 |
 |
 |
| |
PHASE IIB STUDY OF BALAPIRAVIR (RG1626; NUCLEOSIDE ANALOGUE INHIBITOR OF HCV POLYMERASE) PLUS PEGINTERFERON ALFA-2A (40KD) AND RIBAVIRIN FOR CHRONIC HEPATITIS C GENOTYPE 1: FINAL RESULTS
|
| |
| |
Reported by Jules Levin
EASL Apr 14-18 2010 Vienna Austria
S. Zeuzem,1 D.R. Nelson,2 P. Andreone,3 P. Ferenci,4 R. Herring,5 D.M. Jensen,6 P. Marcellin,7 P.J. Pockros,8 M. Rodriguez-Torres,9 L. Rossaro,10 V.K. Rustgi,11 T. Sepe,12 M. Sulkowski,13 I.R. Thomason,14 E. Yoshida,15 A. Chan,16 G. Hill17
1J.W. Goethe University Hospital, Frankfurt, Germany; 2University of Florida, Gainsville, FL, USA; 3Dipartimento di Medicina Clinica, Universita di Bologna, Bologna, Italy; 4Medical University of Vienna, Vienna, Austria; 5Nashville Gastrointestinal Specialists, Inc., Nashville, TN, USA; 6Center for Liver Diseases, Chicago, IL, USA; 7Hopital Beaujon, Clichy, France; 8Scripps Clinic, Torrey Pines, San Diego, La Jolla, CA, USA; 9Fundacion de Investigacion De Diego Santurce, Puerto Rico and Ponce School of Medicine, Santurce, PR, USA; 10Division of
Gastroenterology and Hepatology, University of California, Davis Medical Center, Sacramento, CA, USA; 11Georgetown University Medical Center, Fairfax, VA, USA; 12University Gastroenterology, Providence, RI, USA; 13Johns Hopkins University School of Medicine, Baltimore, MD, USA; 14MountainWest Gastroenterology, Transplant Department, Intermountain Medical Center, Salt Lake City, UT, USA; 15Division of Gastroenterology, University of British Columbia, Vancouver, BC, Canada; 16Roche, Nutley, NJ, USA; 17Roche, Palo Alto, CA, USA
Discussion
· The severity of the haematological toxicity associated with longer term administration of balapiravir in combination with peginterferon alfa-2a (40KD) and ribavirin in HCV infected patients was unexpected from non-clinical toxicity studies, where balapiravir had been well tolerated up to 3000 mg/kg/day (highest dose tested) for three months in mice, up to 2000 mg/kg/day (highest dose tested) in rats for six months, and up to 1500 mg/kg/day (highest dose tested) in female dogs for nine months. These studies had not identified any target organ toxicity. In vitro studies in primary human cells had indicated measurable inhibition of human bone marrow stem cell differentiation with low intrinsic cytotoxicity. Bone marrow stem cell differentiation into erythroid precursor cells had been the most sensitive to inhibition, as compared to differentiation into myeloid or megakaryocytic lineages. Inhibition of erythroid differentiation had occurred with a potency more than 80-fold lower as compared to the comparator nucleoside analogue zidovudine. Consistent with the in vitro analyses, the most obvious dose-dependent haematological effect of balapiravir in monotherapy was a reduction in haemoglobin, erythrocyte counts, and reticulocyte counts that was reversible upon discontinuation, consistent with inhibition of precursor differentiation.3
· The mechanism responsible for the haematological toxicity associated with balapiravir in combination with peginterferon alfa-2a (40KD) and ribavirin is unknown at this time, although it appears that this combination may have exacerbated a direct and broad-based suppressive effect of balapiravir on bone marrow and also resulted in an uncompensated reduction in lymphocytes. The safety profile of balapiravir alone and in combination with peginterferon alfa-2a (40KD) and ribavirin was unique and different from those observed with other nucleoside inhibitors of HCV polymerase. An investigation into possible mechanisms related to these observations is in progress and will be published in due course.
