 |
 |
 |
| |
A Pilot Study to Determine the Effect on Dyslipidemia of the Addition of Tenofovir to Stable ART in HIV-infected Subjects: Results from A5206 Study Team
|
| |
| |
Reported by Jules Levin
CROI 2009 Feb 8-12 Montreal
Marisa Tungsiripat1*, Douglas Kitch2, Marshall Glesby3, Samir Gupta4, John Mellors5, Laura Moran6, Lynne Jones7, Beverly Alston-Smith8, James F. Rooney9, and Judith A. Aberg10
1Cleveland Clinic and Case Western Reserve University, 2Harvard School of Public Health, 3Cornell University, 4Indiana University School of Medicine,
5University of Pittsburg Medical Center, 6ACTG Operations Center, 7Frontier Science and Technology Center, 8National Institute of Allergy and Infectious Diseases, 9Gilead Sciences, and 10New York University
AUTHOR SUMMARY & CONCLUSIONS
The addition of TDF to existing virologically-suppressed ART regimens improved lipid parameters, supporting an independent lipid-lowering effect of TDF.
The mechanism of the lipid-lowering effect and potential rebound effect of increased TG with discontinuation of TDF warrants further study.
ABSTRACT
Background: Several studies have demonstrated improvements in lipids after antiretroviral therapy (ART) switches to tenofovir (TDF). We aimed to isolate the effect of TDF (in regards to lipid-lowering) by adding it to a stable ART regimen.
Methods: In a double-blond, placebo-controlled, randomized crossover design, two 12-week treatment periods randomized to order of treatment (TDF and placebo, respectively) were separated by a 4-week washout period. Virologically-suppressed HIV-infected individuals with dyslipidemia were evaluated with fasting lipids [total cholesterol, HDL cholesterol (HDL-C), LDL cholesterol (LDL-C) and triglycerides (TG)] prior to the start of each treatment period and every 4 weeks throughout each treatment period.
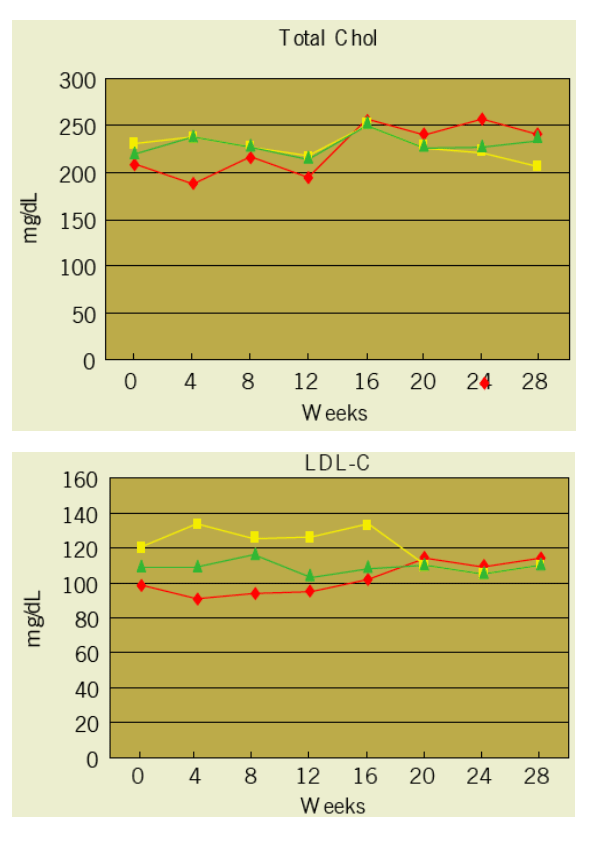
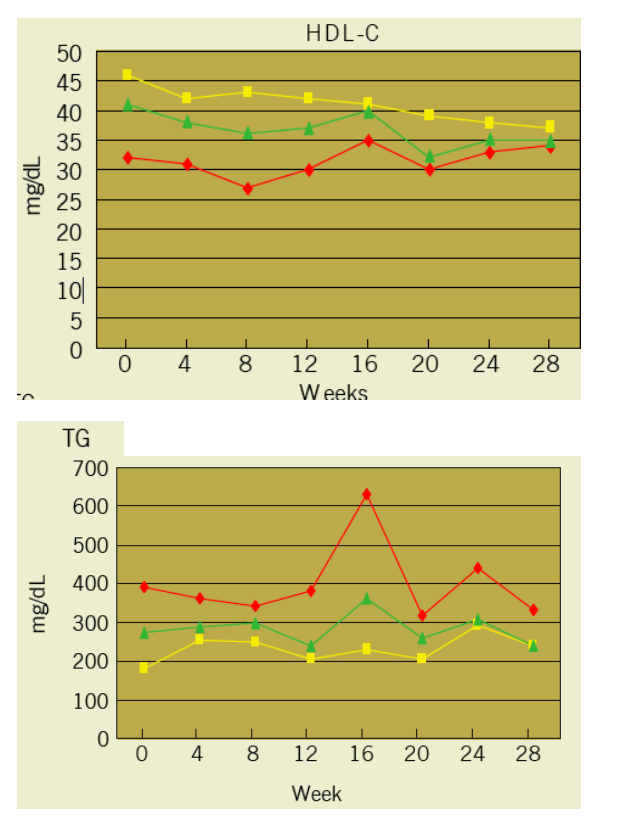
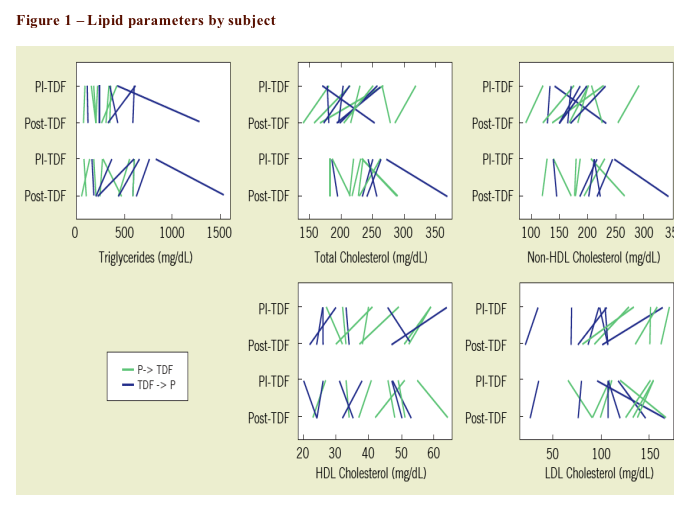
Results: 17 subjects enrolled. 4 (24%)were female; 6 (35%) were black; and 3 (18%) were Hispanic. Lipid parameters (non-HDL-C, total cholesterol, and LDL-C) with the exception of HDL-C and TG improved significantly over the TDF treatment period in comparison to the placebo treatment period.
