 |
 |
 |
| |
Daclatasvir (DCV; BMS-790052) Has No Clinically Significant Effect on the Pharmacokinetics of a Combined Oral Contraceptive Containing Ethinyl Estradiol and Norgestimate in Healthy Female Subjects
|
| |
| |
Reported by Jules Levin
AASLD
Nov 5-8 2011 SF
Bifano M, Sevinsky H, Persson A, Hwang C, Kandoussi H, Jiang H, Grasela D, Bertz R
Research & Development, Bristol-Myers Squibb, Hopewell, NJ
CONCLUSIONS
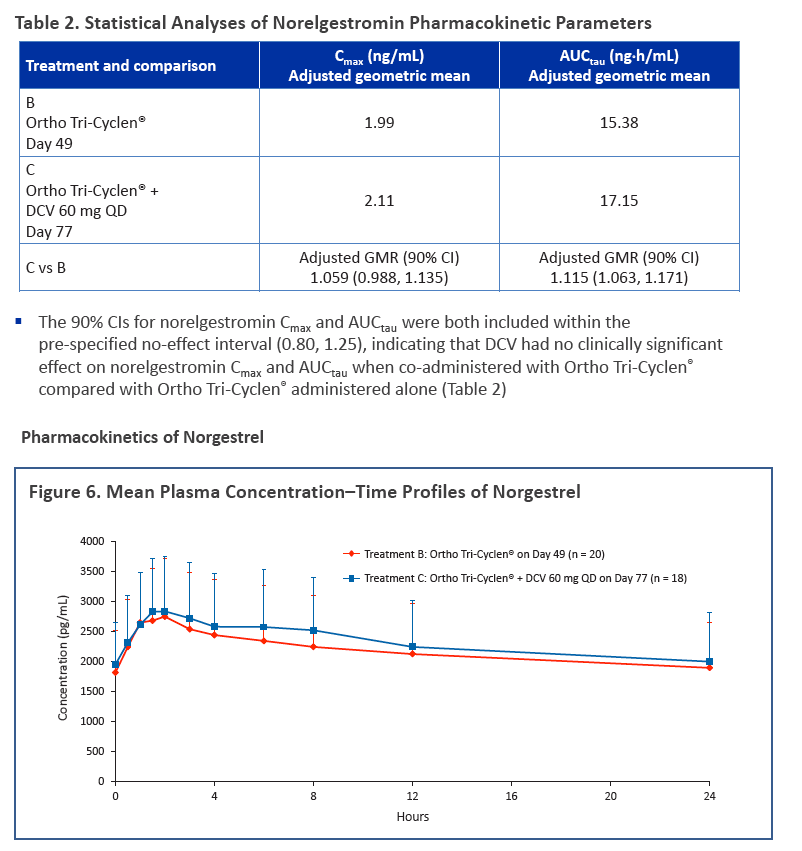
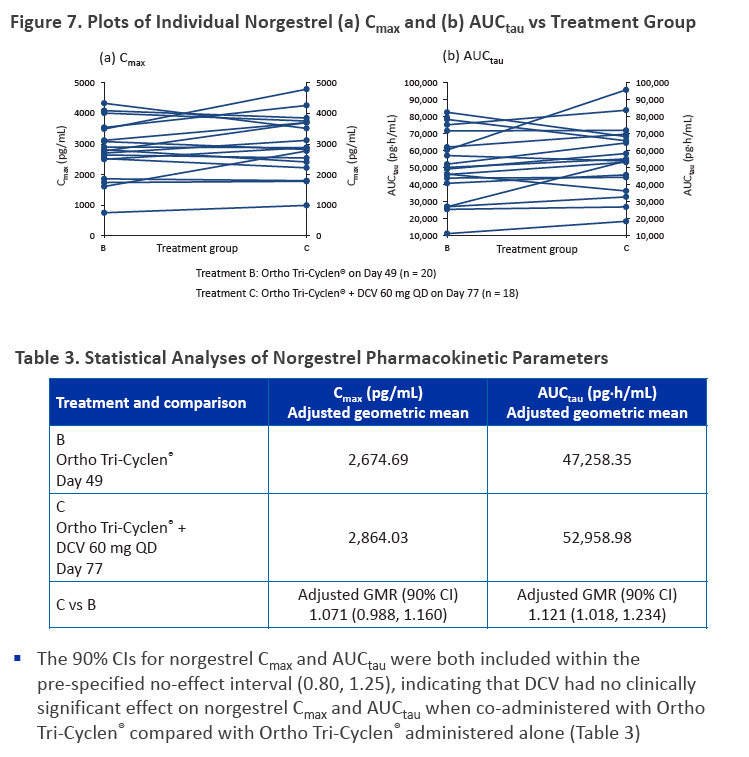
·Co-administration of DCV and Ortho Tri-Cyclen® did not result in any clinically significant pharmacokinetic drug interactions, as assessed by ethinyl estradiol, norelgestromin, and norgestrel exposures
·Contraceptive efficacy is likely to be maintained when a combined oral contraceptive containing ethinyl estradiol and norgestimate is co-administered with DCV
·No significant pharmacokinetic drug interactions between other combined oral contraceptives and DCV are anticipated ·DCV was safe and generally well-tolerated in this study



|
| |
|
 |
 |
|
|