 |
 |
 |
| |
Combination of two complementary nucleotide analogues, PSI-7977 and PSI-938, effectively clears wild type and NS5b: S282T HCV replicons - Comparison with combinations of other antiviral compounds
|
| |
| |
Reported by Jules Levin
EASL Apr 14-18 2010 Vienna Austria
V. Zennou, A.M. Lam, M. Keilman, C. Espiritu , H. Micolochick Steuer, M.J. Sofia, PG Reddy, W. Chang, S. Rachakonda, M.J. Otto, P.A. Furman Pharmasset, Inc., Princeton, NJ, USA.

ABSTRACT
Background: In vitro studies combining direct acting antivirals (DAAs) targeting different HCV proteins have demonstrated varying abilities to suppress the emergence of resistant variants from a wild type replicon. The combination of two complementary DAAs, RG7128, (NS5b) and RG7227 (NS3/4a) suppressed the emergence of variants resistant to either compound over 13 days in genotype 1 patients. Like HIV, it could be expected that complementary nucleosides/tides would effectively suppress the emergence of resistant variant as well. We examined the capacity of various combinations of DAAs, including combinations of NS5b inhibitors and combinations of NS5b plus NS3/4a inhibitors to suppress the emergence of resistant variants in the HCV replicon.
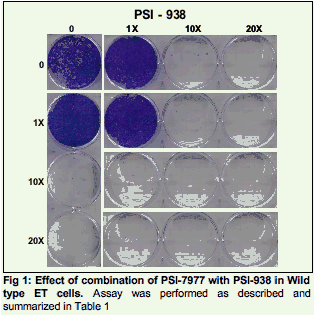
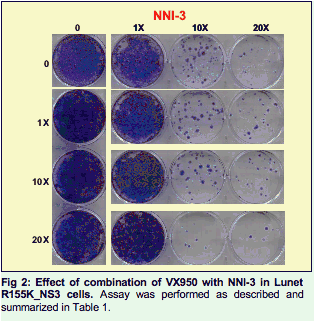
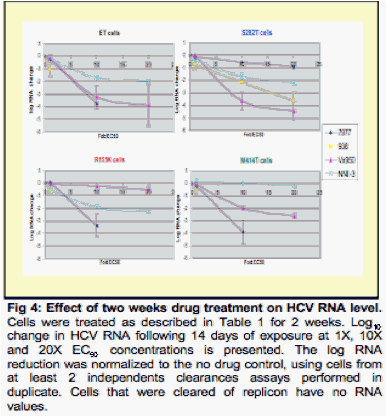
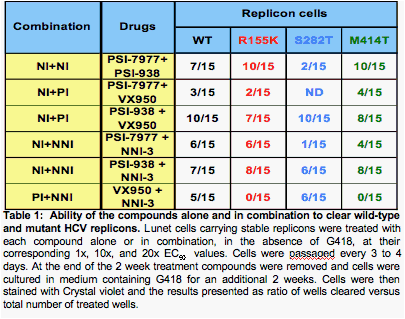
Methods: The studies compared combinations of two nucleotide prodrugs, PSI-7977 a pyrimidine analog and PSI- 352938 (PSI-938) a purine analog, with combinations of either a protease inhibitor (VX-950 / telaprevir) plus a benzothiadiazine non-nucleoside polymerase inhibitor or a nucleotide prodrug plus either a protease inhibitor or a non-nucleoside inhibitor. Combination studies were performed using the wild-type or mutant (NS3:R155K, NS5b: S282T, NS5b:M414T) luciferase expressing ET replicons. Drug-drug combinations were evaluated by combining each inhibitor at various molar ratios based on their EC50 values. After 4 days, the activity of the combinations was quantified using the luciferase activity in replicon cells. Combinations were also evaluated in replicon clearance assays. Wild-type and mutant replicon cells were treated with 1x EC50, 10x EC50 and 20x EC50 with either drug alone or in combination for 2 weeks, followed by G418 selection without drug for two weeks. Surviving cells were stained with crystal violet and the levels of HCV RNA were quantified using real-time PCR.
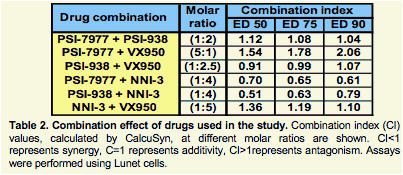
Results: The combination of the two nucleotide prodrugs, PSI-7977 and PSI-938, was additive to synergistic and was able to clear wild type and S282T replicon cells. The combinations of PSI-7977 or PSI-938 with either the non-nucleoside or a protease inhibitor were also able to clear wild type cells. A comparison of these combinations is presented.
Conclusion: These in vitro results indicate that combinations of two nucleosides/tides targeting NS5b as well as DAAs targeting different HCV proteins effectively suppress resistant replicons.
INTRODUCTION
The current standard of care is Pegylated interferon plus ribavirin. Because of the adverse effects often associated with standard of care serious attention is being given to combining two direct acting antiviral agents. Because of their high barrier to resistance, nucleoside/tide combinations could become the backbone of HCV therapy.
Two nucleotides prodrugs, PSI-7977 and PSI-938, were combined together or with either a protease inhibitor (VX950) or a benzothiadiazine non nucleoside site 3 Inhibitor (NNI-3).
Each combination was analyzed for its ability to clear a wild-type HCV 1b replicon or replicons containing mutations that confer resistance to nucleoside analogs (S282T), VX-950 (R155K) or a non nucleoside inhibitor (M414T).
EC50 values of the compounds used
- PSI-7977: 0.03µM
- PSI-938: 0.1µM
- VX950: 0.4µM
- NNI-3: 0.1µM
RESULTS

|
| |
|
 |
 |
|
|