 |
 |
 |
| |
Pharmacokinetics, Safety and Tolerability of the HCV Polymerase Inhibitor ABT-333 Following Multiple Ascending Doses and Effect of Co-Administration of Ketoconazole in Healthy Subjects
|
| |
| |
44th Annual Meeting of the European Association for the Study of the Liver ( EASL), Copenhagen, Denmark, 22-26 April 2009
Reported by Jules Levin
Rajeev Menon*, Daniel Cohen, Adel Nada, Yi-Lin Chiu, Thomas Podsadecki, Walid Awni, Barry Bernstein, Cheri Klein
Abbott Laboratories, Abbott Park, IL
AUTHOR CONCLUSIONS
ABT-333 was safe and well tolerated following multiple dosing with 200, 400, 600, and 1000 mg BID for 10 days.
The Pharmacokinetic parameters of ABT-333 increased proportionally with increase in dose after multiple dosing of ABT-333 200, 400, 600, and 1000 mg BID for 10 days.
The mean Cmax and AUC12 values on Days 1 and 10 were similar, indicating that minimal accumulation occurs following multiple dosing of ABT-333 200 to 1000 mg BID. Consistent with no accumulation, steady state levels of ABT-333 are reached by Day 2 following dosing of ABT-333 200 to 1000 mg BID.
The dose normalized Cmax and AUC values following multiple dosing are consistent with those from the single ascending dose study.
The pharmacokinetic parameters of ABT-333 following morning and evening doses on Day 10 are similar, indicating that there is minimal diurnal variation in ABT-333 pharmacokinetics.
When ABT-333 was co-administered with ketoconazole, the mean ABT-333 Cmax and AUC12 values were only 53 and 64% higher as compared to ABT-333 administration without ketoconazole. As the fold increase in AUC was between 1.25 and 2, based on FDA guidance, ABT-333 is not a sensitive substrate of CYP3A.
ABSTRACT
Background: ABT-333 is a novel non-nucleoside NS5B polymerase inhibitor being developed for the treatment of HCV genotype 1 infection. This study evaluated the safety, tolerability, and pharmacokinetics of multiple oral doses of ABT-333 and assessed the effect of a potent CYP3A inhibitor on the pharmacokinetics of ABT-333 in healthy subjects.
Materials and Methods: Healthy adult subjects received ABT-333 from 200 to 1000 mg BID for 10 days in a double-blind, placebo-controlled fashion in sequential groups. In each dose group, subjects were randomized to receive oral doses of either ABT-333 (8) or Placebo (4) under nonfasting conditions. Serial plasma samples were collected on Day 1 and Days 10 to 13. Plasma samples were also collected prior to the morning dose on Days 2, 3, 5, 7, 8, and 9. Pharmacokinetic parameters of ABT-333 (Cmax, Ctrough, Tmax, and
AUC12) were calculated using WinNonlin version 5.2.1 (Pharsight, Mountain View, CA). Safety and tolerability were monitored throughout the study. In the 200 mg dose group, on Day 11, an additional morning dose of ABT-333 was co-administered with 400 mg ketoconazole.
Results: The mean maximum plasma concentration (Cmax) and area under the concentration-time curve (AUC12) for the 200 mg dose group on Day 10 were 405 ng/mL and 2807 ng·hr/mL, respectively. The dose-normalized maximum plasma concentration (Cmax) and area under the concentration-time curve AUC were comparable for the 200 to 1000 mg BID dose groups. Minimal
accumulation between Days 1 and 10 was noted with multiple dose administration. The Cmax and AUC values for the evening dose and morning dose on Day 10 were comparable indicating no significant diurnal variation in the pharmacokinetics of ABT-333.
Ketoconazole co-dosing resulted in a modest increase in ABT-333 Cmax (50%) and AUC (64%). ABT-333 was well tolerated with few adverse events and no clinically significant dose-related changes from baseline in laboratory values, vital signs, or ECG recordings.
