 |
 |
 |
| |
4-Week Virologic Response and Safety of ABT-450 Given with Low-dose Ritonavir (ABT-450/r) First As 3-Day Monotherapy Then in Combination with Pegylated Interferon Alpha-2a and Ribavirin (SOC) in Genotype 1 (GT1) HCV-infected Treatment-naïve Subjects
|
| |
| |
Reported by Jules Levin
AASLD Oct 29-Nov 3 2010, Boston
Eric Lawitz1, Isabelle Gaultier2, Fred Poordad3, Edwin DeJesus4, Kris V. Kowdley5, Gladys Sepulveda6, Daniel Cohen2, Rajeev Menon2, Lois M. Larsen2, Thomas Podsadecki2, Barry Bernstein2
1Alamo Medical Research, San Antonio, TX, United States; 2Global Pharmaceutical R & D, Abbott Laboratories, Abbott Park, IL, United States; 3Hepatology, Cedars-Sinai Medical Center, Los Angeles, CA, United States;
4Orlando Immunology Center, Orlando, FL, United States; 5Digestive Disease Institute, Virginia Mason Medical Center, Seattle, WA, United States

INTRODUCTION
· ABT-450 is a potent acylsulfonamide protease inhibitor of the hepatitis C virus (HCV) identifi ed as a lead compound by Abbott and Enanta, and being developed for the treatment of HCV genotype 1 infection in combination with
other anti-HCV agents
· ABT-450 has inhibitory concentrations in the sub-nanomolar range in genotype 1a and 1b subgenomic replicon systems in the absence of human
serum
· Ritonavir (RTV) co-administration boosted the pharmacokinetics of ABT-450 with ABT-450 Cmax and AUC increased 28- to 48-fold1; therefore, ABT-450 is being developed with low dose ritonavir (ABT-450/r) to enhance exposure and allow once-daily dosing
· ABT-450/r was safe and well tolerated in single and multiple dose studies in healthy volunteers1, 2
· We present here preliminary results at week 4 of the fi rst study of ABT-450/r in HCV-infected subjects
OBJECTIVE
To analyze the effi cacy and safety of various doses of once-daily ABT-450/r
given alone for 3 days of monotherapy, followed by co-administration of
ABT-450/r with standard of care (SOC) through 4 weeks of treatment.
METHODS
Study Design
· Study M11-602* is an on-going randomized, placebo-controlled, blinded
(active versus placebo), dose ranging, phase 2a clinical trial. In this study, three
cohorts of subjects were randomized to receive various doses of 1 of 3 direct
acting antiviral (DAA) agents currently in clinical development: ABT-450/r, or one of 2 non-nucleoside polymerase inhibitors (ABT-072 or ABT-333). This study is fully enrolled.
· Data from the non-nucleoside polymerase inhibitor-containing arms will be
presented elsewhere. We are presenting here the preliminary results from the
first 4 weeks of treatment with ABT-450/r or placebo.
· To be eligible for enrollment in study M11-602, subjects had to meet the
following inclusion criteria:
- age 18 to 65 years
- body mass index (BMI) ≥18 and <35 kg/m2
- chronic HCV genotype 1 infection for at least 6 months prior to study
enrollment
- plasma HCV RNA level ≥100,000 IU/mL at screening
- liver biopsy within the past 3 years with histology consistent with HCV
induced liver damage
· Exclusion criteria included:
- liver biopsy with a METAVIR fi brosis score of 3 or 4
- positive test result for hepatitis B surface antigen or anti-HIV antibodies
- history of major depression within the 2 years prior to enrollment
- unresolved clinically signifi cant diseases other than HCV
· Subjects were randomized to one of 3 doses of ABT-450/r (50/100 mg,
100/100 mg or 200/100 mg) or placebo once daily for 3 days, followed by
ABT-450/r or placebo in combination with standard of care (SOC) consisting
of pegylated interferon alfa-2a 180 µg/week + weight-based ribavirin 1000-
1200 mg/day through week 12. At week 12, ABT-450/r or placebo was
discontinued and subjects received SOC alone through week 48 as shown in Figure 1.
· Subjects were confi ned to the study site from study day -1 until after the study procedures were completed on day 4. Subjects had subsequent out-patient study visits on days 5, 11, 18, and 28, and at weeks 6, 8, 10, and 12, followed by monthly visits from weeks 16 to 48 while on SOC treatment, then at weeks 52, 56, 60 and 72 during the follow-up period.
