 |
 |
 |
| |
New HCV Drugs II: Schering's protease boceprevir phase 2; Abbott/Enanta HCV Protease Inhibitors; Progenics PRO-206 HCV Entry Inhibitor
|
| |
| |
EASL
Reported by Jules Levin
In the early afternoon presentation Schering Plough reported phase II results from the SPRINT Study of their protease inhibitor boceprevir currently in phase III. The data is very interesting. You can see in the table below overall SVR was 55% with 28 weeks of boceprevir+peg/rbv with a 4-week peg/rbv lead-in before starting boceprevir SVr was 56%. Patients with 48 weeks and no lead-in had 67% SVR and patients with 4 week lead-in and 48 weeks therapy had 75% SVR. Of interest they presented patients response who had little response to peg-RBV lead-in. So patients with < .5 log reduction had 44% SVR and patients with .5 to 1 log reduction had 62% SVR. These rates are surprisingly good but of course these patients are susceptible to greater risk for PI resistance. Of note, patients with RVR and lead-in had 82% SVR after 28 weeks and 94% SVR with 48 weeks therapy and lead-in. Without lead-in therapy resulted in 74% and 86% in 28 & 48 weeks, respectively. Still, the most interesting study at this conference will be the INFORM Stud presented saturday which will report the results from combination of 2 orals R7128 the polymerase and ITMN191 the protease being developed by Roche. They are looking at quad therapy combining peg/RBV with these 2 orals. This is the first data of 2 orals in patients. Stay tuned.
HCV SPRINT-1 FINAL RESULTS : SVR 24 FROM A PHASE 2 STUDY OF BOCEPREVIR PLUS PEGINTRON (PEGINTERFERON ALFA-2B)/RIBAVIRIN IN TREATMENT-NAµVE SUBJECTS WITH GENOTYPE-1 CHRONIC HEPATITIS C
P. Kwo1, E. Lawitz2, J. McCone3, E. Schiff4, J. Vierling5, D. Pound6, M. Davis7, J. Galati8, S. Gordon9, N. Ravendhran10, L. Rossaro11, F. Anderson12, I. Jacobson13, R. Rubin14, K. Koury15, C. Brass15, E. Chaudhri15, J. Albrecht15
1Department of Medicine, Indiana University School of Medicine, Indianapolis, IN, 2Alamo Medical Research, San Antonio, TX, 3Mount Vernon Endoscopy Center, Alexandria, VA, 4Center for Liver Diseases, University of Miami Miller School of Medicine, Miami, FL, 5Professor of Medicine and Surgery, Baylor College of Medicine, Houston, TX,6Indianapolis Gastroenterology Research Foundation, Indianapolis, IN, 7Digestive Care/South Florida Center of Gastroenterology, Wellington, FL, 8Liver Specialists of Texas, Houston, TX, 9Department of Hepatology, Henry Ford Health Systems, Detroit, MI, 10Digestive Disease Associates, Baltimore, MD, 11Internal Medicine, University of California-Davis Medical Center, Sacramento, CA, USA, 12The Liver & Intestinal Research Center, Vancouver, BC, Canada, 13Weill Cornell Medical College, New York, NY, 14Digestive Healthcare of Georgia, Atlanta, GA, 15Schering-Plough Research Institute, Kenilworth, NJ, USA
Background: HCV SPRINT-1 assessed the safety and efficacy of boceprevir, an oral inhibitor of HCV-NS3 protease, plus PegIntron (P) (1.5 µg/kg/QW) and ribavirin (R) (400-1400 mg/day).
Methods: P/R(800-1400 mg/day) for 48 weeks compared to five boceprevir(800 mg TID) regimens: 4 weeks of P/R lead-in followed by P/R(800-1400 mg/day)/boceprevir for 24 or 44 weeks; P/R(800-1400 mg/day)/boceprevir for 28 or 48 weeks; and P/low-dose R(400-1000 mg/day)/boceprevir for 48 weeks. The primary endpoint was sustained virologic response (SVR) at 24 weeks post-treatment (Roche TaqMan LLD=15 IU/mL).
