 |
 |
 |
| |
Slow Responders Benefit From 72 Weeks Peg/Rbv -
Predicting Treatment Outcome Among Slow Responders: A Retrospective Analysis of the SUCCESS Study
|
| |
| |
Reported by Jules Levin
EASL Apr 14-18 2010 Vienna Austria
M. Buti,1 V. G. Morozov,2 V. V. Rafalskiy,3 F. Wong,4 J. Sumskiene,5 R. Faruqi,6 L. D. Pedicone,6 R. Esteban1
1Liver Unit Hospital General Universitario Valle Hebron and Ciber-ehd del Instituto Carlos III, Barcelona, Spain; 2Hepatolog Medical Center, Samara, Russia; 3Smolensk State Medical Academy, Smolensk, Russia; 4Toronto General Hospital, Toronto, ON, Canada;
5Clinic of Gastroenterology, Kaunas University of Medicine, Kaunas, Lithuania; 6Schering-Plough Corporation, now Merck & Co., Inc., Whitehouse Station, New Jersey, U.S.A.
from Jules: in other words this study suggests discontuning therapy at week 12 if viral load decline is marginally less than desired may be premature and if continuing therapy shows signs of improved response & undetectable at week 24 then 72 weeks might do it for the patient. I always thought that anyway!

ABSTRACT
Background: Several studies have examined a 72-week (wk) treatment duration with peginterferon plus ribavirin for patients with chronic hepatitis C (CHC) genotype 1 infection; however, there is no consensus regarding the subgroup of patients for which extended treatment should be considered.
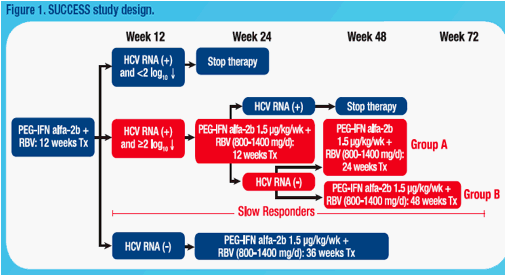
Aim: To identify on-treatment predictors of sustained virologic response (SVR) in slow responders (≥2log decline in HCV RNA yet detectable at wk 12 and
undetectable at wk 24) treated for 48 or 72 wks.
