 |
 |
 |
| |
New CCR5 Drug PRO 140 Reduced Viral Load by 2 Logs With Single 10 mg Intravenous Dose; 5/5 patients with single 10 mg dose had >2 log reduction in viral load
|
| |
| |
SEE FULL POSTER BELOW
Progenics Reports Positive Interim Phase 2 Results for Two Dosage Forms 0f Novel HIV Therapy PRO 140
Both intravenous and subcutaneous forms of PRO 140 showed potent and prolonged activity and were generally well tolerated
Washington, DC and Tarrytown, NY - October 26, 2008 - Progenics
Pharmaceuticals, Inc. (Nasdaq: PGNX) today announced positive interim results from two separate phase 2 clinical trials of PRO 140 for the treatment of HIV infection. Both intravenous and subcutaneous forms of PRO 140, a novel viral-entry inhibitor, exhibited potent and prolonged activity as measured by reductions in plasma levels of virus in individuals with HIV infection. Interim results (based on the first 15 of 31 patients treated) from the study of intravenous PRO 140 were presented in Washington, D.C. today as a "late-breaker" poster at the joint meeting of the Interscience Conference on Antimicrobial Agents and Chemotherapy (ICAAC)
and the Infectious Diseases Society of America (IDSA).
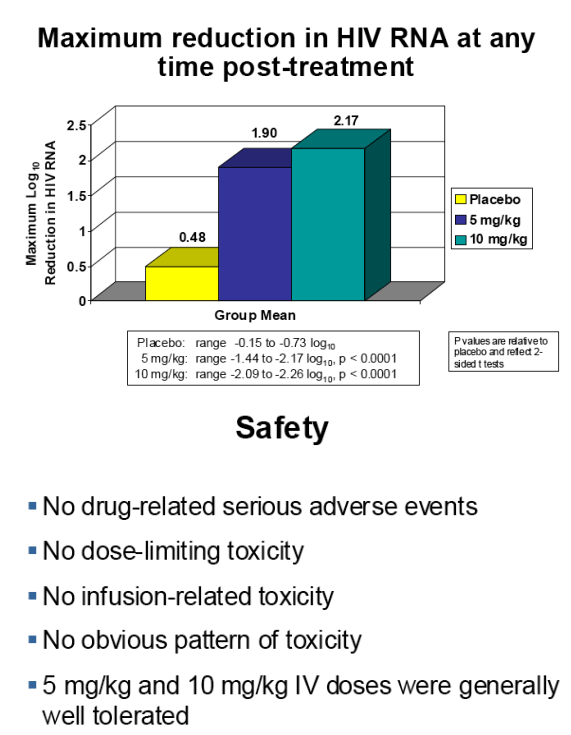
In this study, a 2.17 log10 mean maximum reduction in viral load was observed in patients treated intravenously with a single 10 mg/kg dose, and the antiviral response persisted for three weeks post-treatment.
The Company separately announced positive interim results (based on the first 20 of 44 patients treated) from the first clinical trial of subcutaneous PRO 140. In this study, the mean maximum viral load reduction was 1.77 log10 for patients treated subcutaneously with three weekly doses of 324 mg PRO 140, and the antiviral response persisted for two weeks after the last dose. Both intravenous and subcutaneous forms of PRO 140 were generally well tolerated in these studies.
"The viral load reductions observed for intravenous PRO 140 are similar in magnitude to those observed for our most potent HIV drugs at this stage of
development," said Jeffrey M. Jacobson, M.D., Professor of Medicine, Microbiology and Immunology, Chief, Division of Infectious Diseases and HIV Medicine, Drexel University College of Medicine and lead author of the ICAAC presentation. "The findings underscore the efficiency with which PRO 140 blocks HIV's access to CCR5 and healthy cells in vivo.
In addition, the antiviral and safety data for subcutaneous PRO 140 are highly encouraging. Many patients are excited by the potential for a potent and well-tolerated therapy that could be self-administered and yet not provide a daily reminder of their disease."
PRO 140 is a humanized monoclonal antibody that blocks viral infection of healthy cells by binding to CCR5, a co-receptor that is the principal molecular portal used by HIV to enter and infect immune system cells. Some strains of HIV use the CXCR4 co-receptor as a portal of entry either exclusively or in addition to
CCR5. CCR5 viral-entry inhibitors, such as PRO 140, are active in blocking infection in the majority of HIV patients whose virus uses the CCR5 portal, but do not block the entry of virus that uses the CXCR4 portal.
