 |
 |
 |
| |
Pharmacokinetics of the HCV Polymerase
Inhibitor ABT-333 in US and Japanese Healthy Volunteers
|
| |
| |
Reported by Jules Levin
EASL 2010, Vienna, Austria, 14-18 April 2010
Rajeev Menon, Daniel Cohen, Walid Awni, Yi-Lin Chiu, Cheri Klein
Corresponding author: Rajeev Menon, Abbott, 100 Abbott Park Road, Dept. R4PK, AP13A, Abbott Park, IL 60064, USA

INTRODUCTION
ABT-333 is a novel non-nucleoside NS5B polymerase inhibitor being developed for the treatment of HCV genotype 1 infection.
ABT-333 exhibits highly potent and selective activity against genotype 1a and 1b HCV polymerases.
ABT-333 was safe and well tolerated in single and multiple ascending doses up to the highest doses studied in healthy volunteers: 2000 mg as a single dose and 1600 mg BID following multiple-dose administration.
When ABT-333 or placebo was administered to HCV patients as monotherapy for 2 days, followed by ABT-333 or placebo plus peginterferon and ribavirin for 26 Days, an HCV RNA decrease of > 3.5 log10 was seen in the ABT-333 treated group as compared to 1.4 log10 in the placebo plus peginterferon and ribavirin treated group.1
OBJECTIVE
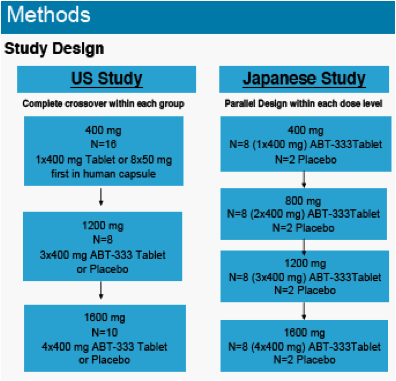
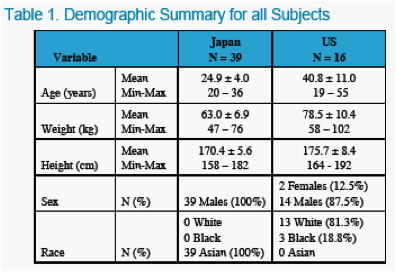
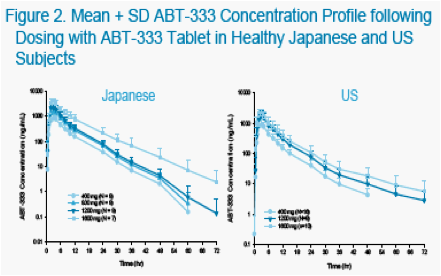
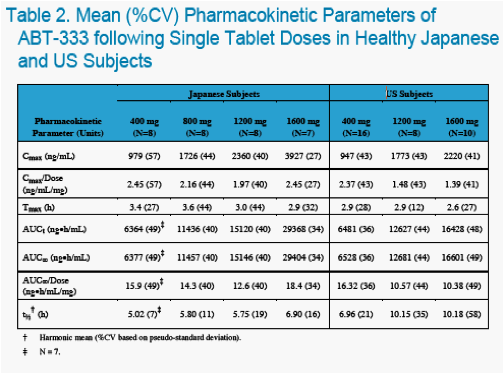
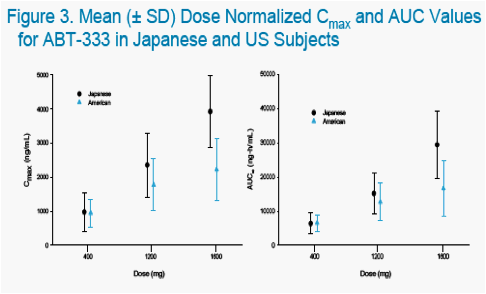
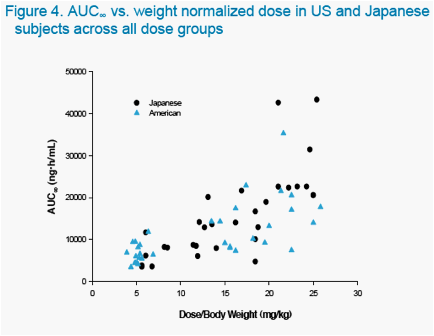
The objectives of the studies were to characterize the pharmacokinetics and tolerability of ABT-333 administered as a 400 mg tablet in non-fasting, healthy US and Japanese adults at increasing doses.




Rodriguez-Torres M et al. Treatment-naïve, genotype-1 HCV-infected subjects show significantly greater HCV RNA decreases when treated with 28 days of ABT-333 plus peginterferon and ribavirin compared to peginterferon and ribavirin alone. 60th Annual Meeting of the American Association for the Study of Liver Diseases (AASLD); 30 October - 03 November 2009 Boston, MA, USA.
|
| |
|
 |
 |
|
|