Conclusions
· When combined with SOC, balapiravir significantly increased the rate of RVR at week 2 and 4 and cEVR at week 12 in genotype 1 patients; however safety-related dose reductions and discontinuations for both balapiravir and SOC significantly impacted the SVR in this Phase IIb study.
· The safety profile of balapiravir alone and in combination with peginterferon alfa-2a (40KD) and ribavirin was unique and different from those observed with other nucleoside inhibitors of HCV polymerase.
· Further development of balapiravir in combination with peginterferon alfa-2a (40KD) and ribavirin for the treatment of chronic hepatitis C has been halted because of the unfavourable benefit:risk ratio revealed in this study. Non-clinical investigations are ongoing to explore the mechanism of balapiravir-related toxicity.
· Longer-term experience with other investigational nucleoside polymerase inhibitors is currently lacking and close monitoring of safety in ongoing studies is warranted.
Introduction
· Several classes of direct-acting antiviral (DAA) agents have been discovered and are being evaluated in human clinical trials with the goal of
increasing cure rates for chronic hepatitis C.
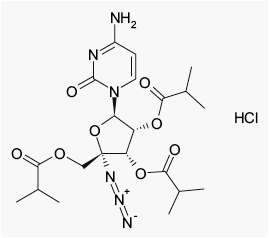
· Balapiravir (RG1626) (Figure 1) is an orally administered prodrug of a potent nucleoside analogue inhibitor (RG1479) (4'-azidocytidine) of the
RNA-dependent RNA polymerase of the hepatitis C virus (HCV).1, 2
Figure 1: Chemical structure of balapiravir (RG1626): 4'-azidocytidine-2',3',5'-triisobutyrate hydrochloride (common name)
· When administered for 14 days in a Phase I monotherapy study at dosages ranging from 1000 to 9000 mg/day balapiravir produced mean reductions in serum HCV RNA levels up to 3.7 log10 with an acceptable safety profile.3 Dose-dependent haematological abnormalities were found to be the dose-limiting toxicity.
· When administered for 28 days in a Phase IIa combination study at a dosage of 3000 mg/day in combination with the standard of care (SOC; peginterferon alfa-2a [40KD] plus ribavirin, [RBV]), the rapid virological response (RVR) rate (HCV RNA <15 IU/mL at week 4) was 74%.4 A synergistic effect of balapiravir and interferon on haematological safety parameters was detected in the Phase IIa study, but these events were manageable through dose-reductions and were reversible upon discontinuation of balapiravir.
· Importantly, balapiravir-resistant HCV variants were not detected during these short-term clinical trials.3, 4
· Dose-dependent severe neutropenia and other haematological abnormalities were observed over short administration in early clinical trials; however, these
events were effectively managed with dose reductions and were reversible upon discontinuation of balapiravir and/or peginterferon alfa-2a (40KD). Rigorous
dose modifications were implemented in order to manage these side effects in this Phase IIb study.
Objective
· The objective of this Phase IIb study was to determine the efficacy and safety of balapiravir (RG1626) in combination with peginterferon alfa-2a (40KD) plus ribavirin in treatment-naive genotype 1 patients.
Methods
Patients
· Adult patients aged 18 to 65 years with HCV genotype 1 infection who had never received treatment for chronic hepatitis C were eligible for the trial.
Treatment
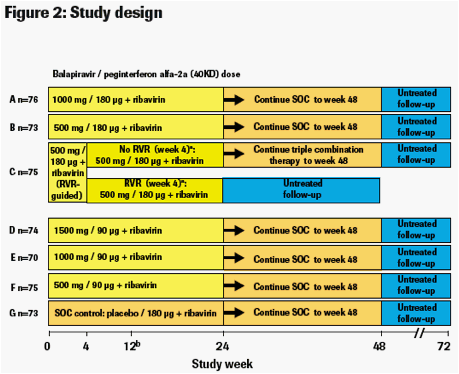
· Eligible patients were randomised to 1 of 7 groups (Figure 2).
