 |
 |
 |
| |
Interrupting antiretroviral therapy does not reduce CVD risk and activates the immune system
|
| |
| |
Reported by Jules Levin
ICAAC Sept 17-20, 2007, Chicago
"Antiretroviral Treatment Interruption, Immune Activation and Cardiovascular Risk"
P.TEBAS1, K. HENRY2, R.MATINING3 , D. CHERNG3, J. SCHMITZ4, H. VALDEZ5, N. JAHED6, L. MYERS7,W.G. POWDERLY8, D. KATZENSTEIN9, and the ACTG A5102 Study Team
1University of Pennsylvania, Philadelphia, PA, 2Hennepin County Medical Center- University of MN, 3Harvard School of Public Health, Boston, MA, 4University of North Carolina, Chapel Hill, NC, 5Case Western Reserve University, Cleveland, OH, 6Social & Scientific Systems Inc, Silver Spring, MD, 7FSTRF, 8University College, Dublin, Ireland, 9Stanford University, Stanford, CA
"Our results suggest that treatment interruption would not reduce CVD risk, as any improvements in lipid parameters are modest and overshadowed by the decreased HDL levels and the increased immune cell activation due to systemic inflammatory responses associated with returning HIV viremia. These effects could put the patients at a greater cardiovascular risk than when they were receiving antiretroviral treatment."
AUTHOR CONCLUSIONS
The lack of significant changes in glucose and insulin suggests that the effect of ART on those parameters is limited.
The rapid changes of lipid levels indicates that these effects are due directly to the medications, and most likely are not mediated by changes in body composition which require a lengthier period of time. It is also unlikely that these initial lipoprotein changes are related to HIV replication, as they occurred before the return of HIV viremia.
The decrease in total and LDL cholesterol might be associated with a decreased cardiovascular risk, those benefits may be offset by the marked decrease in HDL cholesterol observed. The net effect of the observed lipid changes would predict little if any improvement in the cardiovascular risk after the discontinuation of ART.
Following treatment interruption there is a prompt and obvious increase in cellular activation, which may negate the modest potential cardiovascular benefits of TI.
The simultaneous combination of these two phenomena (modest decreases in lipids with no net cardiovascular benefit and significant pro-inflammatory changes) could partially explain the increase in cardiovascular events observed in the SMART trial and other studies after the discontinuation of ART.
Abstract
Background: Concern about costs and ART-associated toxicities , led to the consideration of CD4 driven strategies for the management of HIV. That approach was evaluated in the SMART trial that reported an unexpected increase of CVD events after treatment interruption.
Objective: Our goal was to evaluate fasting metabolic changes associated with interruption of ART (TI) and relate them to changes of immune activation markers and cardiovascular risk.
Design: ACTG 5102 enrolled 47 HIV-1-infected subjects on stable ART, with < 200 HIV RNA copies/mL, and CD4 cell count > 500 cells/mm3. Subjects were randomly assigned to continue ART for 18 weeks with or without 3 cycles of IL-2 (cycle= 4.5 million IU sc BID x 5 days every 8 weeks). After 18 weeks ART was discontinued in all subjects until the CD4 cell count dropped below 350. Glucose and lipid parameters were evaluated every 8 weeks initially and at weeks 2, 4, 8 and every 8 weeks after TI. Immune activation was evaluated by flow-cytometry and soluble TNFR2 levels.
Results:
LIPIDS DECREASED: By week 8 of TI, total cholesterol (TC) decreased (median(Q1, Q3) = -28 (-46, -7) mg/dl, p<0.0001) as well as triglycerides (-35 (-74, 6) mg/dl, p=0.005). LDL and HDL cholesterol also decreased (-14(-28,-1), p=0.0007 and -2(-10,1), p=0.0033, respectively).