"The interim analyses were conducted to provide timely information on the potential feasibility of two attractive treatment options for PRO 140: monthly intravenous and weekly subcutaneous dosing," said Paul J. Maddon, M.D., Ph.D., Progenics Pharmaceuticals' Founder, Chief Executive Officer and Chief
Science Officer. "These interim data support the potential feasibility of both options.
Importantly, in our first clinical study of the subcutaneous form of PRO 140,
we observed significant and prolonged reductions in viral load. Based on our discussions with the HIV community, we believe that weekly subcutaneous dosing of a drug with a favorable tolerability profile would be a desirable new treatment paradigm. Our intravenous form of PRO 140 also exhibited substantial and sustained reductions in viral loads with a favorable tolerability profile.
We expect to obtain results from all patients enrolled in both of these studies by early 2009, and these data will be used to select a dosage form (intravenous or subcutaneous) for further clinical development."
Intravenous PRO 140: Summary of interim phase 2 results
In the intravenous phase 2 trial, single infusions of PRO 140 were evaluated in HIV-infected individuals with early-stage disease, who had received no antiretroviral therapy for at least three months and who had HIV-1 RNA viral loads of greater than 5,000 copies/mL. Prior to treatment, all patients were screened for the presence of virus that uses CCR5, and the absence of virus that uses CXCR4, as the entry co-receptor. Subjects were monitored for 58 days following treatment for safety and antiviral effects. A total of 31 individuals were enrolled and approximately evenly randomized to receive a single dose of placebo,
PRO 140 5 mg/kg or PRO 140 10 mg/kg on day one. An interim analysis was performed on safety and efficacy data from the first 15 subjects treated. There were five subjects in each of the three dose groups.
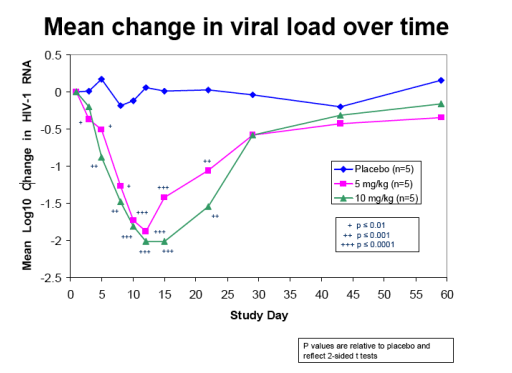
Interim results from this study reconfirmed the substantial antiviral activity observed at the 5 mg/kg dose level in the earlier phase 1b trial, and also showed that doubling the dose to 10 mg/kg increased the duration and magnitude of antiviral response. Each of the five subjects treated with the 10 mg/kg dose experienced a greater than 2.0 log10 (100-fold) reduction in viral load.
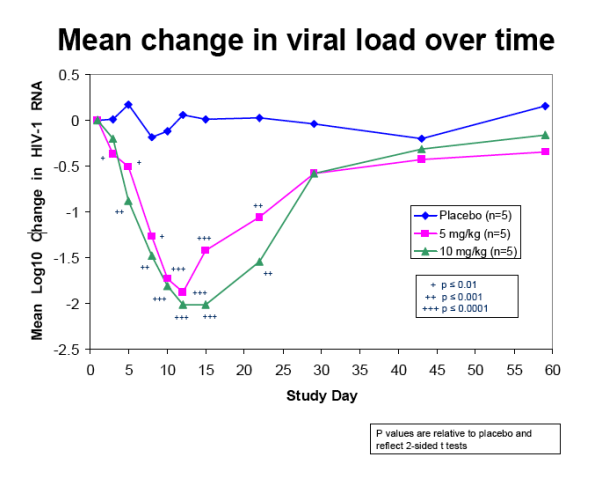
Figure 1 depicts the mean viral load reductions over time for the three dose groups. A greater than 1.0 log 10 (10-fold) mean reduction in viral load was sustained
until three weeks post-treatment at the 10 mg/kg dose level, compared with two to three weeks at the 5 mg/kg dose level in the present study and in the previous phase 1b trial.
