 |
 |
 |
| |
Short-term monotherapy with UK-453,061, a novel NNRTI, reduces viral load in HIV-infected patients
|
| |
| |
Reported by Jules Levin
4th IAS Conference, 22-25 July, 2007, Sydney, Australia
G Fatkenheuer1; S Staszewski2; A Plettenburg3; F Hackman4; G Layton4; L McFadyen4; J Davis4 and T Jenkins4
1Cologne University, Germany; 2JH Goethe University, Frankfurt, Germany; 3Institute for interdisciplinary Infectiology, Hamburg, Germany & 4Pfizer Global R&D, Sandwich, UK
AUTHOR CONCLUSIONS
UK-453,061 appears generally safe and well tolerated at all doses evaluated in this study in HIV-1 infected men
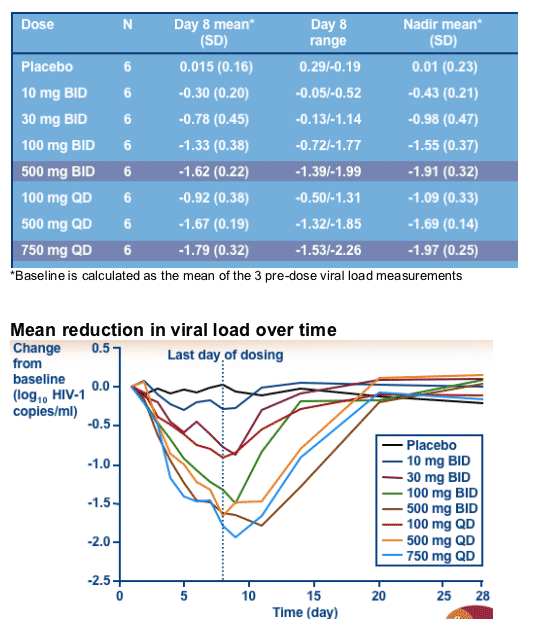
Doses of 500 mg BID & 750 mg QD achieved mean decreases from baseline HIV RNA nadir values of 1.9 & 2.0 log10 respectively
Further evaluation of QD and BID doses of UK-453,061 for the treatment of HIV infection is merited
UK-453,061: preclinical profile
-- WT AV IC90 about12 nM in primary isolates
-- Active against clinically significant NNRTI mutations: e.g. K103N, Y181C/I & G190A/S
-- Resistance required 2-3 mutations - still efavirenz sensitive
-- No CYP3A4 inhibition @ 25 mM
-- Cytotoxicity selectivity index >1000
-- Good CNS penetration predicted
UK-453,061: summary of healthy volunteer data
Safe and well tolerated in single doses (SD) up to 1800 mg and multiple dose (MD) studies up to 1000 mg BID for 12 days
Most frequent adverse events (AEs) after MD: diarrhoea, abnormal bowel sounds, abdominal discomfort, flatulence, dyspepsia and headache
Maximum tolerated dose not identified in SD or MD escalation studies
No significant trends in any liver / renal function, lipid, clinical chemistry or urinalysis parameters
Predictable PK and no clinically-relevant food effect on PK
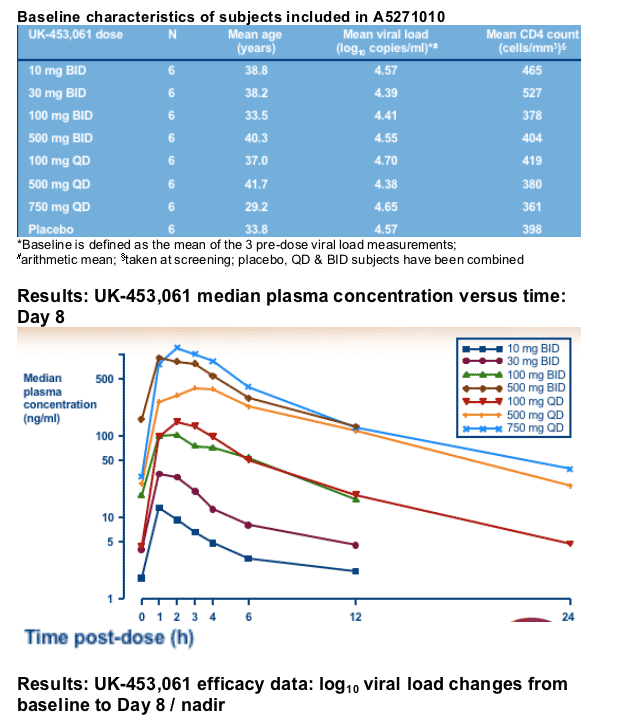
Study A5271010 objectives and patient population
Objectives
--effect of 7-day monotherapy on viral load
--PK / PD relationships
--safety and tolerability of UK-453,061
Patient population
--HIV-infected men with no prior exposure tfo an NNRTI and absence of NNRTI-resistant mutations (determined using VircoGen virtual phenotyping assay)