There was no significant difference in TG changes between the two treatment periods. In the six subjects randomized to receive TDF first, the TG levels decreased from a median of 390 (237, 604) mg/dL at baseline to 381 (241, 586) mg/dL at week 12 when TDF was discontinued. Over the 4-week washout period, the TG levels increased by a median of 145.5 (35, 176) mg/dL and 34% (28%, 51%) to a median value of 630 (365, 762) mg/dL.
At the end of the second phase the median TG level was 331 (208, 621) mg/dL, similar to baseline levels.
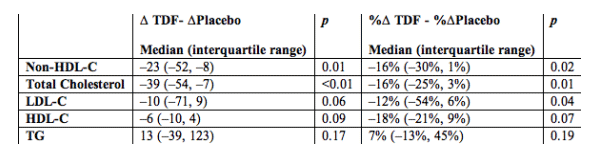
Conclusion: The addition of TDF to existing virologically-suppressed ART regimens improved lipid parameters, supporting an independent lipid-lowering effect of TDF. The mechanism of the lipid-lowering effect and potential rebound effect of increased TG with the discontinuation of TDF warrants further study.
INTRODUCTION
Several recent studies have highlighted the importance of cardiovascular issues in HIV-infected individuals. Traditional interventions often do not fully achieve lipid-lowering goals in HIV-infected population. Additional lipid-lowering options are needed in this population.
TDF may provide a needed addition to the available lipid-lowering options in this population.
Prior studies suggest that TDF may by associated with lipid-lowering which has prompted many clinicians to use TDF as a component of 'lipid-friendly' regimens.
Several studies have demonstrated that switching from other antiretroviral agents (mainly d4t) to TDF results in improvements in lipids except HDL-C.
Our study differs from prior studies in that we have aimed to isolate the effect of TDF (in regards to lipid-lowering) by adding it to a stable, virologically suppressive regimen
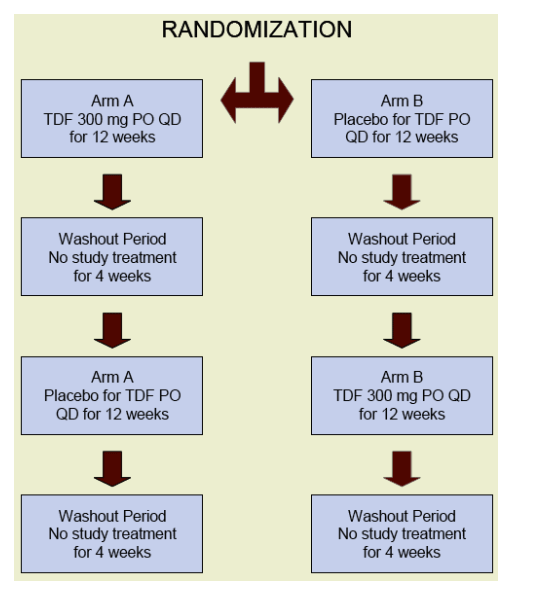
ACTG A5206 was a double-blind, randomized placebo-controlled, crossover, pilot study of HIV-infected individuals. This study added TDF to a stable antiretroviral regimen to evaluate the effect on lipids.
