 |
 |
 |
| |
Summary from CROI for Hepatitis Coinfection -
New drugs for treatment of hepatitis C are becoming available: what does that mean for the HIV coinfected patient?
|
| |
| |
Jurgen K. Rockstroh M.D., Professor of Medicine
University of Bonn, Germany
Correspondence:
Prof. Dr. J.K. Rockstroh
Department of Medicine I
University of Bonn
Sigmund-Freud-Str. 25
53105 Bonn
Germany
Tel.: +49-228-287 16558
FAX: +49-228-287 15034
e-mail: juergen.rockstroh@ukb.uni-bonn.de
Introduction
With the advent of the so called direct acting antivirals (DAAs) for treatment of hepatitis C mono infection treatment paradigms for HCV therapy are about to change soon. Under consideration of the to be expected registration of the first two oral HCV protease inhibitors in the coming months by the FDA, new treatment options for HCV clearly have drawn considerable attention (The agency's Antiviral Drugs Advisory Committee will review Merck's boceprevir on April 27 and Vertex's telaprevir on April 28 of 2011). Therefore, maybe not so surprisingly at this 18th Conference on Retroviruses and Opportunistic Infections in Boston a plenary delivered by Stefan Zeuzem from the University of Frankfurt was devoted to "New antiviral therapies in the management of HCV infection". In an outstanding overview professor Zeuzem introduced the audience to an extraordinary active field of current drug development including compounds from various drug classes including HCV protease inhibitors, nucleoside and non-nucleoside polymerase inhibitors, NS5A-inhibitors, cyclophyllin inhibitors as well as immune based therapies. Clearly, the addition of the soon to become available HCV protease inhibitors (telaprevir and boceprevir) to the current gold standard of pegylated interferon and ribavirin combination therapy with an on average 30% higher sustained virological response rate compared to standard combination therapy in treatment naïve genotype 1 patients will allow to cure two thirds of our naïve HCV mono infected patient population. However will this be the same in HIV/HCV coinfected patients with higher baseline HCV viral loads, overlapping toxicities between HIV and HCV agents and multiple possible drug-drug interactions? Unfortunately up to now data from trials with DAAs in HIV/HCV coinfection has not been presented as companies were reluctant to move into this potentially more challenging patient population despite the highly agreed upon unmet medical need in this particular patient group. Currently two pilot trials (one with boceprevir in about 90 patients and one with telaprevir in 60 patients) are ongoing in HIV coinfection where recruitment has been completed. Interim results of the telaprevir trial were presented at CROI this year publically for the first time.
Efficacy and safety of boceprevir in HCV monoinfection
At this year CROI the final results of the phase III trials with the HCV-NS3 protease inhibitor boceprevir (BOC) in HCV monoinfected individuals were presented (1). The so called SPRINT-2 study assessed the safety and efficacy of pegylated interferon-a-2b/ribavirin (PEG/RBV) ± BOC in treatment-naïve genotype-1 patients. As phase II studies showed a benefit with regard to percentage of successfully treated patients after a lead-in of 4 weeks of pegylated interferon and ribavirin combination therapy before addition of the HCV protease inhibitor the phase III studies with boceprevir all were designed accordingly with a 4 week lead-in of PEG/RBV. This has been unique to BOC so far; indeed the lead-in concept has not yet been demonstrated to further increase SVR rates with any other new HCV compound. Overall, SPRINT-2 consisted of 3 study arms: a 4-week lead-in treatment (LI) period with P/R for all study arms, followed by: P/R+placebo for 44 weeks (48 P/R) in the control arm; a response-guided therapy arm-BOC+P/R for 24 weeks with an additional 20 weeks of P/R only if detectable HCV RNA during week 8 to 24 (LI+24BOC/P/R± 20P/R); or a third arm of BOC+P/R for 44 weeks (LI+44BOC/P/R). Interferon was dosed 1.5 μg/kg subcutaneously weekly; the ribavirin dose was weight-based (600 to 1400 mg/day) by mouth divided twice daily, and the BOC dose was 800 mg by mouth 3 times a day. Patients with detectable HCV RNA at week 24 were discontinued for futility. The primary efficacy endpoint was sustained virologic response 24 weeks post-therapy in all patients receiving ≥1 dose of any study medication. Non-black (cohort 1) and black (cohort 2) patients were enrolled and analysed separately per protocol.