· Study procedures included monitoring of adverse events, physical
examination, vital signs, 12-lead ECGs, and phlebotomy for analysis of
pharmacokinetic parameters, HCV RNA level, and hematology and clinical
chemistry testing
*The M11-602 study is registered with ClinicalTrials.gov, NCT01074008.
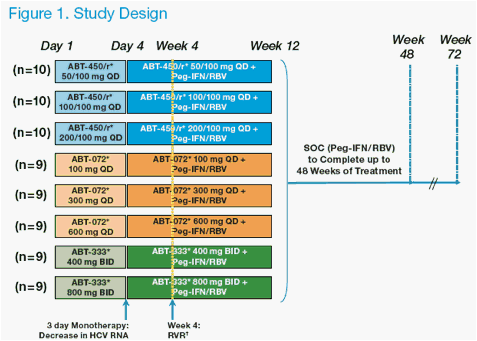
*Placebo Controlled: n=2 placebo per ABT-450/r group and n=1 placebo per ABT-072 and ABT-333 group
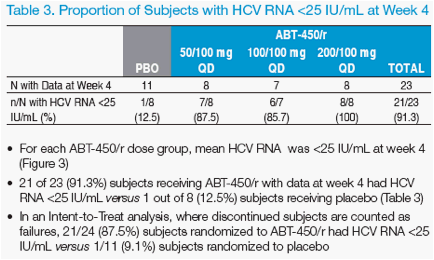
RVR: HCV <25 IU/mL (LOQ) at Week 4
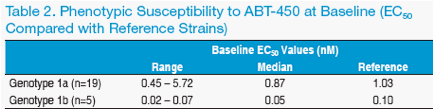
Determination of Baseline Susceptibility to ABT-450
· HCV viral RNA was isolated from a baseline sample collected prior to the fi rst
dose of ABT-450/r, and the region of the virus encoding the NS3 protease
domain was amplifi ed by RT-PCR
· After cloning of this region into the appropriate HCV genotype 1a or 1b
subgenomic replicon shuttle vector, EC50 values were determined using a
transient transfection replicon assay with a fi refl y luciferase reporter
· Replicon reference strains were H77 and Con1 for genotypes 1a and 1b,
respectively
Efficacy Analyses
· HCV RNA was measured using Roche COBAS TaqMan (LLOQ = 25 IU/mL
and LLOD = 10 IU/mL)
· Virologic response was assessed as HCV RNA decrease from baseline in
log10 IU/mL at each time point
· The study primary endpoint was the mean maximum decrease in HCV RNA
during the 3-day monotherapy period (through day 4 predose), which was
compared among ABT-450/r treatment groups and placebo using a one-way
ANCOVA with treatment group as factor and baseline HCV RNA levels as
covariate3
· The proportion of subjects with HCV RNA <25 IU/mL was assessed at week
4 (protocol-defi ned rapid virologic response, RVR)
· Pair-wise comparisons to placebo in mean change from baseline in HCV
RNA to each time point though week 4 were performed using ANCOVA with
treatment group as factor and baseline HCV RNA levels as covariate
RESULTS
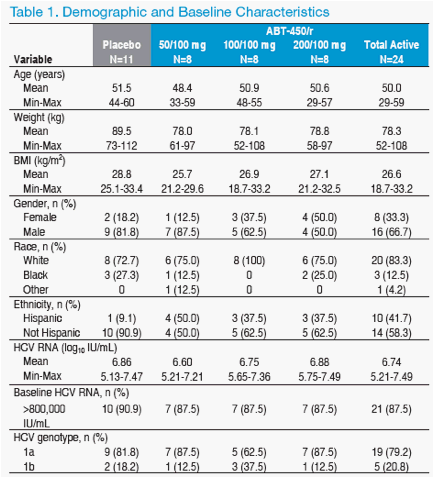
Subject Disposition and Baseline Characteristics
· A total of 24 HCV genotype 1-infected subjects were enrolled and
randomized to 1 of 3 doses of ABT-450/r
· Eleven subjects in total in the study were randomized to receive placebo; all
11 are pooled in these analyses
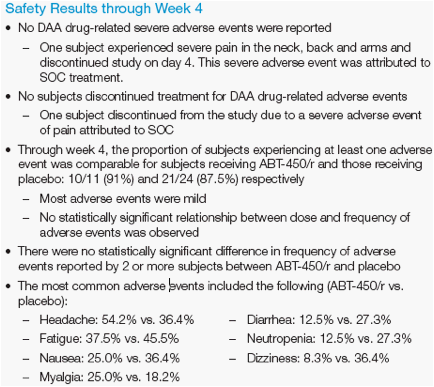
· Four subjects discontinued study before week 4, none for DAA drug-related
adverse events: one was lost to follow-up, two withdrew consent and one
discontinued all treatment due to a severe adverse event of pain attributed to
SOC
· Demographic and baseline characteristics were similar between groups
(Table 1)
· 80% of subjects overall were infected with genotype 1a
· 89% of subjects overall had baseline HCV RNA >800,000 IU/mL
Baseline phenotypic susceptibility of isolates from subjects randomized to
ABT-450/r ranged from <1 to 5.7 times the EC50 of the reference strains
(Table 2).