Inclusion/ exclusion
- HIV-infected individuals at least 18 years
- Dyslipidemia (defined as fasting TG >/= 150 and < 1000 mg/dL or fasting non-HDL cholesterol levels >/= 100 and < 250 mg/dL)
- Virologically suppressed with HIV-1 RNA <400 copies/mL
- On stable ART for at least 90 days prior to study entry. (DDI and 'unboosted' ATV not allowed due to concerns of interaction with TDF)
- No hx of active hepatitis B infection
- No hx of cardiovascular disease, uncontrolled DM, untreated hypothyroidism, or use of hormonal anabolic therapy
- Lipid-lowering agents could not be changed or added
Study treatment
12-week treatment periods randomized to order of treatment (TDF and placebo, respectively) were separated by a 4-week washout period
Subjects continued all antiretroviral medications in their regimens
Evaluations- at Start of each Treatment Period and Every 4 weeks thoughout each Treatment Period (Weeks 0, 4, 8, 12, 16, 20, 24, 28):
Fasting (> 8 h) triglycerides, total cholesterol, HDL and LDL cholesterol
Statistical Methods:
The primary endpoint was the change in non-HDL-C over 12 weeks of active TDF minus the change over 12 weeks of placebo for TDF.
The primary analysis used Wilcoxon signed rank tests [change (Δ) or %Δ over 12 weeks of placebo] and was a per protocol analysis limited to subjects with lipid-level data for baseline and week 12 of both treatment periods. As this was a pilot study, the type I error rate was relaxed to 0.10.
In order to confirm our results, 2 sample t-tests were used for 1) treatment effect adjusted for period effect, 2) period effect adjusted for treatment effect, and 3) interaction of treatment and period effects
RESULTS
17 Subject enrolled. 4 (24%) were female; 6 (35%) were black; and 3(18%) were Hispanic.
8 subjects were randomized to TDF->Placebo; 9 received the opposite sequence
One subject from each arm was lost to follow-up (1 started lipid-lowering agent; 1 wished to DC medication
Did not have week 28 lipid-level data from 1 subject in each arm (1 did not have week 28 fasting lipids; 1 started lipid-lowering agent in violation of protocol)
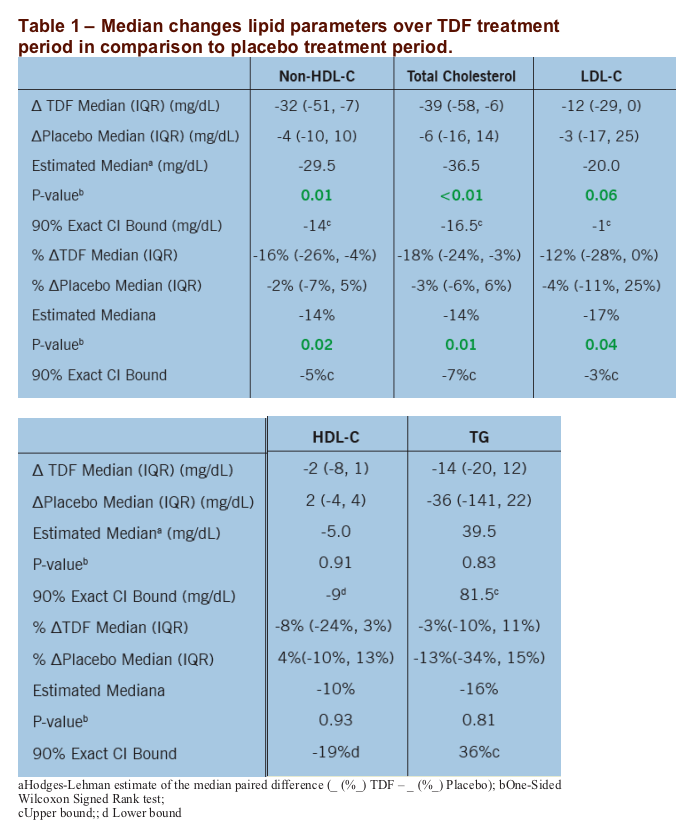
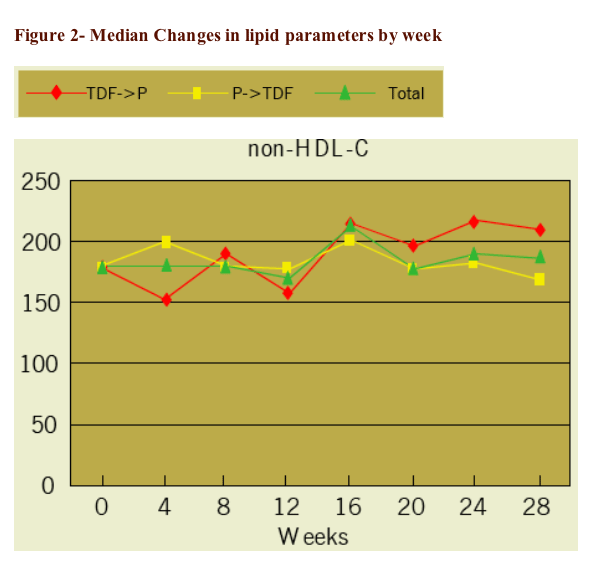
|
| |
|
 |
 |
|
|