938 non-black and 159 black patients were enrolled. The majority (92%) had a high HCV viral load >400,000 IU/mL. Only few patients with more advanced fibrosis stages (9% biopsy-proven F3/4) were included. In the combined cohort analysis (all patients) sustained virologic response (SVR) was significantly higher (p <0.0001) in both BOC arms (63% in the response guided arm and 66% in the 44 weeks triple therapy arm) vs control (38%). Relapse rates in the combined cohort analysis were highest in the control arm with 22% vs only 9% in each of the boceprevir containing arms, respectively. Sustained virologic response in the two separate cohorts was also significantly higher in both BOC arms vs control. For cohort 1 patients (non-black) completing the LI and receiving ≥1 dose of BOC or placebo, respective sustained virologic response were 42%, 70%, and 71% for the 3 study arms. At the end of the LI period, ~25% of patients had <1 log decline in baseline HCV RNA. Regardless of week-4 decline, sustained virologic response was consistently higher in the BOC arms than control.
Discontinuation for adverse events occurred in 16%, 12%, and 16% in the 3 arms, respectively. Anemia was reported in 29% of controls vs 49% in BOC arms but rarely led to treatment discontinuation. In summary, BOC/P/R significantly increased sustained virologic response in both the response-guided therapy and 48-week treatment arms over standard of care by ~70% (67% vs 40%). BOC was well tolerated. Compared to 44 weeks of triple therapy, response-guided therapy produced comparable sustained virologic response highlighting that in patients who are good initial responders shorter treatment durations appear to be feasible than currently recommended for combination HCV therapy of genotype 1 patients.
The second major phase III study (RESPOND-2) evaluated efficacy and safety of boceprevir in the context of a retreatment of HCV genotype 1 patients who had previously failed a PEG/RBV HCV therapy (2). Two predefined patient populations of previous treatment failure were studied: relapsers and PEG/RBV non-responders who had shown at least a 2log drop in HCV RNA by week 12 of prior therapy but with detectable HCV-RNA throughout the course of therapy. Overall, in this double-blind placebo controlled trial, genotype-1 P/R treatment failure patients from the US, Canada, and Europe were randomized 1:2:2 to receive either P/R control (arm 1; n=80), 4 weeks of P/R (lead-in) then response-guided therapy with P/R + BOC (arm 2; n=162), or P/R lead-in for 4 weeks followed by 44 weeks of P/R + BOC (arm 3; n=161). All patients with detectable HCV RNA at week 12 were discontinued for futility. The primary endpoint was sustained virologic response at 24 weeks post-treatment (Roche TaqMan LLD <15 IU/mL). Patient distribution was predominatly male (67%), 12% black, and 12% cirrhotic.
Sustained virologic response was significantly higher (p <0.0001) in both BOC arms (59% in the BOC response guided arm and 66% in the 44 weeks BOC/PEG/RBV arm) vs control (21%). For all arms, previous relapsers had higher sustained virologic response than previous non-responders. The highest sustained virologic response (79%) was observed in patients with a ≥1 log decline at week 4 following the 4 week P/R lead-in, who were then treated with 800 mg BOC 3 times per day for 44 weeks. Notably, sustained virologic response in arms 2 and 3 was significantly higher (33% and 34%, respectively) than arm 1 (0%) for patients with a <1 log decline at week 4. Discontinuation due to adverse events occurred in 3%, 8%, and 12% of patients in arms 1, 2 and 3, respectively. The most common reason for discontinuation was lack of response at week 12.
In summary this study shows that in particular in previous relapsers BOC added to P/R leads to high sustained virologic response rates in genotype-1 patients. In partial non-responders again triple therapy leads to significant SVR improvements. Which impact the addition of BOC to PEG/RBV could have in previous null-responders (less than one log drop in HCV RNA under combination therapy) remains unclear as these patients were excluded from this study.
What happens after stopping boceprevir therapy in the setting of treatment failure and resistance emergence?