· Balapiravir was given twice daily.
· Standard of care (SOC) = peginterferon alfa-2a (40KD) 180 µg/week +
ribavirin 1000/1200 mg/day. Based on the significant neutropenia seen in
earlier studies with balapiravir3,4 this Phase IIb study explored higher doses
of balapiravir with half doses of peginterferon alfa-2a (40KD) plus ribavirin
or lower doses of balapiravir with full doses of peginterferon alfa-2a (40KD)
plus ribavirin in conjunction with rigorous dose modification guidelines to
manage the neutropenia.
a. Therapy was stopped at week 24 in patients with an RVR in group C,
provided that their HCV RNA had remained undetectable at all visits
through to week 22.
b. Due to safety signals that were detected after treatment was initiated, the
protocol was amended on 29 May 2008 specifying that treatment with
balapiravir was to be stopped immediately in all patients in group D, and
that treatment with balapiravir was to be stopped at week 12 rather than
week 24 in all other treatment groups with a corresponding increase in the
length of treatment with the SOC from 24 to 36 weeks. Patients who had
completed more than 12 weeks of treatment with balapiravir were required
to stop balapiravir immediately and switch to SOC to complete 48 weeks.
Outcomes
· The primary efficacy outcome was SVR (HCV RNA <15 IU/mL 24 weeks after the end of scheduled treatment). Undetectable HCV RNA (<15 IU/mL) at week 4
(RVR) and week 12 (complete early virological response, cEVR) were secondary outcomes.
· Efficacy results are presented according to the intention-to-treat principle. Patients who withdrew prematurely and those with missing results were considered not to have had a virological response.
Results
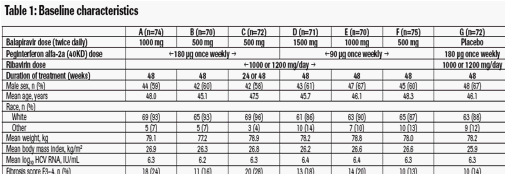
· A total of 516 patients were randomised, and 504 received study medication. The majority of patients were male (58%-67%) and White (86%-96%), the mean age
ranged from 45 to 48 years, the mean weight ranged from 77 to 79 kg, and 13%-28% of patients had advanced fibrosis (F3-4) (Table 1).
The most common reason for withdrawal from treatment with balapiravir or placebo was 'administrative or other reasons' (338, 67%), largely due to the protocol
amendment that resulted in termination of balapiravir treatment in patients who received 12 or more weeks of treatment with the drug.
Efficacy
· High rates of safety-related dose discontinuations and dose reductions with all three study drugs impacted the SVR rates achieved.
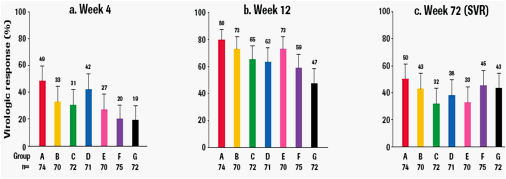
· Virological response rates up to week 12 were consistently higher in balapiravir-containing regimens compared with the SOC group:
- At week 2, the percentage of patients with undetectable HCV RNA (<15 IU/mL) ranged from 3% with SOC group as compared to up to 25% with balapiravircontaining
regimens
- Beyond week 2, safety events began to impact the ability of patients to remain on full dose balapiravir and/or peginterferon alfa-2a (40KD). RVR at week 4
ranged from 19% with SOC group as compared to up to 49% with balapiravir-containing regimens (Figure 3a) and rates of cEVR ranged from 47% with SOC
as compared to up to 80% with balapiravir-containing regimens (Figure 3b).
