 |
 |
 |
| |
SILEN-C2: Early antiviral activity and safety of BI 201335 combined with peginterferon alfa-2a and ribavirin (PegIFN/RBV) in chronic HCV genotype 1 patients with non-response to PegIFN/RBV
|
| |
| |
Reported by Jules Levin
EASL Apr 14-18 2010 Vienna Austria
M. Sulkowski,1 M. Bourliere,2 J.-P. Bronowicki,3 A. Streinu-Cercel,4 L. Preotescu,4 T. Asselah,5 J.-M. Pawlotsky,6 S. Shafran,7 S. Pol,8 F.A. Caruntu,4 S. Mauss,9 D. Larrey,10 C. Häfner,11 Y. Datsenko,11 J.O. Stern,12 R. Kubiak,11 G. Steinmann11
On behalf of the SILEN-C2 study group
1Department of Viral Hepatitis, Johns Hopkins University, Baltimore, MD, USA; 2Hopital Saint Joseph, Marseille, France; 3Hopital de Brabois, Vandoeuvre, France; 4Prof. Dr. Matei Bals, Institute of Infectious Diseases 1, Bucharest, Romania; 5Hopital Beaujon, Clichy, France; 6Hopital Henri Mondor, Créteil, France; 7University of Alberta, Edmonton, AB, Canada; 8Hopital Cochin, Paris, France; 9Center for HIV and Hepatogastroenterology, Düsseldorf, Germany; 10Hopital Saint-Eloi, Montpellier, France; 11Boehringer Ingelheim Pharma GmbH & Co. KG,
Biberach/ Riβ, Germany; 12Boehringer Ingelheim Pharmaceuticals, Ridgefield, CT, USA



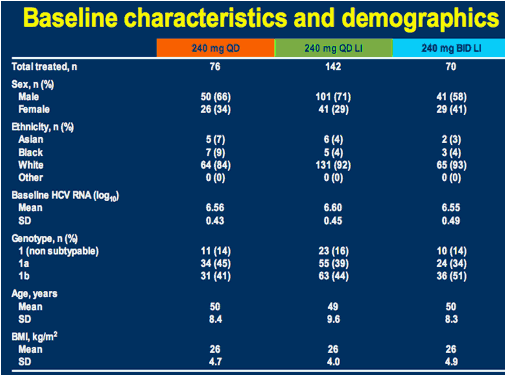
LI = 3-day lead-in; BMI = body mass index
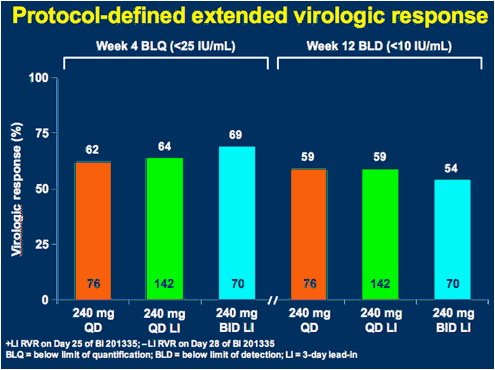
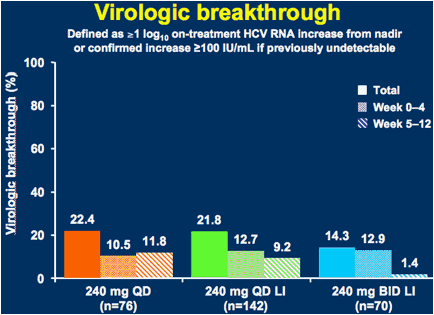
LI = 3-day lead-in
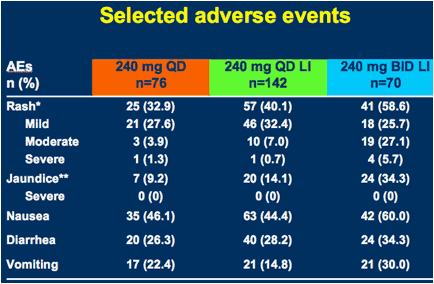
*Including photosensitivity
**Due to isolated unconjugated hyperbilirubinemia
AEs = adverse events; LI = 3-day lead-in
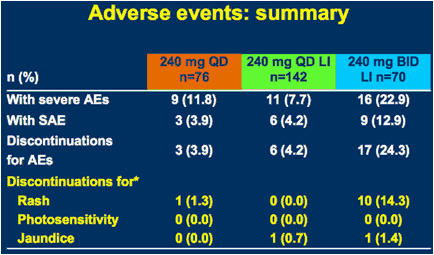
*Other discontinuations mainly due to general disorders and administration site conditions, GI and others
AE = adverse event; SAE = serious adverse event; LI = 3-day lead-in
|
| |
|
 |
 |
|
|