 |
 |
 |
| |
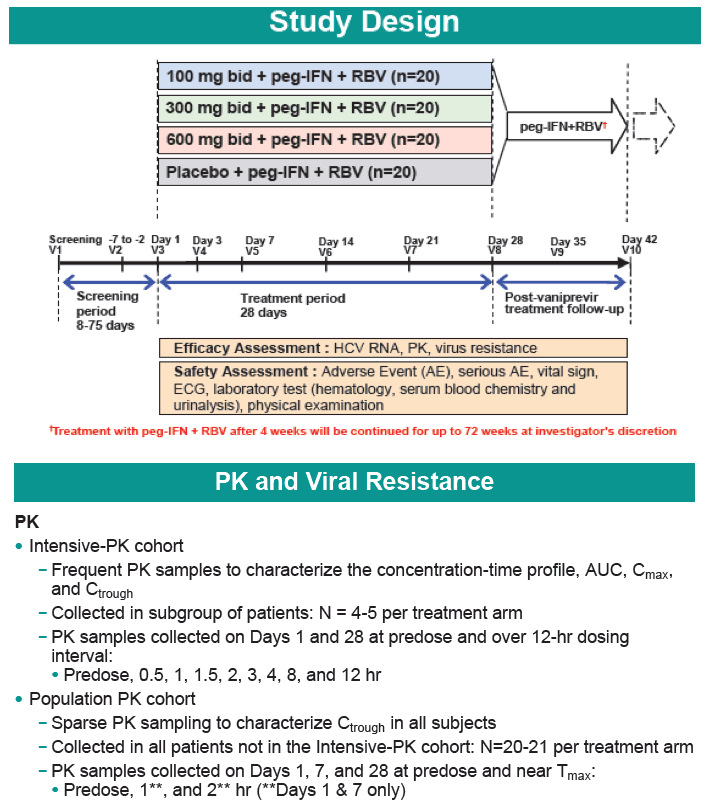
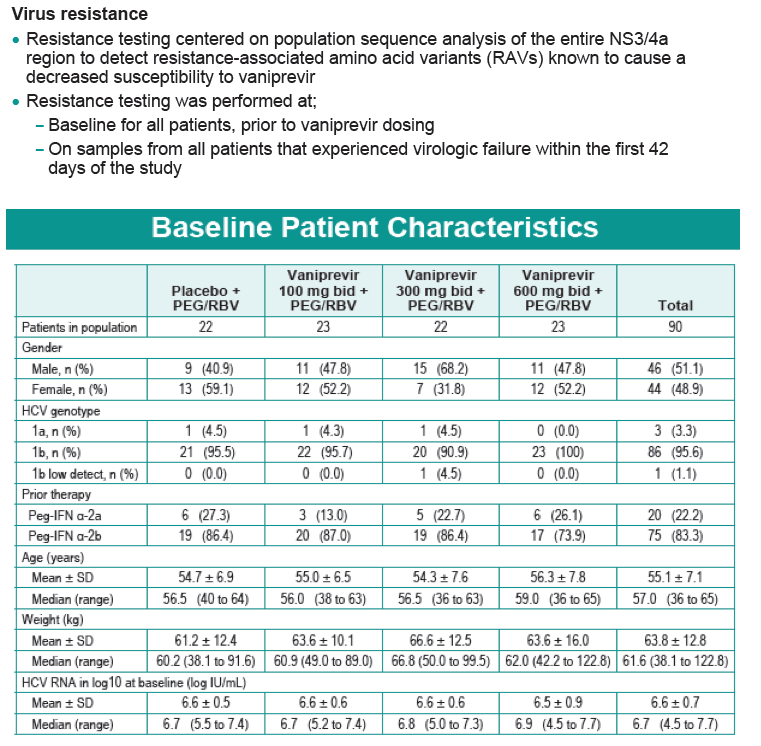
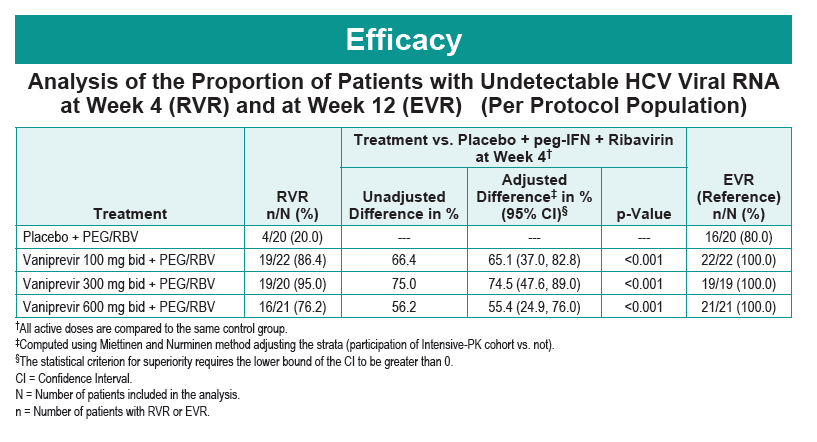
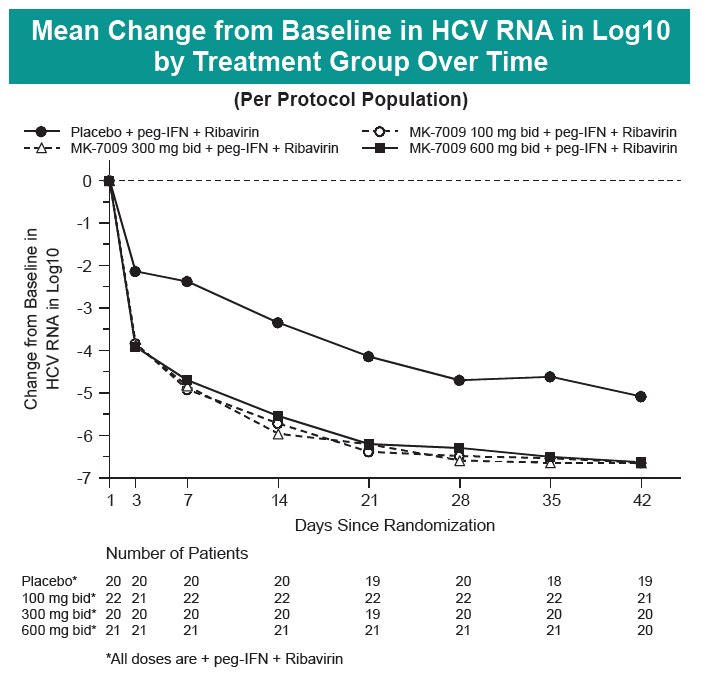
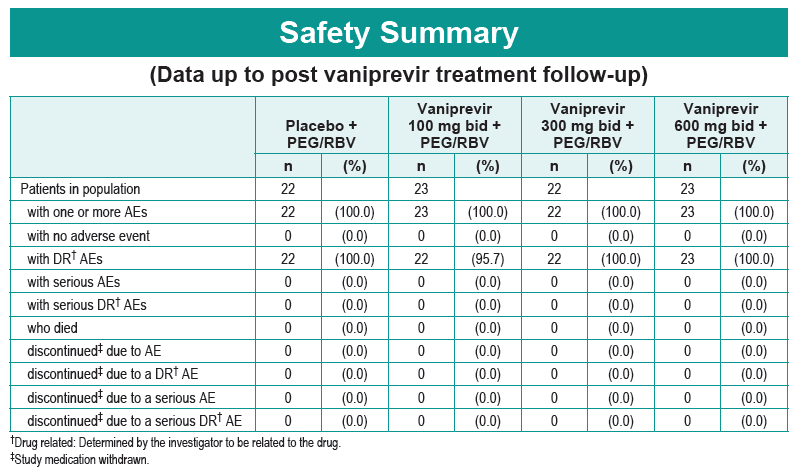
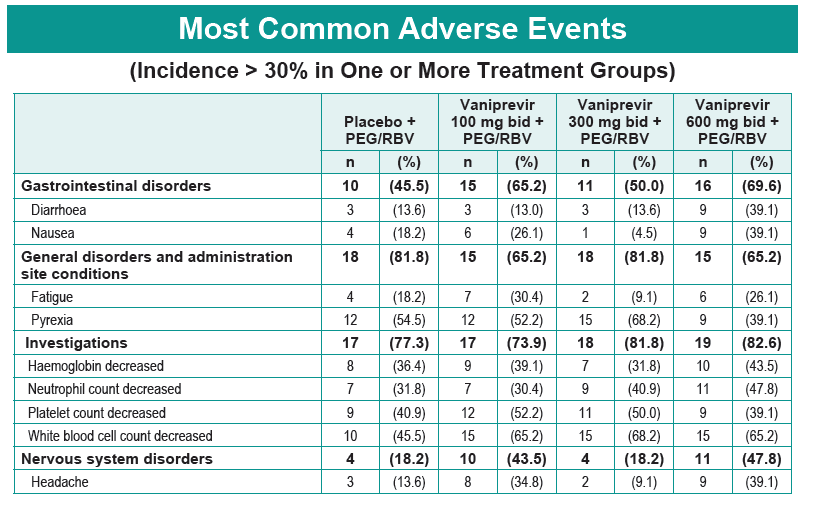
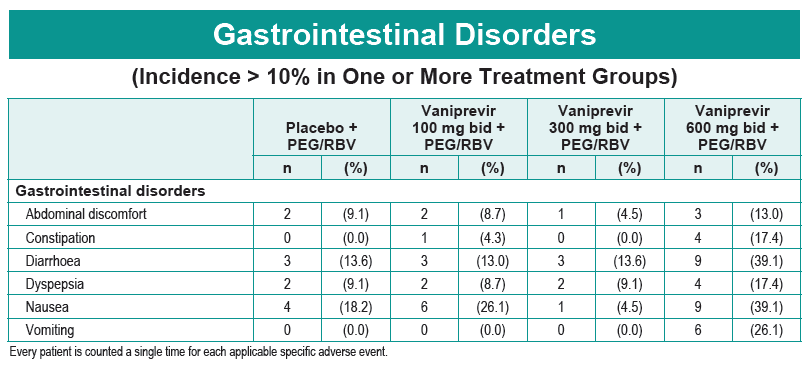
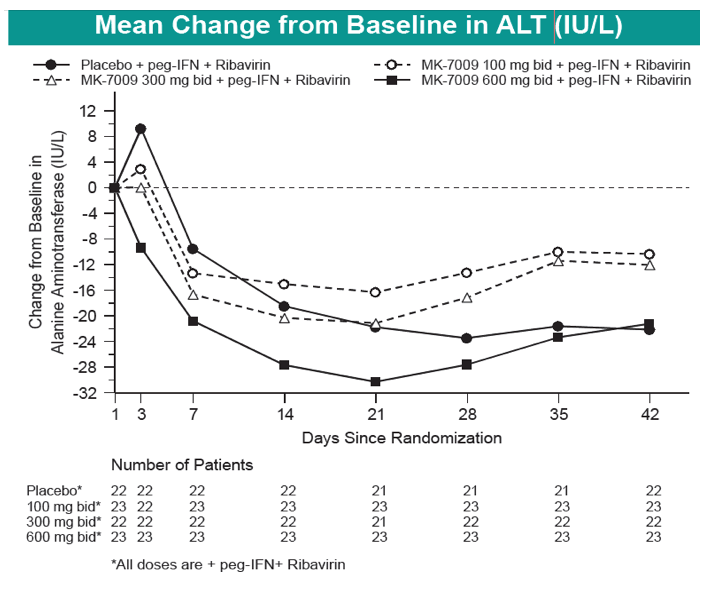
Safety and Efficacy of Vaniprevir (MK-7009) in Combination with Peg-interferon a-2a (Peg-IFN)/Ribavirin (RBV) in Genotype 1 Treatment-Experienced HCV-Infected Japanese Patients
|
| |
| |
Reported by Jules Levin
AASLD Nov 5-8 2011 SF
Norio Hayashi1, Keisuke Nakamura2, Duncan Hamish Wright3, Luzelena Caro3, Richard J Barnard3, Niloufar Mobashery3, Yoshiyuki Tanaka2
1Kansai Rosai Hospital, Hyogo, Japan, 2MSD KK, Tokyo, Japan, 3Merck Sharp & Dohme Corp, Whitehouse Station, NY, USA
AASLD: A Phase 2b Study of MK-7009 (vaniprevir) in Patients with Genotype 1 HCV Infection Who HaveFailed Previous Pegylated Interferon and Ribavirin Treatment - (11/15/11)
Safety, Tolerability and Antiviral Activity of MK-7009, a Novel ...