· SVR rates ranged from 32% to 50% in the 6 balapiravir-containing groups and was 43% with SOC (Figure 3c).
Figure 3: Virological response (HCV RNA <15 IU/mL) during treatment and at the end of follow-up by intention-to-treat.
Vertical bars show the upper limit of the 95% confidence interval.

Safety
· Overall, 28-36% of patients in the balapiravir-containing treatment groups discontinued balapiravir due to safety reasons compared with 6% of patients who discontinued the matching placebo in the SOC group.
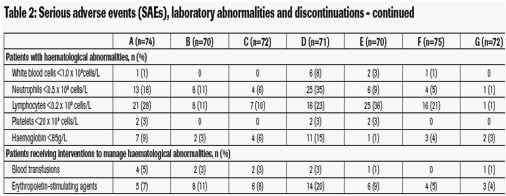
· Serious haematological adverse events, particularly neutropenia and lymphopenia, were observed during the study (Table 2)
- Lymphopenia was the most common reason for discontinuation of balapiravir as a requirement of the protocol amendment
- Lymphopenia resolved in most patients after all study medication was stopped, but some patients remained lymphopenic 1 year after the end of treatment
-- Severity of lymphopenia was not associated with the incidence of infection.
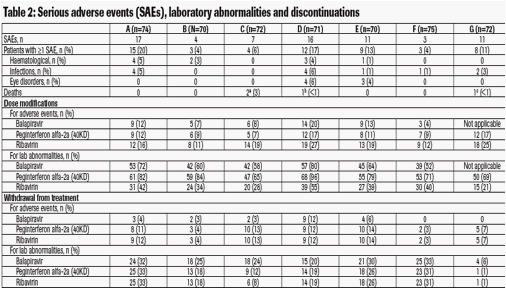
a. One death was attributed to cardiac arrest unrelated to treatment in the opinion of the investigator in a patient with metastatic lymphoma in whom progression of lymphoma was considered to be possibly related to treatment; the second patient death was attributed to suicide (possibly related to treatment);
b. The death was attributed to primary fulminant disseminated varicella infection
(possibly related to treatment);
c. The death was attributed to a road traffic accident (unrelated to treatment)
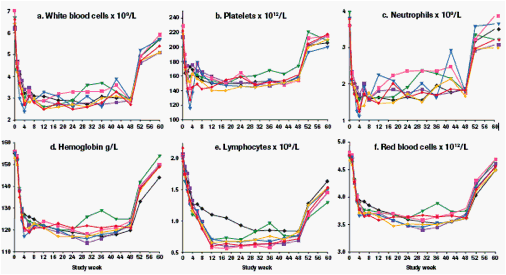
The time course of haematological parameters is shown in Figure 4.
Figure 4: Mean haematological parameters during treatment and follow-up. a) White blood cells, b) platelets, c) neutrophils, d) haemoglobin, e) lymphocytes, f) red blood cells

References
1. Klumpp K, Leveque V, LePogam S. et al. The novel nucleoside analog R1479 (4'-azidocytidine) is a potent inhibitor of NS5B-dependent RNA synthesis and hepatitis C virus replication in cell culture. J Biol Chem 2006;281(7):3793-3799.
2. Brandl M, Wu X, Holper M et al. Physicochemical properties of the nucleoside prodrug R1626 leading to high oral bioavailability. Drug Dev Ind Pharm 2008;34(7):683-691.
3. Roberts SK, Cooksley G, Dore GJ et al. Robust antiviral activity of R1626, a novel nucleoside analog: a randomized, placebo-controlled study in patients with chronic hepatitis C. Hepatology 2008;48(2):398-406.
4. Pockros PJ, Nelson D, Godofsky E et al. R1626 plus peginterferon alfa-2a provides potent suppression of hepatitis C virus RNA and significant antiviral synergy in combination with ribavirin. Hepatology 2008;48(2):385-397.
|
| |
|
 |
 |
|
|