Poster
Antiviral Activity and Tolerability of 5 mg/kg and 10 mg/kg Doses of PRO 140, a Humanized Monoclonal Antibody to CCR5
Reported by Jules Levin
Chemotherapy (ICAAC) and the Infectious Diseases Society of America (IDSA) - October 25-28, 2008 - Washington, D.C Sunday, October 26, 2008
Jeffrey M. Jacobson, M.D.
Drexel University College of Medicine
245 N. 15th Street, MS 461
Philadelphia, PA 19102
Tel: 215-762-6555
Fax: 215-762-3031
jeffrey.jacobson@drexelmed.edu
Jeffrey M. Jacobson1, Melanie A. Thompson2, Margaret A. Fischl3, Michael S. Saag4, Barry S. Zingman5, Ralph Liporace6, Jacob P. Lalezari7, Carl J. Fichtenbaum8,
Daniel S. Berger9, Nancy Stambler10, Yakov Rotshteyn10, Paul D'Ambrosio10, Marti Michael10, Stephen A. Morris10, Paul J. Maddon10 and William C. Olson10
1Drexel U. Coll. of Med., Philadelphia, PA, 2ARCA, Atlanta, GA, 3U. Miami, Miami, FL, 4U. Alabama, Birmingham, AL, 5Montefiore Med. Ctr., Bronx, NY,
6Albany Med. Ctr., Albany, NY, 7Quest Clinical Res., San Francisco, CA, 8U. Cincinnati, Cincinnati, OH, 9NorthStar Med. Ctr., Chicago, IL, 10Progenics Pharmaceuticals, Tarrytown, NY.
AUTHOR CONCLUSIONS
High-level activity in subjects with R5 virus is consistent with previous findings
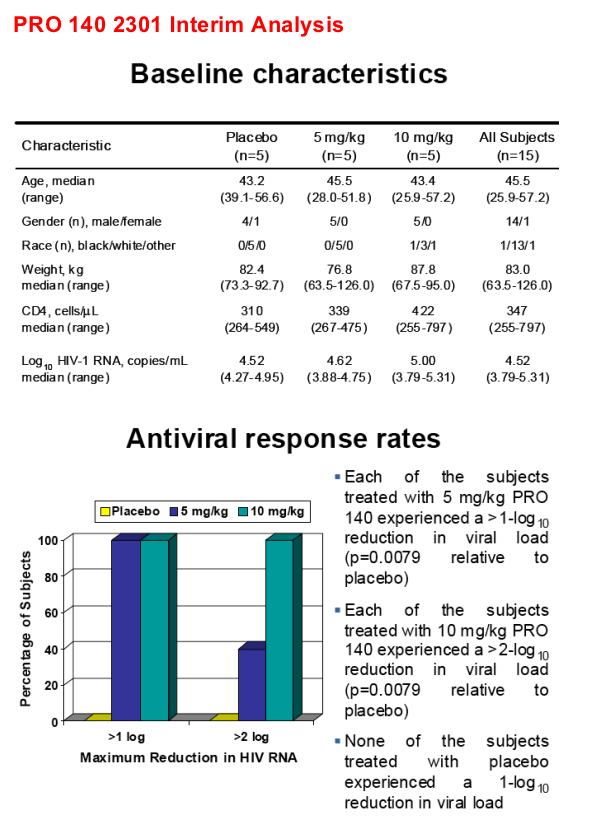
100% antiviral response rate in PRO 140-treated subjects
- >1 log10 reduction in HIV RNA in 5 of 5 subjects treated with 5 mg/kg
- >2 log10 reduction in HIV RNA in 5 of 5 subjects treated with10 mg/kg
Duration of antiviral effects increased with increasing dose
- >1 log10 mean reduction in viral load maintained until nearly 4 weeks at 10 mg/kg v. 3 weeks at 5 mg/kg
- Antiviral effects observed at 10 mg/kg support evaluation of repeat dosing at 3-week intervals
No dose-limiting toxicity or apparent safety signal
Modeling suggests that a 20 mg/kg dose may maintain a >1 log10 reduction in viral load beyond one month
PRO 140 Next Steps
Evaluate the safety, PK, antiviral effects and duration of antiviral effects of up to 20 mg/kg PRO 140 administered intravenously
Complete phase 2a evaluation of PRO 140 administered subcutaneously
Select route of administration and dose levels
for phase 2b/3 testing
ABSTRACT
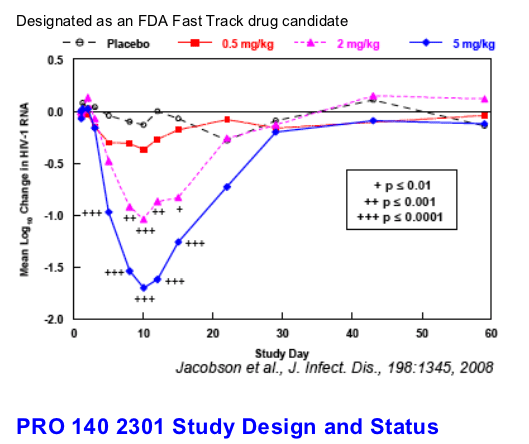
Background: PRO 140 is a humanized anti-CCR5 monoclonal antibody that broadly and potently inhibits CCR5-tropic (R5) HIV-1 in vitro. A previous phase 1b study examined single ascending intravenous doses of PRO 140 ranging to 5
mg/kg in subjects with early-stage infection and only R5 HIV-1 detectable. In the prior study, the mean maximum reduction in HIV-1 RNA was 1.83 log10 at 5 mg/kg, and all subjects treated at this dose experienced a >1 log10 reduction