MONOTHERAPY 8 Days
Safety, Tolerability and Antiviral Activity of MK-7009, a Novel Inhibitor of the Hepatitis C Virus NS3/4A Protease, in Patients with Chronic HCV Genotype 1 Infection


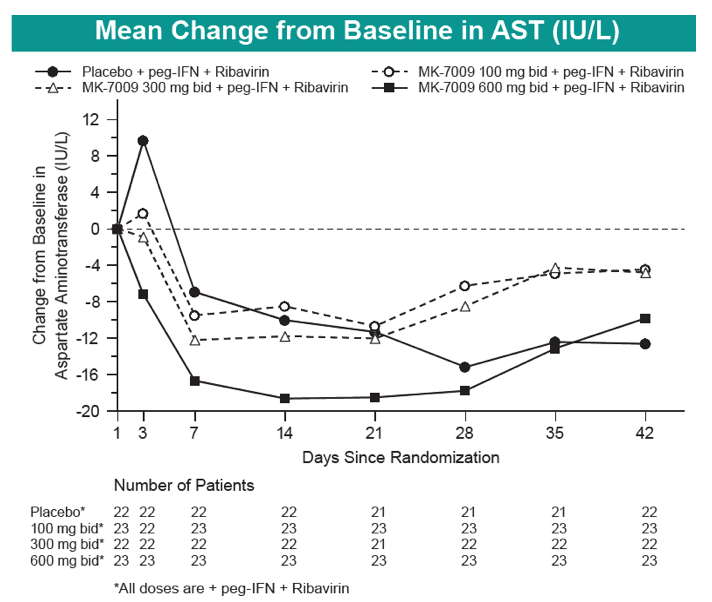
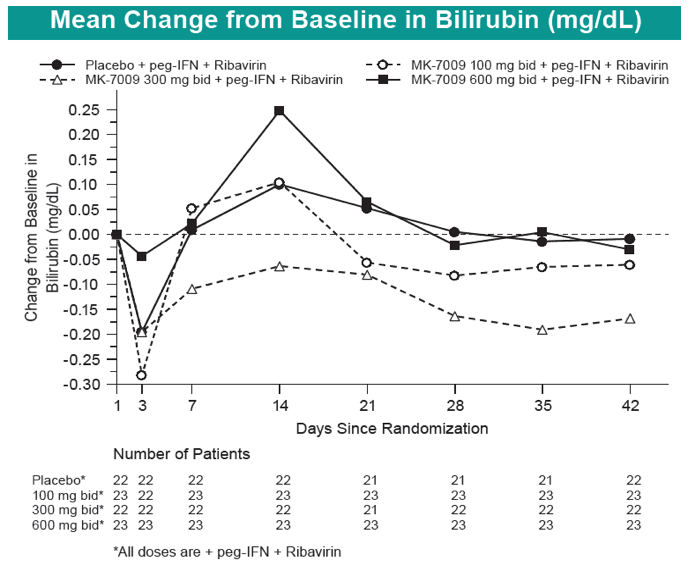
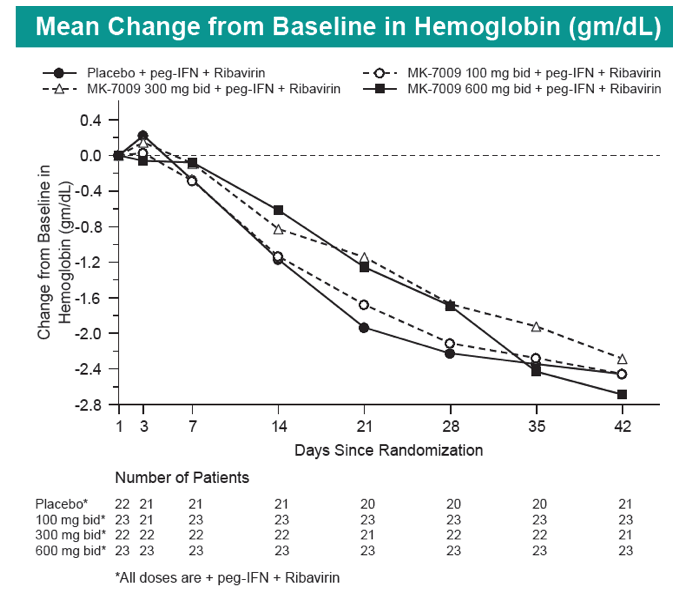
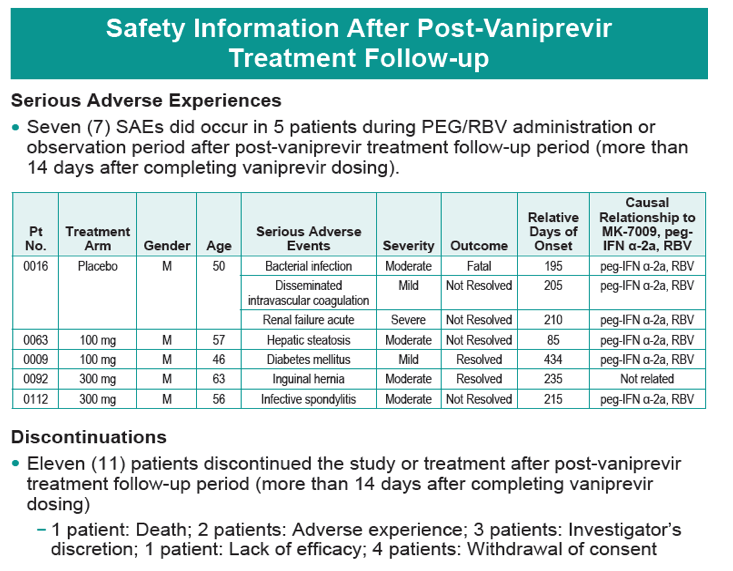
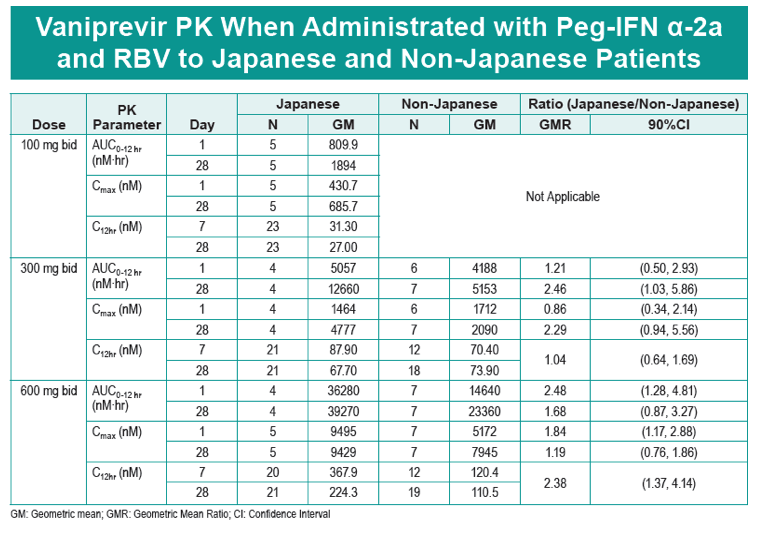

|
| |
|
 |
 |
|
|