 |
 |
 |
| |
CHARACTERIZATION OF THE HEPATOSELECTIVE DISTRIBUTION OF ACH-1625: A POTENT, CLINICAL STAGE HCV NS3 PROTEASE INHIBITOR
|
| |
| |
Reported by Jules Levin
EASL 2010
Kathe Stauber, Preeti Kapoor, Christopher Marlor, Gohar Mushtaq, Venkat Gadhachanda, Akihiro Hashimoto, Godwin Pais, Dawei Chen, Avinash Phadke, Mingjung Huang and Milind Deshpande
Achillion Pharmaceuticals, New Haven, CT 06511, USA.
CONCLUSIONS
ACH-1625 shows rapid and selective distribution to the liver. After a single oral dose of 50 mg/kg to the rat, liver concentrations of ACH-1625 at 24 hr post dose were about 500-fold higher than plasma values and 30-fold higher than the
anti-viral EC50. Similarly, in the dog, liver concentrations at 24 hr post dose were 12-fold higher than the antiviral EC50.
The extensive distribution to liver is due, in part, to a rapid, transporter-mediated uptake process as demonstrated with hepatocyte suspensions and OAT P-transfected cells. Uptake characteristics are similar between human and the
preclinical species tested. These data suggest that the partitioning to the liver seen in nonclinical species should occur in humans and is compatible with a once-a-day dosing strategy.
While active uptake is an apparent mechanism for liver partitioning, the data show that < 1% of the dose remains in the liver after 24 hr post dose, indicating that elimination/redistribution of the drug occurs. There was no indication of
prolonged half-life in the liver compared to plasma.
Preclinical safety studies and Phase 1 clinical trials have shown that the drug is well tolerated, highly efficacious and support continued development of ACH-1625.
Background
ACH-1625 is a small molecule inhibitor of the HCV NS3 protease, a viral-specific protein necessary for viral poly-protein processing, and a clinically validated target. ACH-1625 displays a potency of 11 nM (Huang, et al; Abstract 492 ) and demonstrates favorable pharmacokinetic and safety profiles in animals. In recent clinical studies (Detishin et al; LB 2012), ACH-1625 showed a maximum decrease of 4.25 and 3.94 log10 drop of plasma HCV RNA in the 500 mg BID and 600 mg BID dosing groups, respectively, after the 4.5 day treatment. In addition, sustained viral load suppression was observed up
to day 12. ACH-1625 is safe and well-tolerated at doses up to 2000 mg/day for 4.5 days duration.
Interaction with membrane transport proteins, found predominantly in the intestine, brain, kidney and liver, can significantly effect the disposition and pharmacokinetics of many drugs (1,2). The organic anion transporting peptides (OAT Ps) are a large family of cellular uptake transporters (for review see ref 3). At least 3 OAT P proteins are expressed in the liver sinosoid and mediate uptake of endogenous compounds and xenobiotics into hepatocytes. Furthermore, OAT P1B1 and OAT P1B3 are liver specific and show affinity for drugs such as rosuvastatin and fexofenadine. The effectiveness of directly acting anti-HCV small molecules, such as ACH-1625, is dependent on achieving and maintaining effective concentrations in the hepatocyte (the main site of viral replication) for continued suppression of viral replication.
Current work shows the results of in vitro and in vivo studies designed to explore ACH-1625 selective distribution to the liver.

RESULTS
ACH-1625 SHOWS EXCELENT METABOLIC STABILITY
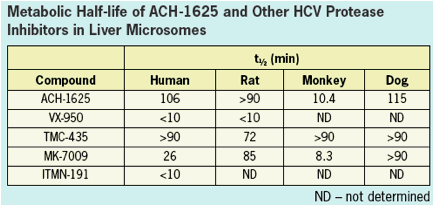
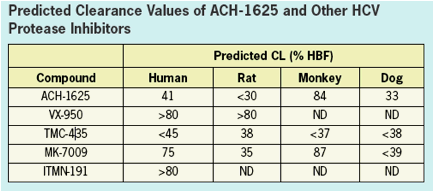
ACH-1625 shows excellent metabolic stability with predicted human metabolic clearance approximating less than
one-half of hepatic blood flow.