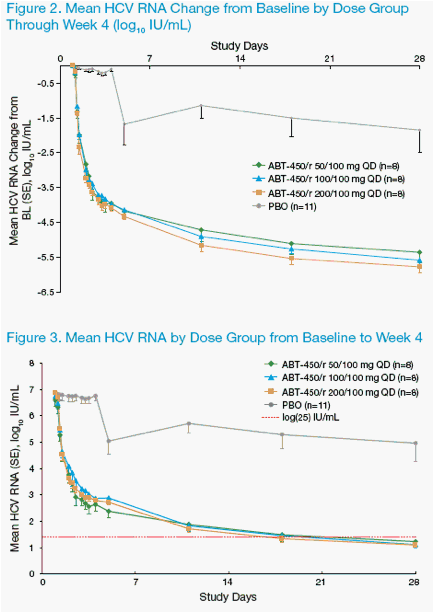
Efficacy Through Week 4
· The primary endpoint of the study was the mean maximum change in HCV
RNA during the 3-day monotherapy with ABT-450/r or placebo. Through 3
days of monotherapy, response was similar in the 3 ABT-450/r dose groups:
Mean maximum decreases from 3.89 to 4.11 log10 IU/mL were observed in all
3 dose groups compared to 0.36 log10 IU/mL for the placebo group (p<0.001
for each comparison)3.
· Beginning at 8 hours post the fi rst dose on day 1 through week 4, all
3 ABT-450/r dose groups had mean HCV RNA decreases from baseline
statistically signifi cantly different from placebo at every time point
· At week 4, the mean (± SD) HCV RNA decrease from baseline was 5.58 ±
0.65 log10 IU/mL for subjects on ABT-450/r versus 1.86 ± 1.90 log10 IU/mL for
subjects on placebo (p<0.001)
· The mean HCV RNA (range) at week 4 was 1.15 (1.00 - 2.87) log10 IU/mL for
subjects on ABT-450/r versus 4.97 (1.00 - 7.21) log10 IU/mL for subjects on
placebo
· Through week 4, virologic response was similar in the 3 ABT-450/r dose
groups (Figures 2 and 3)
· Although few subjects with genotype 1b enrolled, there were no apparent
differences in virologic response to ABT-450/r or placebo between genotypes
1a and 1b
· No subject receiving ABT-450/r experienced a virologic rebound (increase
≥0.5 log10 IU/mL from nadir) through the 4 weeks of ABT-450/r + SOC
treatment

REFERENCES
1. Menon R, et al. Pharmacokinetics and Tolerability of the HCV Protease Inhibitor ABT-450 Following Single Ascending Doses in Healthy Adult Volunteers with and Without Ritonavir. Annual Meeting of HepDART 2009; Poster 57.
2. Bernstein B, et al. Pharmacokinetics, Safety and Tolerability of the HCV Protease Inhibitor ABT-450 with Ritonavir Following Multiple Ascending Doses in Healthy Adult Volunteers. Annual Meeting of HepDART 2009; Poster 58.
3. Lawitz E. et al. Initial antiviral activity of the HCV NS3 protease inhibitor ABT-450 when given with low dose ritonavir as 3-day monotherapy: Preliminary results of study M11-602 in genotype 1 (GT1) HCVinfected treatment-naïve subjects. 61st Annual Meeting of the AASLD, Boston, 2010; Abstract #1855.
|
| |
|
 |
 |
|
|