 |
 |
 |
| |
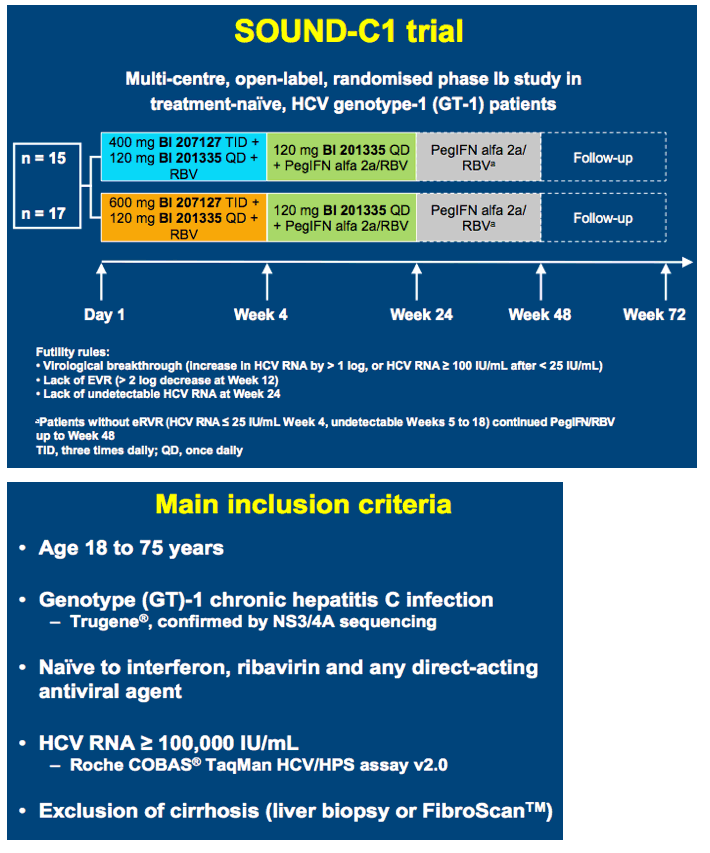
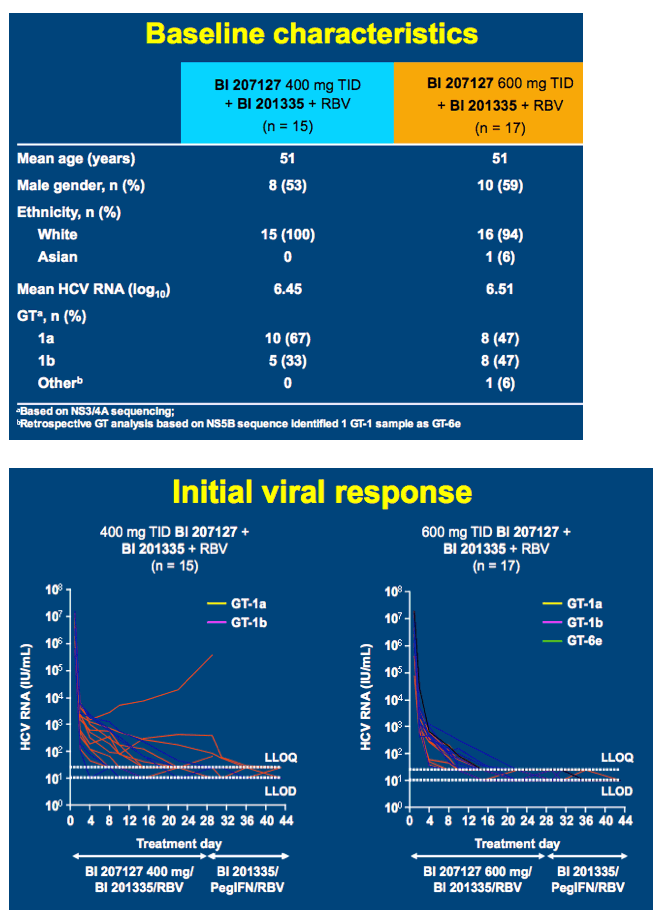
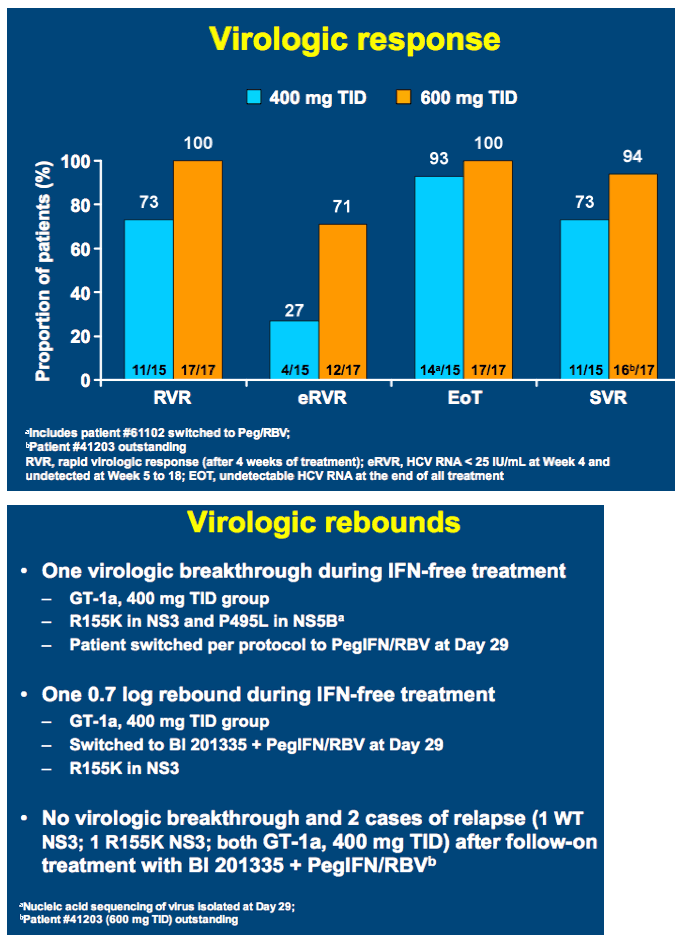
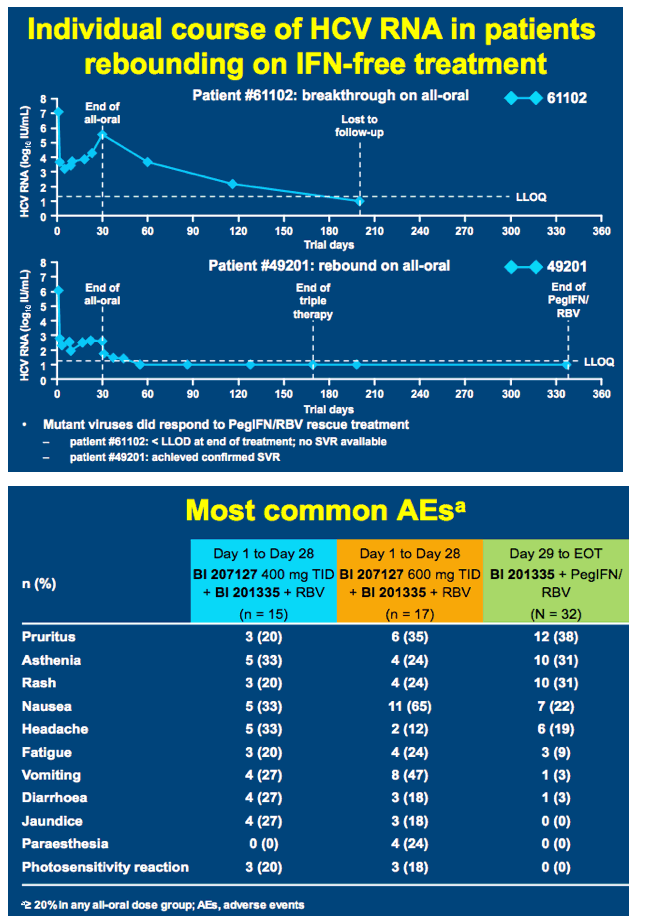
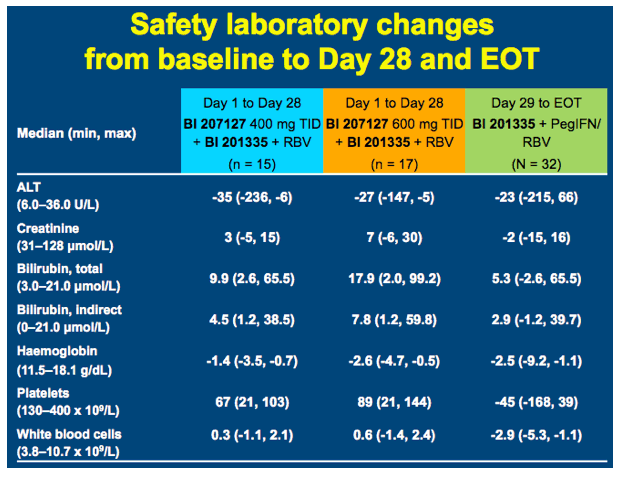
High SVR following IFN-free treatment of chronic HCV GT1 infection for 4 weeks with HCV protease inhibitor BI 201335, polymerase inhibitor BI 207127 and ribavirin, followed by BI 201335 and PegIFN/ribavirin - the SOUND-C1 study
|
| |
| |
Reported by Jules Levin
AASLD Nov 5-9 2011 SF
Stefan Zeuzem,1 Tarik Asselah,2 Peter Angus,3 Jean Pierre Zarski,4 Dominique Larrey,5 Beat Mullhaupt,6 Ed Gane,7 Marcus Schuchmann,8 Ansgar Lohse,9 Stanislas Pol,10 Joseph Moussalli,11 Jean-P. Bronowicki,12 Stuart Roberts,13 Keikawus Arasteh,14 Fabien Zoulim,15 Jerry O. Stern,16 Federico Mensa,16 Gerhard Nehmiz,17 Carla Hafner,17 Wulf Otto Bocher17
1J.W. Goethe University Hospital, Frankfurt, Germany; 2Hopital Beaujon, Paris, France; 3Austin Hospital, Heidelberg, VIC, Australia; 4Hopital Albert Michallon, Grenoble, France; 5Hopital Saint-Eloi, Montpellier, France; 6University Hospital of Zurich, Zurich, Switzerland; 7Auckland City Hospital, Auckland, New Zealand; 8University Medical Center Mainz, Mainz, Germany; 9University Hospital Hamburg-Eppendorf, Hamburg, Germany; 10Hopital Cochin, Paris, France; 11Hopital La Pitie Salpetriere, Paris, France; 12University Hospital of Nancy, Vandoeuvre-les-Nancy, France; 13Alfred Hospital, Melbourne, VIC, Australia; 14Epimed GmbH, Berlin, Germany; 15Hospices Civils de Lyon, Lyon University, Lyon, France; 16Boehringer Ingelheim, Ridgefield, CT, United States; 17Boehringer-Ingelheim Pharma GmbH&Co. KG, Biberach, Germany

|
| |
|
 |
 |
|
|