Results: Of 595 patients treated in US (77%), Canada and Europe, 60% were male, 16% Black, 7% cirrhotic, 56% genotype 1a and 89% had high viral load (>600,000 IU/mL). SVR was significantly increased in the 28 and 48 week boceprevir arms compared to P/R Control. RVR and EVR were highly predictive of SVR with boceprevir combinations. Rash-related AEs were similar for boceprevir regimens and P/R Control.
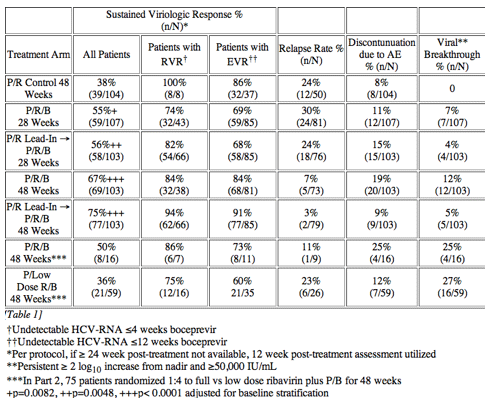
Conclusions: Both 28 and 48 week boceprevir regimens significantly increased SVR with very low relapse rates in 48 week regimens. However, low dose ribavirin with PegIntron and boceprevir was associated with increased viral breakthrough, relapse and lower efficacy. In contrast, P/R lead-in prior to boceprevir substantially increased SVR and reduced viral breakthrough.
In the late afternoon oral session Progenics reported preclinical data for their HCV entry inhibitor which looks promising. But Abbott is presenting their preclinical data for 2 HCV protease inhibitors. Both PIs are potent against all genotypes and in vitro are more potent than some other HCV PIs. Abbott PIs are active against resistant mutants with potential for once daily dosing.
Session Title: Parallel Session 1: HEPATITIS C VIRUS DRUG DEVELOPMENT I
Presentation Date: Apr 23, 2009
POTENT HCV PROTEASE INHIBITORS WITH THE POTENTIAL FOR ONCE-DAILY DOSING AND BROAD GENOTYPE COVERAGE
L.J. Jiang1, Y. Gai1, T. Middleton2, K. Kurtz2, L. Lu2, S. Liu1, T. Phan1, X. Luo1, T. Poon1, B. Brasher1, K. McDaniel2, D. Kempf2, Y.S. Or1
1Enanta Pharmaceuticals, Inc., Watertown, MA, 2Abbott Laboratories, Chicago, IL, USA
Background: Chronic hepatitis C (HCV) is becoming a major global health burden; current therapies have sub-optimal response rates and are associated with significant side effects. The design of HCV protease inhibitors (PI) EA-058 and EA-063 was undertaken in response to this unmet medical need.
Methods: IC50 and EC50 values were determined in a biochemical assay using recombinant HCV protease and in a replicon system using a luciferase readout, respectively. Rats were dosed orally at 20 mg/kg of EA-058, and plasma and tissue samples were analyzed by LC/MS/MS. Intrinsic clearance in human liver microsomes was calculated by disappearance of parent compound.