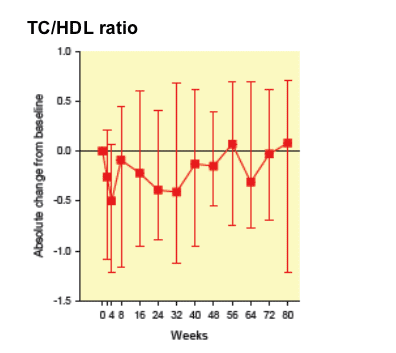
However the TC/HDL ratio remained unchanged (-0.09 (-1.2, 0.5), p=0.20).
Glucose and insulin levels did not change (p=0.6 and 0.8, resp.).
After TI there was marked increase in immune activation (CD8+/HLA-DR+/CD38+ cells, 34% (13, 43), p<0.0001) and soluble TNFR2 (1089 ng/L (-189, 1655), p=0.0008) coinciding with the rebound of HIV viremia.
Conclusions:
Interrupting antiretroviral therapy does not reduce CVD risk, as the improvements in lipid parameters are modest and overshadowed by the decreased HDL levels, the increased immune cell activation and systemic inflammatory responses associated with HIV viremia. These effects could put the patients at a greater cardiovascular risk than when they were receiving antiretroviral treatment.
RESULTS
IL-2 did not have durable effects on lipids or glucose and the two arms are presented combined after treatment discontinuation.
GLUCOSE
After the discontinuation of ART there was no significant changes in either the fasting glucose or insulin levels (Figure 1A).
LIPIDS
After the discontinuation of ART there was a rapid and dramatic decrease of the concentrations of total cholesterol and other lipid fractions. (Figure 1B).
These decreases persisted for the duration of the follow up. All of these changes were evident before the return of detectable HIV plasma RNA, defined as levels > 500 copies/ml.
This suggests that lipid changes were mediated directly by the discontinuation of ART and not by HIV replication itself.
TC/HDL ratio remain unchanged after TI (Figure 2A)
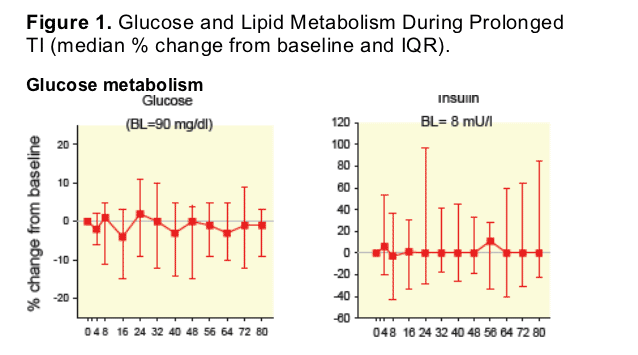
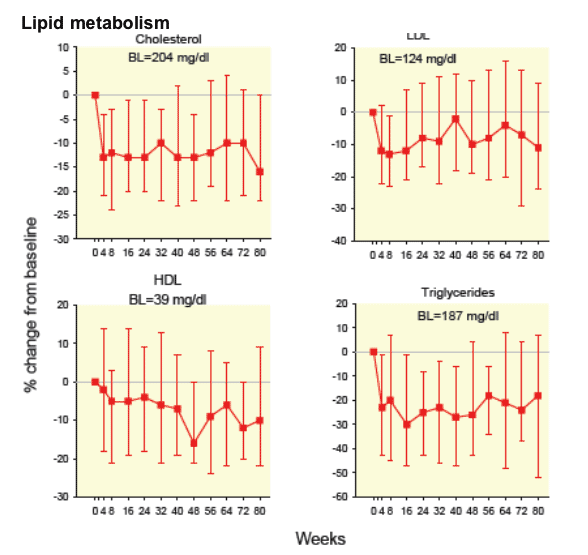
Figure 2. A. Total cholesterol to HDL ratio changes overtime after treatment interruption (median change and IQR).