Conclusions: The pharmacokinetics of ABT-333 were dose proportional following 10 days of administration. ABT-333 showed minimal accumulation with no diurnal variation. As the fold increase in ABT-333 exposure in the presence of ketoconazole was between 1.25 and 2, based on FDA guidance, ABT-333 is not a sensitive substrate of CYP3A. ABT-333 was well tolerated following multiple dosing for 10 days.
Note: Additional doses of 600 and 1000 mg BID were added to the study following abstract submission.
INTRODUCTION
ABT-333 is a novel non-nucleoside NS5B polymerase inhibitor being developed for the treatment of HCV genotype 1 infection.
ABT-333 exhibits highly potent and selective activity against genotype 1a and 1b HCV polymerases [Poster Number: 955].
Metabolite profiles in hepatocytes indicate a primary oxidative pathway followed by subsequent conjugation primarily as a glucuronide.
ABT-333 is highly distributed to the liver in rats and dogs and produces 12-hour drug levels in humans at significant multiples over its IC50.
ABT-333 was safe and well tolerated in single ascending doses up to 2000 mg in healthy volunteers [Poster number: 956].
The effect of CYP3A inhibition on ABT-333 pharmacokinetics was evaluated by co-dosing ketoconazole with ABT-333.
Ketoconazole is a potent CYP3A inhibitor recommended by the FDA to evaluate the effect of CYP3A inhibition on CYP3A substrates. Per the FDA guidance a substrate with less than 2-fold increase in exposure when co-dosed with ketoconazole would not be considered a sensitive substrate of CYP3A.
OBJECTIVES
Determine multiple dose safety, tolerability, and pharmacokinetics of ABT-333 under nonfasting conditions in healthy adult subjects.
Determine the effect of single dose administration of ketoconazole on steady state ABT-333 pharmacokinetics.
METHODS
Study Design
· This was a Phase 1, multiple-dose, randomized, placebo-controlled, parallel group study conducted according to a sequential design.
· The protocol was reviewed and approved by an Independent Review Board prior to the start of the study. All subjects who participated in the study gave written informed consent prior to starting any study procedures.
· Adult male and female subjects in general good health were selected to participate in the study according to the selection criteria. In Groups 1 to 3, twelve subjects were randomized in a 2:1 fashion to receive multiple doses of ABT-333 or matching placebo from Days 1 through 10. In Group 4, ten subjects were randomized in a 4:1 fashion to receive multiple doses of ABT-333 or matching placebo from Days 1 through 10. Safety, tolerability, and preliminary pharmacokinetic results were assessed in each group prior to the start of the next group.
· The ABT-333 doses in Groups 1 to 4 were 200 mg BID, 400 mg BID, 600 mg BID, and 1000 mg BID.
· Subjects in Group 1 received a single dose of ABT-333 (or matching placebo) and 400 mg ketoconazole on Day 11.
Methods
Pharmacokinetic Assessments
· Complete pharmacokinetic profiles for ABT-333 were drawn on Days 1 and 10 following the morning dose. For Group 1, a complete pharmacokinetic profile was also drawn on Day 11.
· ABT-333 trough samples were collected immediately prior to the morning dose on Days 2, 3, 5, 7, 8, and 9.
· ABT-333 concentrations were determined by a validated high performance liquid chromatography tandem mass spectroscopy method (HPLC/MS/MS) with a lower limit of quantitation of 1 ng/mL.
· Pharmacokinetic parameters of ABT-333 alone (Days 1 and 10) and in the presence of ketoconazole (Day 11) were estimated by noncompartmental methods using WinNonlin Professional version 5.2.1. (Pharsight Corporation, CA, USA).
Statistical Methods
· Statistical analysis was conducted using SAS version 9.13 (Cary, NC).
· Dose proportionality analysis was conducted on dose-normalized and log-transformed Cmax and AUC12 values.
· The effect of ketoconazole on ABT-333 pharmacokinetics was assessed by comparing log-transformed Cmax and AUC12 on Day 11 to those on Day 10 using analysis of variance (ANOVA). Least square mean ratios and 95% confidence intervals were calculated to quantify the effect of ketoconazole on ABT-333.