--CD4 count >250 cells/mm3, viral load >5000 copies/ml
--Antiretroviral (ART) treatment-naive or off treatment for ≥8 weeks at time of screening and diagnosed at least 3 months prior to screening
A5271010 study design
Randomised, double-blind, outpatient study in asymptomatic HIV-infected patients in two stages
Modelling and simulation used to select doses and minimise patient numbers
6 patients / arm and 7 days dosing, Day 8 PK and follow-up to Day 40
Viral load measurements at screening, randomisation,
Day 1 (pre-dose) and Days 2-8, 9, 11, 14, 20, 28 & 40
1st stage (suspension - 5 ml dispensed)
Doses: 10 mg BID, 30 mg BID, 100 mg BID or 500 mg QD
2nd stage (tablets - 50 mg and / or 250 mg dispensed)
Doses: 100 mg QD, 750 mg QD or 500 mg BID
Study A5271010 demography data
Total of 81 HIV-infected patients were screened and 49 randomised (one subject withdrew consent prior to first dose and was replaced) 48 completed the study
Genotyping for NNRTI mutations
Patients were excluded from the study if they had:
-- Previous exposure to NNRTIs (experimental or marketed)
-- Presence of virus containing NNRTI mutations
A5271010 data:
During screening: 7/81 patients excluded: (A98S, K103N, E138G, G190A and K238T)
VircoGen virtual phenotyping assay on Days 1, 8 and 40
-- 1 subject (100 mg QD) = Y181Y/C on Day 8 only
-- 2 subjects (750 mg QD) = Y144F on Day 8 only
No other known NNRTI- or UK-453,061-associated resistance mutations where seen in any dose group
Clonal genotypic assay analysis of pre-dose Day 1, 8 and 40 samples for all subjects is currently ongoing
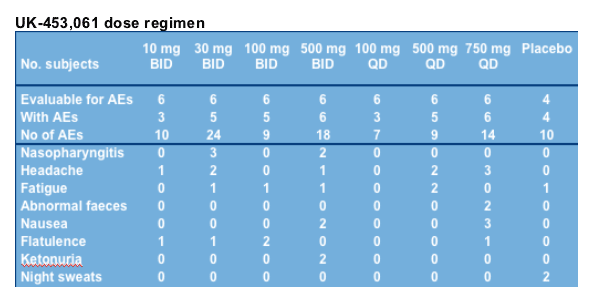
UK-453,061 safety and tolerability data
UK-453,061 appeared safe and well tolerated in this HIV-infected patient population, with 1 (non-treatment related) serious AE and 2 (non-treatment related) severe AEs
No discontinuations due to AEs or lack of toleration
Most common all-causality AEs (3 or more patients; all mild or moderate)
--cold (4)
--flatulence (5)
--headache (9)
--fatigue (6)
--nausea (5)
All causality treatment-emergent AEs occurring in more than one subject in any treatment group
|
| |
|
 |
 |
|
|