Method: This was an open-label, randomized, international study of patients with CHC genotype 1. Slow responders were randomized to receive peginterferon alfa-2b (1.5 µg/kg/wk) plus ribavirin (800-1400 mg/d) for 48 or 72 wks.
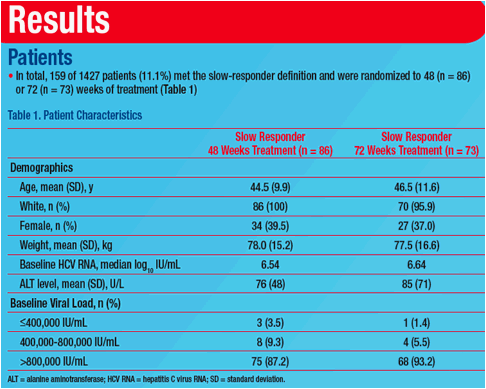
Results: 1,428 patients were enrolled of which 159 slow responders
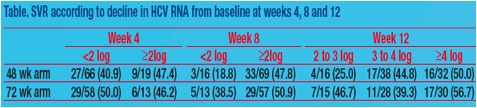
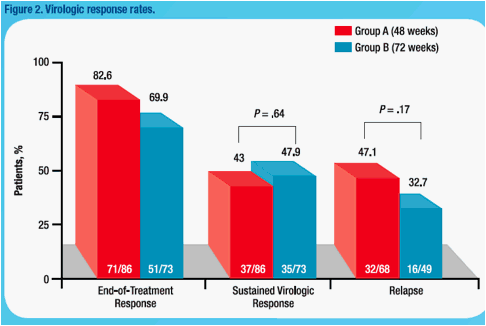
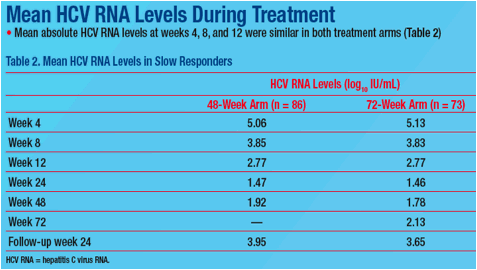
were randomized to 48 (n=86) or 72 wks treatment (n=73). SVR rates were 43.0% and 47.9% in 48 wk and 72 wk arms. Mean absolute HCV RNA levels at wk 4, 8, and 12 were similar in both arms. Change in HCV RNA at wk 4 was
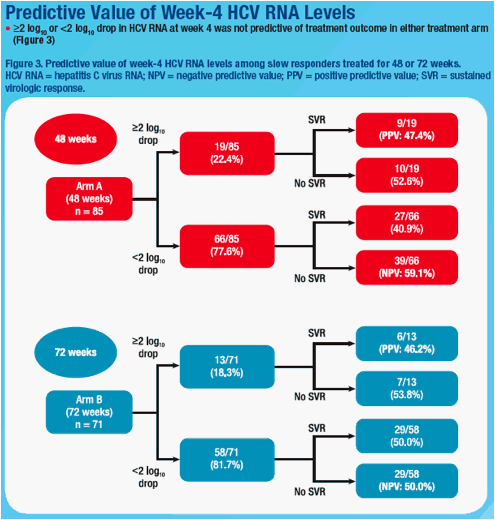
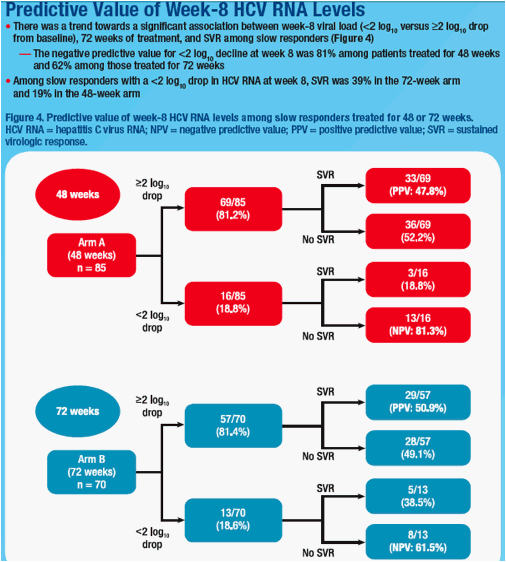
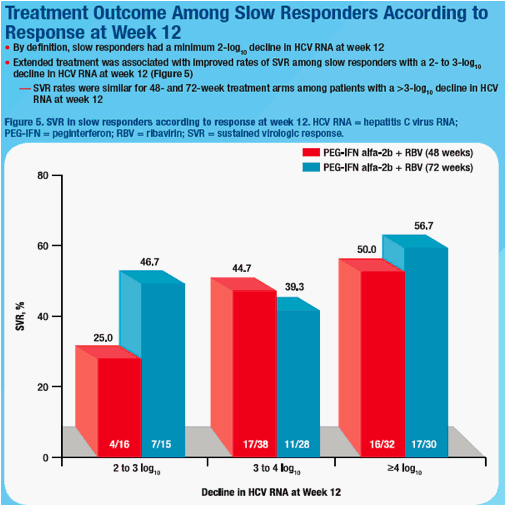
not predictive of SVR (Table). Among slow responders with <2log decline at wk 8, SVR rates were 18.8% and 38.5% in 48 wk and 72 wk arms. Extended treatment did not improve SVR among slow responders with ≥2log decline at
wk 8 or with ≥3log decline at wk 12; however, SVR was 25.0% and 46.7% in slow responders with 2-3log decline at week 12 when treated for 48 or 72 wks. Logistic regression revealed a weak association between week 8 viral load (<2 versus ≥2log decline) and SVR (odds ratio = 2.504; 95% CI, 0.948 to 6.613; P = .064).
Conclusion: Slow responders with <2log decline from baseline at wk 8, 2-3log decline at wk 12 and undetectable HCV RNA at wk 24 represent a group of patients who could benefit most from extended treatment.




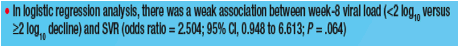

|
| |
|
 |
 |
|
|