ACH-1625 SHOWS SELECTIVE DISTRIBUTION TO LIVER IN THE RAT AND DOG
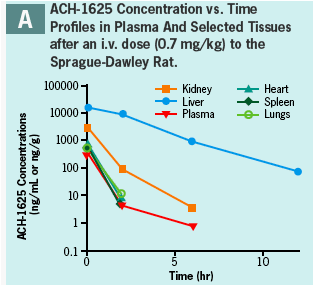
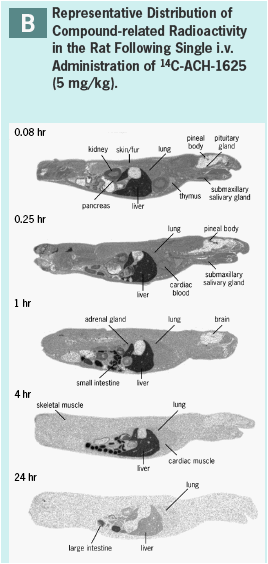
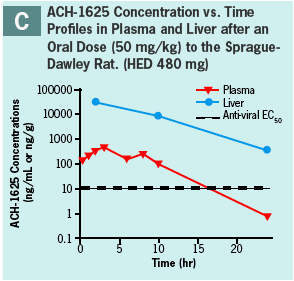
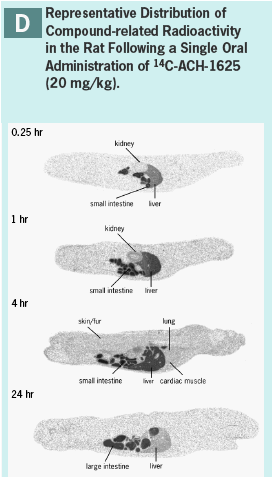
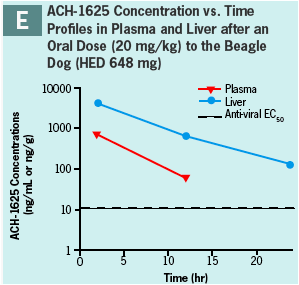
ACH-1625 or 14C-ACH-1625 shows selective distribution to the liver in rats after i.v. dosing (Panels A and B) while drug concentrations in most other tissues show values approximately equivalent to plasma.
This hepatoselective distribution was also observed after a single oral dose to rats (Panels C and D) or dogs (Panel E) at human equivalent doses of 480 and 648 mg, respectively.
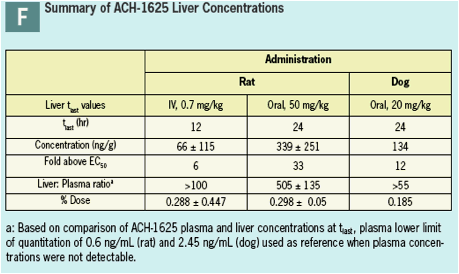
Liver concentrations at 12 or 24 hr after a single oral dose were respectively 33- and 12-fold higher than the anti-viral EC50 (Panel F) and were up to 500-fold higher than respective plasma concentration.
At liver t-last ACH-1625 concentrations were less than 1% of the dose, suggesting elimination or redistribution by 24 hr post dose.
ACH-1625 SHOWS TEMPERATURE DEPENDENT UPTAKE INTO ISOLATED HEPATOCYTES
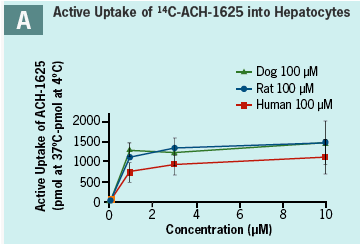
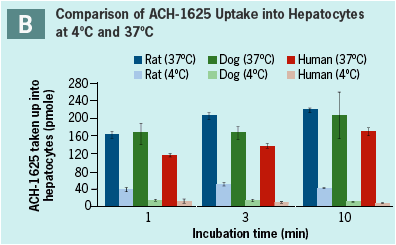
ACH-1625 shows active uptake into hepatocytes from all species (Panel A). Uptake is rapid and reaches maximal velocity by 1 min incubation.
Rate and extent of uptake into human hepatocytes is approximately similar to that in rat and dog (Panel A).
Temperature dependency is shown above (Panel B) by the higher uptake at 37°C compared to 4°C demonstrating an active uptake process.
ACH-1625 Shows Interaction with Hepatic Uptake Transporters
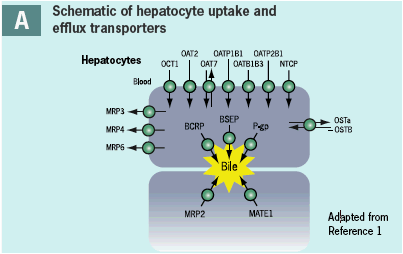
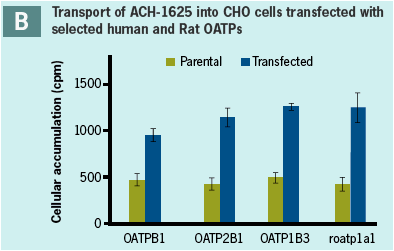
A schematic of the network of hepatic drug transporters is depicted in Panel A. Studies using CHO cells transfected
with each of three hepatic membrane transporters showed 2 - to 3-fold higher uptake of 14C-ACH-1625 into CHO cells
containing expressed transporters than in parental cell line.
Similar uptake of ACH-1625 was observed in cells transfected with both human and rat transporters.
|
| |
|
 |
 |
|
|