 |
 |
 |
| |
Efficacy and Safety of Protease Inhibitor Replacemet for Nevirapine In Patients with High CD4 Lymphocyte Cell Counts: A Systematic Review
|
| |
| |
Reported by Jules Levin
ICAAC Sept 17-20 2007, Chicago
Javier Ena, Angela Leach, Patrick Nguyen
Hospital Marina Baixa. Villajoyosa. Alicante. SPAIN
AUTHOR COMMENTS
Replacement of PIs by nevirapine had similar efficacy to maintain viral suppression as continuing with PI therapy. By intention to treat analysis, 81% of patients in the nevirapine group maintained viral suppression at the end of follow-up compared with 78% in the PI group.
By on treatment analysis, the proportions were 91% vs. 89%, respectively.
Our conclusions were insensitive to a number of potential sources of heterogeneity.
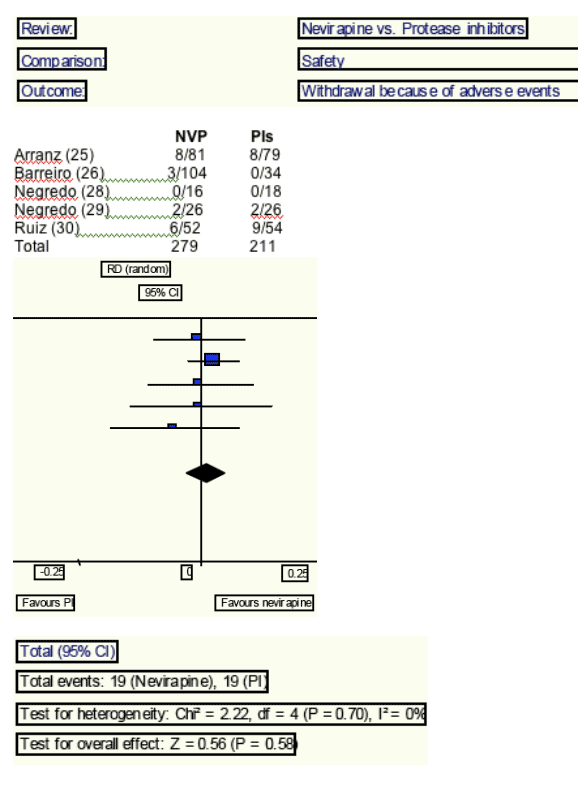
There were no differences in the overall proportion of withdrawals because of side effects in either group (7% vs. 9%%, p=NS).
However, there was greater risk of grade 3-4 liver toxicity in patients treated with nevirapine compared to PI therapy (7% vs. 0%, p=0.002)
ABSTRACT
Background: In order to reverse metabolic side effects and to improve adherence physicians and patients often wish to switch from a complex protease inhibitor therapy either to abacavir, efavirenz or nevirapine. Nevirapine has good long-term tolerability and favourable lipid profile but drug agencies recommended cautious use in patients with hepatic dysfunction and those with high CD4 cell counts due to severe liver toxicity. We carried out a meta-analysis to assess the safety and efficacy of protease inhibitor replacement for nevirapine.
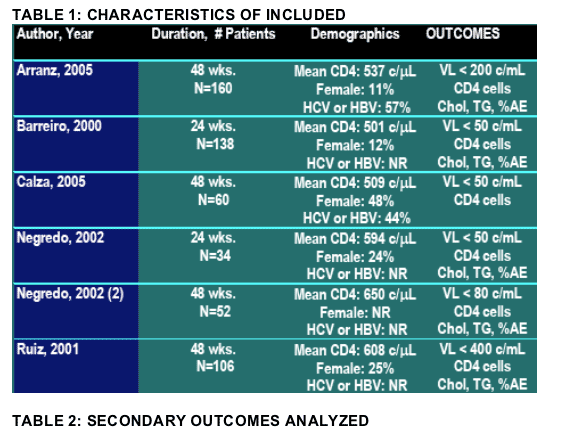
Methods: 6 trials (550 patients) compared replacing protease inhibitors for nevirapine versus continuing with protease inhibitor therapy in adult patients with human immunodeficiency virus in whom virological suppression had been achieved. Demographics at study entry showed patients mean CD4 lymphocytes greater than 500 cells/ _L in all studies, 11-48% of female participants and, 25%-40% of patients infected with hepatitis C virus.
