 |
 |
 |
| |
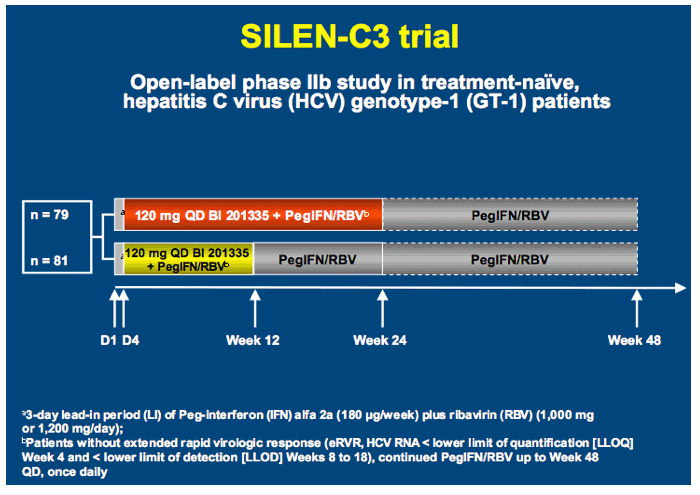
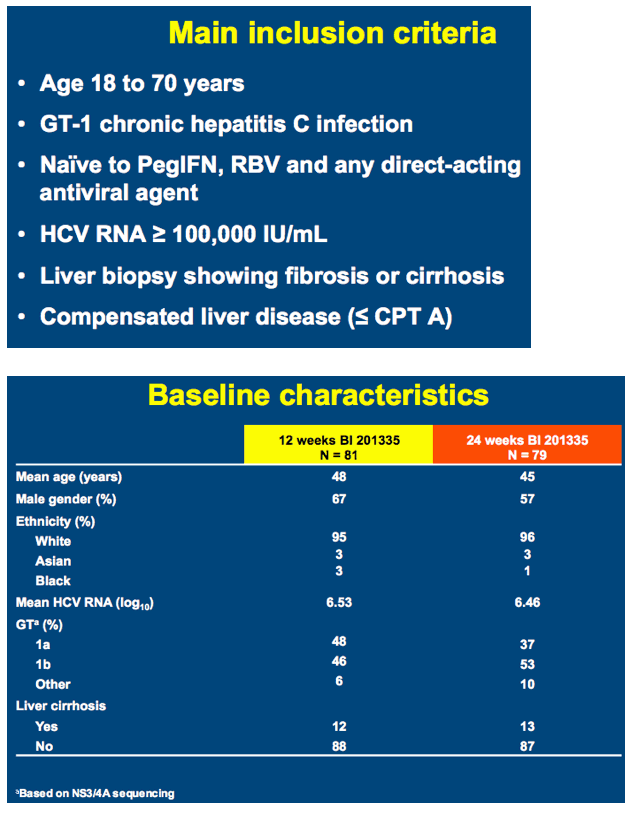
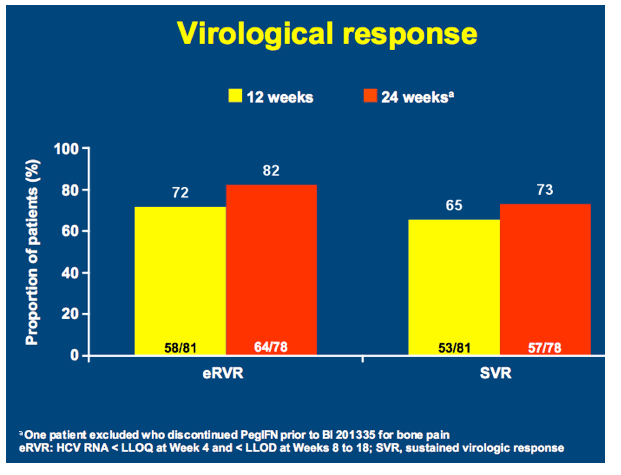
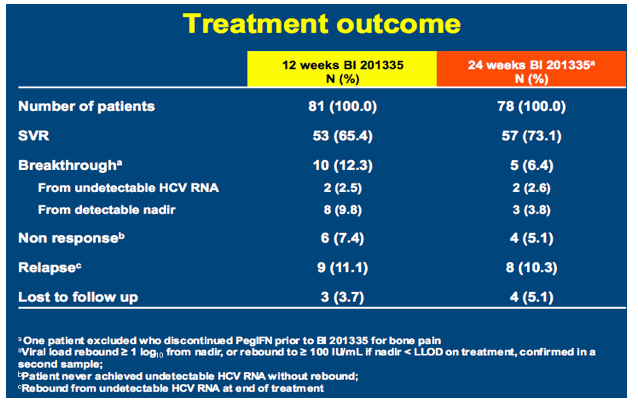
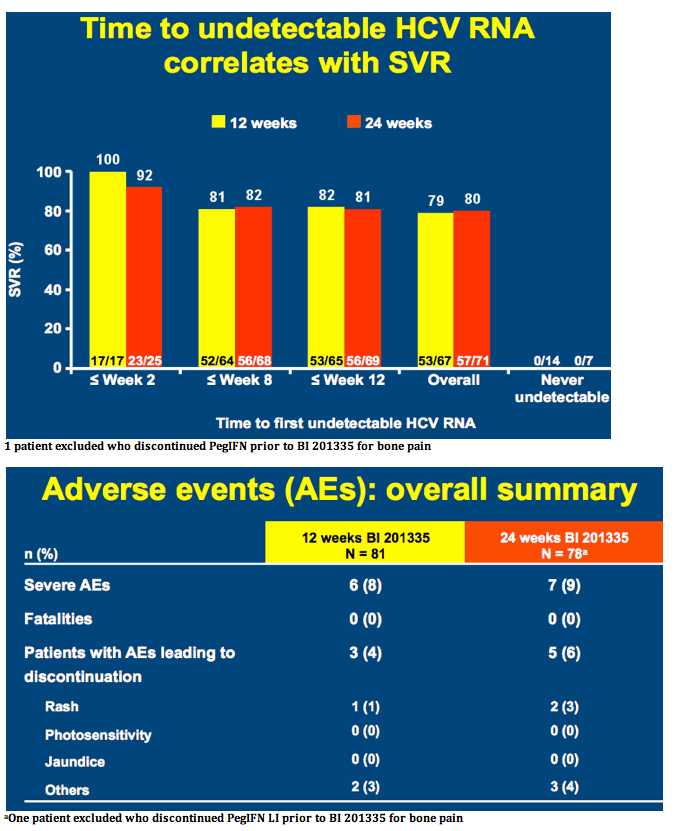
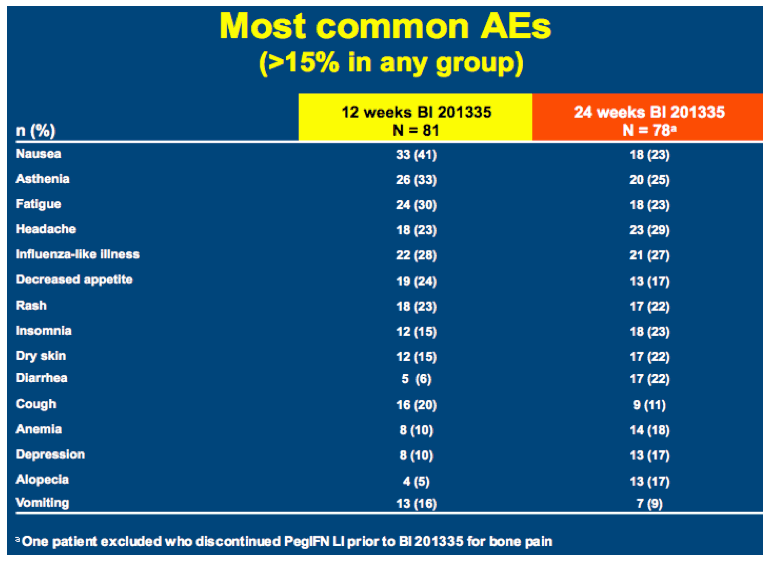
SILEN-C3: treatment for 12 or 24 weeks with BI 201335 combined with peginterferon alfa-2a and ribavirin in treatment-naïve patients with chronic genotype-1 HCV infection
|
| |
| |
Reported by Jules Levin AASLD Nov 5-9 2011 SF
Douglas Dieterich,1 Tarik Asselah,2 Dominique Guyader,3 Thomas Berg,4 Emanoil Ceausu,5 Liliana Preotescu,6 Marcus Schuchmann,7 Stefan Mauss,8 Vlad Ratziu,9 Peter Ferenci,10 Dominique Larrey,11 Andreas Maieron,12 Michael Biermer,4
Jerry O. Stern,13 Joe Scherer,13 Luyan Dai,13 Wulf Bocher14
1Mount Sinai School of Medicine, New York, USA; 2Hopital Beaujon, Clichy Cedex, France; 3University of Rennes, Rennes Cedex, France; 4Charité, Campus Virchow-Klinikum, Berlin, Germany; 5 'Dr. Victor Babes' Hospital for Infectious and Tropical Diseases, Bucharest, Romania; 6'Prof. Dr. Matei Bals' Institute of Infectious Diseases, Bucharest, Romania; 7Internal Medicine 1, University Hospital Mainz; Germany; 8Center for HIV and Hepatogastroenterology, Düsseldorf, Germany; 9Service d'Hepato-Gastroentérologie, Hôpital Pitié-Salpêtrière, Paris, France; 10Medical University, Wien, Austria; 11INSERM632-CIC, Hôpital Saint Eloi, Montpellier, France; 12Department of Gastroenterology and Hepatology, Elisabethinen Hospital Linz, Linz, Austria; 13Boehringer Ingelheim Pharmaceuticals, Ridgefield, USA; 14Boehringer Ingelheim Pharma, Biberach, Germany.

|
| |
|
 |
 |
|
|