable between the PC & PR treatment arm. With only 3 patients in the PRC treatment arm efficacy analysis was not performed.
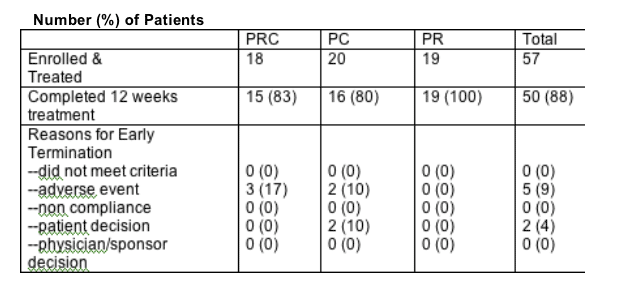
Patient Disposition by Treatment Group
88% of the 57 patients enrolled completed the 12 weeks of study. Adverse events included fatigue, flu-like symptoms, injection site reactions, 1 diarrhea.
SAFETY SUMMARY
All 3 combination treatments generally well tolerated. No serious adverse events reported. No new treatment-emergent toxicities were observed:
--as is common with peginterferon alfa-2b and ribavirin, fatigue and flu-like symptoms were reported equally across all treatment groups.
--diarrhea, flatulence and CK elevations in celgosivir-treated groups were mostly mild to moderate in severity, manageable, reversible and consistent with previous clinical experience.
EFFICACY in NON RESPONDERS Cohort at Week 12
On-Treatment Analysis
The triple therapy group had a mean viral load reduction of -1.62 log (n=12); this did not reach statistical significance but was very close to it. The Peg/RBV group had a -0.92 log reduction. The peginterferon plus Celgosivir group had a -0.6 log reduction in viral load. There was a steep early decline in viral load in the non-responder group, about 1 log by day 2. Patients receiving the triple celgosivir containing regimen who had a mean -1.62 reduction in HCV RNA previously had a mean HCV RNA reduction by prior therapy of -0.6 log.
Efficacy in Non Responder Cohort
Log reduction comparison
Number (%) of Patients with Viral load Reductions at Study Week 12
Close to 45% of patients on triple therapy achieved early viral response or minimum 2 log drop, while in n the other 2 groups EVR was 10%.
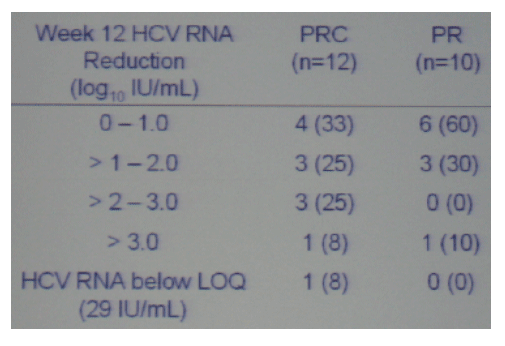
Of note, of the 11 previous null responders all but 1 patient had much better responses with the triple therapy in this study, they had 1 to 4 log better responses than they had to previous therapy. Only 1 patient had no response to the new therapy, they had the same response as to the previous therapy.
EFFICACY SUMMARY
Non Responders
--42% EVR in PRC vs 10% in PR
--Mean viral load drop in PRC of 1.63 log compared to 0.92 in PR
--Rapid onset of treatment effect observed in PRC-treated patients comtinuing through week 12.
Null Responders (subgroup)
--3 PRC-treated patients (43%) achieved EVR in this difficult to treat subgroup.
-- >2 log reduction by week 4 in 1 additional PRC patient (did not complete 12 weeks).
-- 1.86 log mean viral load drop at week 12 in PRC (n=6) vs 0.32 log in PR (n=2).
-- 6/7 patients in PRC group had >1 log reduction in viral load.
DISCUSSION
Celgosivir is a first-in-class oral antiviral compound that targets the a-glucosidase I host enzyme.
Safety: Celgosivir + peginterferon alfa-2b with or without ribavirin is well tolerated with no new combination treatment emergent toxicities.
Rapid Onset: Addition of celgosivir to PR resulted in a rapid onset of treatment effect that continued through the 12 week study treatment period.
Clinical Benefit: Celgosvir added to peginterferon alfa-2b and ribavirin results in improved viral load reductions and clinically significant benefit in genotype-1 non-responders.
|
| |
|
 |
 |
|
|