· Adverse events were coded using the Medical Dictionary for Regulatory Activities (MedDRA version 11.1). The number and percentage of subjects having treatment-emergent adverse events were tabulated with a breakdown by ABT-333 regimen.
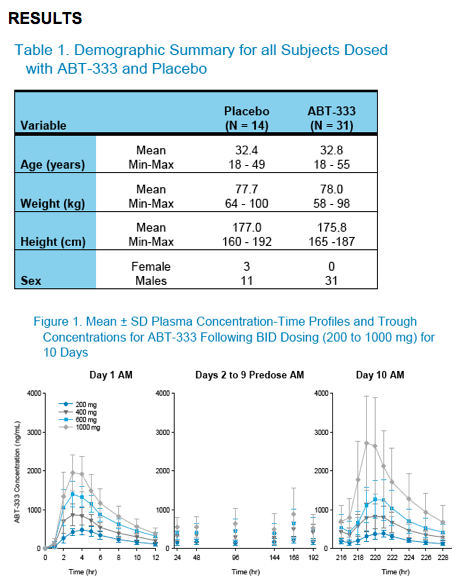
Cmax and AUC12 values for ABT-333 increased with increasing dose from 200 mg to 1000 mg in a dose-proportional manner as shown in Table 2.
Mean Tmax values were 3-4 hours across all dose groups.
Steady state levels of ABT-333 were reached by Day 2 following BID dosing of ABT-333 200 mg to 1000 mg.
Pharmacokinetic parameters of ABT-333 following morning and evening doses on Day 10 were similar indicating that there is minimal diurnal variation in ABT-333 pharmacokinetics (Table 3).
The mean terminal half-life following ABT-333 dosing for 10 days were comparable to the mean terminal half-life from the single ascending dose study for ABT-333 in healthy volunteers [Abstract: 2046].
Effect of Ketoconazole
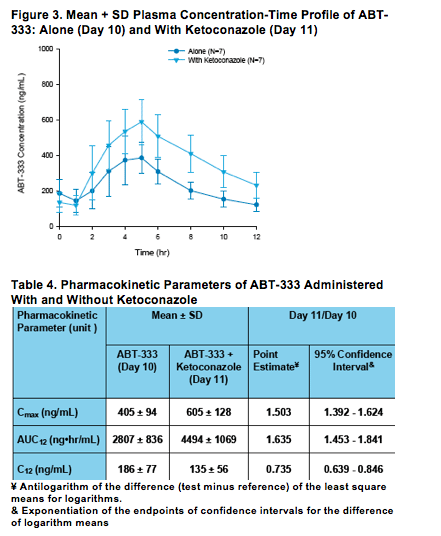
Following co-administration of 400 mg ketoconazole with 200 mg BID ABT-333 at steady-state, the mean ABT-333 Cmax and AUC12 values were 50 and 64% higher as compared to ABT-333 administration without ketoconazole, Figure 2 and Table 4.
Ketoconazole did not appear to affect Tmax or the elimination phase of ABT-333.
As the fold increase in AUC was between 1.25 and 2, based on FDA guidance, ABT-333 is not a sensitive substrate of CYP3A.
Safety Results
ABT-333 was safe and well tolerated across all dose groups following multiple dosing for 10 days (N=31).
No adverse event occurred in more than 2 subjects.
No association between the character or frequency of adverse events or laboratory abnormalities and ABT-333 dose was observed.
Adverse events were mild in the majority of subjects reporting adverse events.
There were no severe or serious adverse events (SAEs).
There were no clinically significant laboratory abnormalities.
Two subjects discontinued the study prematurely.
-- Study-drug related AEs (nausea, diarrhea, vomiting) on Day 5 in the 1000 mg BID dose group. No other subject in this dose group experienced nausea, diarrhea or vomiting.
-- Elevated ALT which occurred prior to dosing in the 1000 mg BID dose group.
|
| |
|
 |
 |
|
|