Results: IC50 values for EA-058 against genotype 1a, 1b, 2a, 2b, 3a and 4a protease were 0.04, 0.1, 0.3, 1.0, 16 and 0.1 nM, and for EA-063 were 0.03, 0.1, 0.09, 0.4, 0.4, 0.7 and 0.08 nM, respectively. EC50 values for EA-058 against genotype 1a and 1b replicons were 0.8 and 1.5 nM, and for EA-063 were 0.1 and 0.2 nM, respectively, providing selectivity indices of >280,000. The EC50's of EA-063 against genotype 1a R155K, D168E, D168K resistant mutants were 0.7, 0.5, and 27 nM, and against genotype 1b R155K, A156T and D168V resistant mutants were 1.2, 15.1, and 4.3 nM, respectively. Following a single PO dose of 20 mg/kg EA-058 in rats, the ratio of liver exposure over plasma exposure was 440. In contrast, EA-058 levels in the heart were below detection limit of 10 ng/mL across 48 hours. The intrinsic clearance values for EA-058, EA-063, TMC-435350, MK-7009 and ITMN-191 in human liver microsomes were 6.2, 3.6, 4.0, 45.9 and 144.3 µL/min/kg, respectively.
Conclusions: EA-058 and EA-063 are highly potent PIs with broad genotype coverage and excellent in vitro safety windows; and their virological profiles are superior to ITMN-191, MK-7009 and TMC-435340 which were tested side by side. EA-058 and EA-063 have equal metabolic stability to TMC-435350, suggesting the potential for once daily dosing. They concentrate in the liver with minimal plasma and heart exposure in rats, providing the potential to minimize extra-hepatic adverse effects in humans.
PRECLINICAL CHARACTERIZATION OF PRO 206, AN ORALLY ACTIVE SMALL-MOLECULE HEPATITIS C (HCV) ENTRY INHIBITOR
D. Qian1, G. Coburn1, A.Q. Han1, J.-M. de Muys1, C. Gauss1, K. Provoncha1, M. Canfield1, D. Paul1, S. Mohamed1, S. Moorji1, D. Fisch1, J. Murga1, Y. Rotshteyn2, P.J. Maddon3, W.C. Olson1
1Research and Development, 2Preclinical Development, 3CEO, Progenics Pharmaceuticals Inc., Tarrytown, NY, USA
Background: Combinations of specific anti-viral drugs, with complementary mechanisms of action and determinants of resistance, offer the potential to improve sustained virologic response rates and to improve tolerability for HCV infected patients. To date, most drug discovery and development efforts have targeted two HCV enzymes: the NS3/4A serine protease and the NS5B RNA-dependent RNA polymerase. Targeting HCV entry represents a novel therapeutic approach that has been validated clinically for other pathogenic viruses. We have discovered a small-molecule inhibitor of HCV entry, PRO 206, and describe herein its preclinical characteristics.
Methods: The activity of PRO 206 was evaluated in an HCV pseudo-particle (HCVpp) assay using fusogenic envelopes isolated from multiple HCV+ patients. Antiviral activity was also measured against authentic HCV in cell culture (HCVcc). Cytotoxicity was assessed in a standard CellTiter Glo assay. Selectivity was determined against a panel of pseudoviral particles that were typed with the envelope glycoproteins from unrelated viruses. PRO 206 was also profiled against a broad panel of human receptor and enzyme targets of pharmacologic interest. Pharmacokinetic properties were characterized in rats after intravenous or oral administration. ADME (absorption/distribution/ metabolism/excretion) properties were assessed in a panel of in vitro assays including Caco2 permeability, human microsomal stability, and inhibition of cytochrome P450s.
Results: PRO 206 demonstrated potent and selective inhibitory activity against HCVpp and HCVcc at concentrations that had no measurable effect on cell viability, entry of unrelated viruses, or a broad panel of human receptors and enzymes. PRO 206 also demonstrated favorable ADME properties, as well as high oral bioavailability and a prolonged pharmacokinetic half-life in animals.
Conclusions: Preclinical data indicate that PRO 206 was a potent, selective, non-cytotoxic and orally bioavailable inhibitor of HCV entry. The antiviral and pharmacokinetic properties of PRO 206 suggest the potential for once-daily dosing in humans. Based on these data, PRO 206 has been selected for preclinical development and is currently undergoing IND-enabling studies to support the initiation of testing in humans for the treatment of HCV infection.
|
| |
|
 |
 |
|
|