in HIV-1 RNA. The present phase 2a study compared 5 mg and 10 mg IV doses of PRO 140 for antiviral activity and tolerability.
Methods: Entry criteria included HIV-1 RNA > 5,000 copies/mL, only R5 virus detectable, CD4 > 300/μL, and no antiretroviral therapy within 12 weeks. Subjects were randomized to receive placebo, 5 mg/kg PRO 140 or 10 mg/kg PRO 140. They were followed for 58 days for safety, antiviral effects and pharmacokinetics. An interim analysis for safety and antiviral activity was performed on data from the first 15 treated subjects.
Results: At the interim analysis, enrollment was equally distributed across the three treatment groups. Baseline HIV-1 RNA values and CD4 counts averaged 35,480 copies/mL and 403/μL, respectively, and were similar for the different
treatment groups.
The mean maximum log10 reductions in HIV-1 RNA were 0.48 (median 0.59, range 0.15-0.73) for the placebo group, 1.90 (median 1.98, range 1.44-2.17, p<0.0001) for 5 mg/kg PRO 140 and 2.17 (median 2.16, range 2.09 to 2.26, p<0.0001) for 10 mg/kg PRO 140. At Day 12, the mean log10 changes in HIV-1 RNA were +0.06, -1.88 (p<0.0001), and -2.01 (p<0.0001) for the placebo, 5 mg/kg and 10 mg/kg groups, respectively.
The mean viral load reduction was >1.5 log10 through Day 22 at 10 mg/kg. Both doses were generally well tolerated. Trial enrollment has since completed, and updated data for all 31 treated subjects will be presented.
Conclusions: The antiviral effects observed to date in this study compare favorably with those reported previously for PRO 140 5 mg/kg. A 10 mg/kg dose of PRO 140 increased the duration of antiviral effect. The findings indicate the
potential for infrequent intravenous dosing with PRO 140. A study to test weekly and biweekly subcutaneous dosing regimens also is ongoing.
Humanized monoclonal antibody targeting CCR5, which serves as a co-receptor for HIV-1 entry
Broadly and potently inhibits R5 HIV-1 replication
Distinct class of CCR5 inhibitor
--Binds extracellular domains vs. transmembrane pocket
--Inhibits HIV via competitive vs. allosteric mechanism
--Inhibits HIV without blocking the natural activity of CCR5 in vitro
--Synergistic with small-molecule CCR5 antagonists
--Limited cross-resistance with small-molecule CCR5 antagonists in vitro
Additional potential advantages over existing therapies
--Improved tolerability without drug-drug or drug-food interactions
--Infrequent dosing
Phase 1b study in HIV-infected individuals (PRO 140 1302)
- 1.83 log10 mean reduction in HIV RNA following single 5 mg/kg IV dose
- >/=1.0 log10 mean reduction in HIV RNA for 2-3 weeks post-treatment
- Individual reductions up to 2.5 log10
- Generally well tolerated at all doses tested


|
| |
|
 |
 |
|
|