 |
 |
 |
| |
ANTIVIRAL ACTIVITY, COMBINATION AND RESISTANCE OF ACH-1625, A POTENT HCV NS3 PROTEASE INHIBITOR
|
| |
| |
Reported by Jules Levin
EASL 2010
Mingjun Huang, Steven Podos, Yongsen Zhao, Joanne Fabrycki, Dharaben Patel, Guangwei Yang, Chris Marlor, Kathe Stauber, Xiangzhu Wang, Atul Agarwal, Milind Deshpande and Avinash Phadke
Achillion Pharmaceuticals, New Haven, CT 06511, USA.


Background
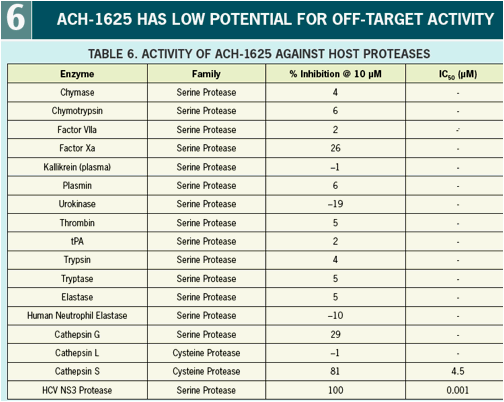
HCV NS 3 protease is a clinically validated target. Analysis of structural information of the target and its ligands led to design and synthesis of linear, competitive reversible inhibitors including the clinical development candidate ACH-1625. ACH-1625 displays good human metabolic stability and high human hepatoselectivity in vitro. ACH-1625 demonstrates favorable pharmacokinetic and good safety profiles in animals (Abstract 172). ACH-1625 is very well tolerated after administration of single and multiple doses up to 2000 mg in healthy volunteers and displays a mean maximal viral load of 4.25 log10 after administration of 500 mg BID in HCV genotype-1 infected patients (LB 2012).

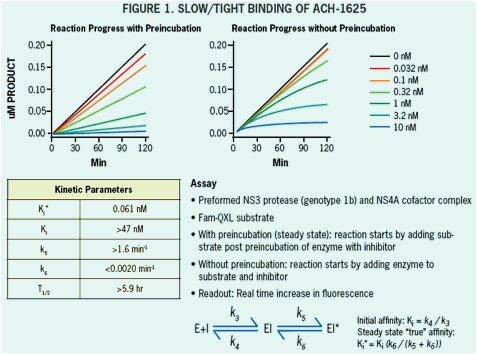

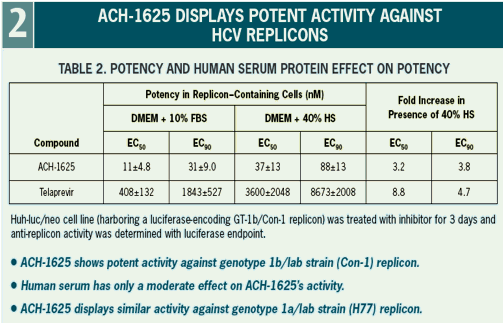
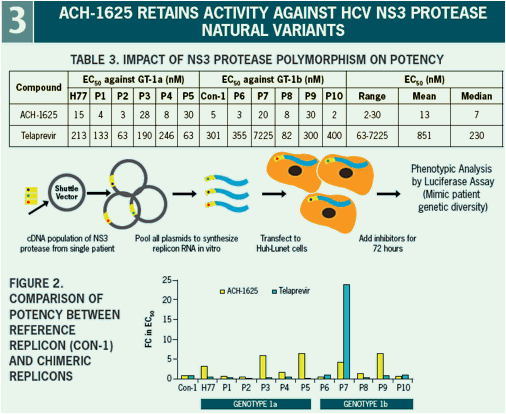

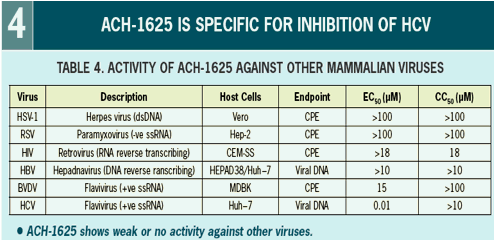
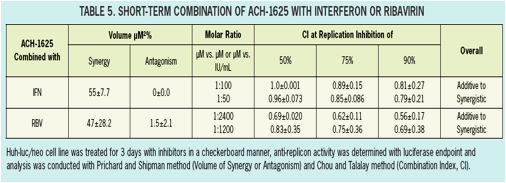
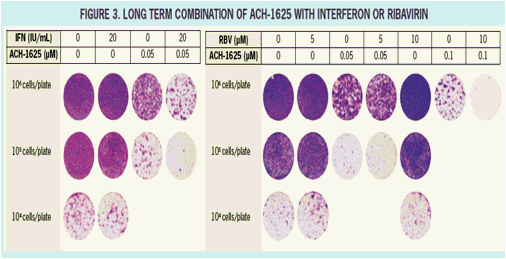
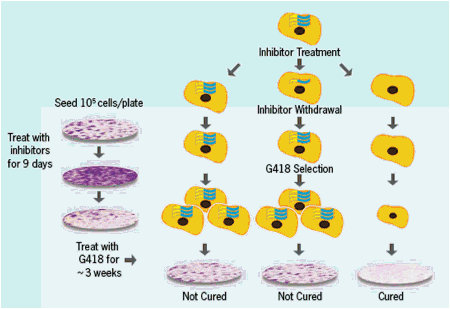
ACH-1625 Enhances Anti-HCV Activity of Interferon-α and Ribavirin


|
| |
|
 |
 |
|
|