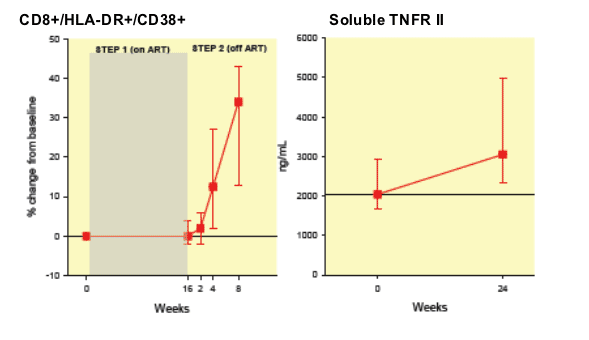
B. Changes in Immune activation.
- Percentage change of CD8+/ HLA-DR+ /CD38+ cells after TI. (median and IQR)
- Absolute change in soluble serum TNFR-II levels (median and IQR). Week 0 (before TI) and 24 w after TI
IMMUNE ACTIVATION
After 8 weeks of TI the percentage of activated CD8 cells in the whole group (HLA-DR+ and CD38+ positive) had increased a median of 34% (IQR 13-43%, Wilcoxon signed rank p<0.0001). The levels of TNF receptor alfa II increased approximately by 30% after 24 weeks after the discontinuation of ART (1089 ng/L (-189, 1655), p=0.0008) (Figure 2B).
There were no significant correlations between the changes in lipid or glucose parameters and the changes in immune activation markers or viral markers during the first weeks of follow up.
Introduction
Treatment associated toxicities together with the costs, both economic and in quality of life terms associated with ART led to the consideration of CD4 driven strategies for the management of HIV infection
The strategy is not without risks, including recrudescent HIV viremia and HIV disease progression.
An unexpected result of the recently discontinued SMART trial, the largest study that evaluated CD4 driven therapy, was an increase in frequency of cardiovascular events in the drug conservation arm
The metabolic effects and overall cardiovascular risk of treatment interruption have not been well defined.
Methods
Subjects
ACTG 5102 enrolled HIV-1-infected adults receiving stable ART, who were virologically fully suppressed (< 200 HIV RNA copies/mL) for at least three months at the time of enrollment and had a CD4 cell count > 500 cells/mm3.
47 subjects were then randomly assigned to continue ART for 18 weeks (Step 1) with (ARM A) or without (ARM B) 3 cycles of IL-2 (cycle= 4.5 million IU sc BID x 5 days every 8 weeks). After 18 weeks ART was discontinued in all subjects (Step 2). Subjects were followed monthly with monitoring of their CD4+ T-cell counts
and plasma HIV-1 RNA levels. ART was restarted when the CD4+ cells reached a confirmed level of < 350 cells/mm3.
Laboratory
CD4+ and CD8+ T-cell counts were measured every 4 weeks during while on ART and at weeks 0,2,4,8, and every 4 weeks thereafter on the treatment interruption phase.
HIV viral load was determined at weeks 0, 8 and 16 of Step 1 and weeks 2, 4, 6, 8, 16 and every 8 weeks thereafter
Fasting metabolic testing (glucose, insulin, total, calculated LDL and HDL cholesterol and triglycerides) were analyzed at each participating site laboratory.
The number and percentage of CD8+ cells expressing activation markers (CD38+/HLA-DR+) were measured at entry and at week 16 of Step 1 while the participant was receiving ART and at weeks 2, 4, and 8 of Step 2 (TI)
Soluble TNF Receptor 2 was measured before TI and 24 weeks after discontinuation of ART.
The metabolic effects of IL-2 had not been described. If there were no changes in the metabolic parameters during the first phase of the study, we planned to combine both arms for the purpose of this analysis.
Statistical analysis
- Summary statistics for continuous variables are presented as median (first quartile, third quartile).
- Comparison between treatment arms was carried out using a Wilcoxon Rank Sum Test for ordinal valued outcomes, and a Fisher's Exact Test or Pearson's Chi-square Test for categorical factors. Within each arm, a Wilcoxon Signed Rank Test was used to assess differences over time.
- Spearman's rank correlation was used to measure the relationship between immunologic and metabolic markers.
- The level of significance used was 0.05. These results are not adjusted for multiple testing and are therefore exploratory.
|
| |
|
 |
 |
|
|