Results:
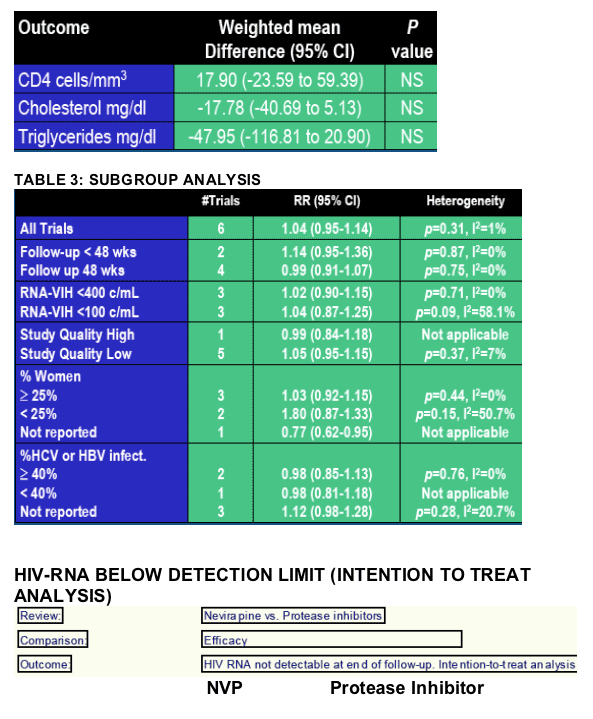
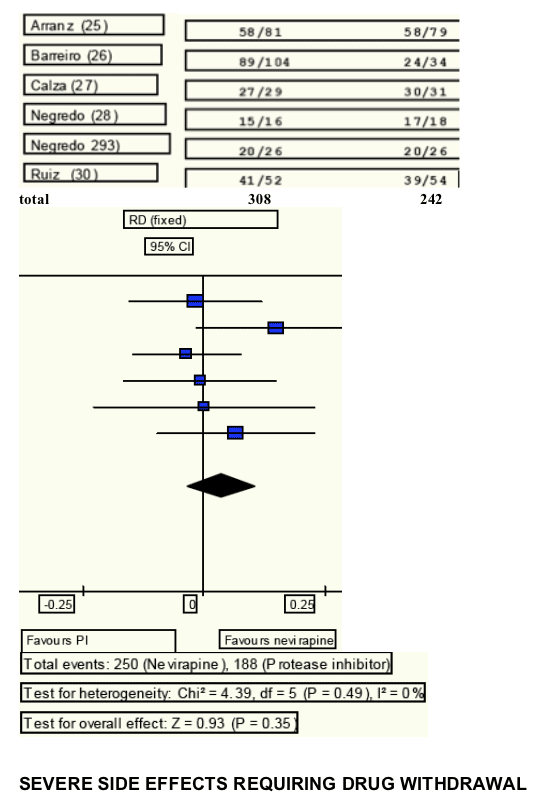
Replacement of protease-inhibitors for nevirapine showed non-inferiority to continuation with protease-inhibitor therapy to maintain virological suppression by ìintention to treat (81% vs. 78%; Risk difference 3%; 95% Confidence Interval [CI]: from -4% to 10%; Heterogeneity I2: 0%) and by ìon treatmentî analyses (91% vs. 89%; Risk difference 3%; CI: from -1% to 11%; Heterogeneity I2: 7.1%).
Discontinuation rate due to adverse events was similar between two groups (6.81% vs. 9.00%, respectively; Risk difference -2%; CI: from -3% to 5%; Heterogeneity I2: 0%).
At end of follow-up there were no differences in CD4 cell counts, cholesterol, triglyceride, and body shape measurements between two groups. Two studies reported greater improvements in quality of life in the nevirapine group compared to those who continued inhibitor protease therapy.
Conclusions: Protease inhibitors replacement with nevirapine is virologically effective and safe in patients with high CD4 lymphocyte cell counts and those with hepatitis C virus infection.
|
| |
|
 |
 |
|
|