Virological rebound or failure to suppress HCV replication under ongoing therapy with HCV protease inhibitors in combination with PEG/RBV has been associated with the emergence of HCV protease inhibitor associated resistance conferring mutations. Somewhat similar to the HIV-field cross resistance appears to be common among HCV protease inhibitors. As HCV however, is not integrated into the cell genome potentially wildtype HCV may reemerge and resistance associated mutations could disappear over time. In the SPRINT-2/RESPONSE-2 studies resistance associated amino acid variants (RAVs) were detected in 53% of non-SVR patients. At CROI results were presented on the rate of reemergence of wild type at BOC resistance-associated variant loci in genotype-1a and -1b patients from an interim analysis of long-term follow-up of patients enrolled in a 3-year post-therapy long-term outcome study (3). Samples from 599 patients (treatment-naive or IFN/RBV nonresponders) who received BOC/IFN/RBV in 2 phase 2 trials were evaluated. Roche Taqman (LLQ = 25 IU/mL) was used for HCV RNA quantitation. RAV were detected by population sequencing of NS3 protease region (codons 1-181) by Virco BVBA (Belgium) or sponsor. Enzyme- and cell-based HCV protease assays evaluated the fold-shift of each variant. Of 307 non-sustained virologic response patients who received BOC 183 had sufficient follow-up for analysis. All 183 subjects had G1a or G1b viruses that had developed RAV during treatment and which were still detectable at end of therapy. In total, 17 BOC RAV were identified in G1a and G1b viruses: R155K (65%), T54S (56%), V36M (53%), T54A (12%), others <11% each. Median time to re-emergence of wild type was shortest for G1b viruses with T54A (0.24 years); reemergence of wild type in viruses with T54S was longer in both G1a and G1b (median time 1.46 and 1.21 years, respectively). In G1a, median time to reemergence of wild type was shortest for V36M, 0.78 years, compared with 1.28 years for the R155K variant. In conclusion wild type reemerged at different rates depending on the specific RAV suggesting differences in relative fitness of the specific RAV. Also resistance emergence may vary between different genotype 1 subtypes. The question which came up in discussion was whether use of more sensitive assays would lead to higher rates of resistance persistence with potentially some clinical implications upon retreatment with this or other HCV protease inhibitors. So far no data exists on patients who developed resistance conferring mutations under oral HCV protease inhibitors and were then exposed again to the same or different HCV protease inhibitors once wild type virus had reemerged. Clearly in clinical practice resistance emergence should be avoided as much as possible. Therefore stopping rules for oral HCV protease inhibitors will need to be established for patients on triple therapy who fail to suppress and continue to show signs of HCV viral replication. A possible stopping rule could be termination of the corresponding HCV protease inhibitor if HCV viral load is still above 1000 IU/mL after 4 weeks of triple therapy or in treatment-naïve patients if HCV viral load is still detectable at week 24 (for boceprevir). Final results of the ongoing first trials in coinfection will be needed to further define these corresponding stopping rules.
Efficacy and safety of telaprevir in HCV monoinfection
At CROI in a poster presentation the pooled analysis of 2 phase III trials with telaprevir (TVR) were presented (ADVANCE and ILLUMINATE) (4). ADVANCE was a randomized, placebo-controlled, phase 3 clinical trial that evaluated efficacy and safety of TVR in combination with PEG/RBV in chronic HCV genotype 1-infected treatment-naïve patients.
Patients were randomized either to a control arm of placebo + pegIFNa-2a subcutaneously, every week + weight-based RBV and then an additional 36 weeks of PEG/RBV or triple therapy with TVR for either 8 or 12 weeks. Patients in the TVR arms who achieved an eRVR (extended RVR, undetectable HCV RNA at weeks 4 and 12) received a total of 24 weeks of therapy, while those who did not achieve an eRVR received a total of 48 weeks of therapy. HCV RNA was evaluated using the TaqMan assay (limit of quantification 25 IU/ml). ILLUMINATE was an open-label, randomized, non-inferiority Phase 3 clinical trial that evaluated efficacy and safety of 24 weeks of TVR-based treatment versus 48 weeks in chronic HCV genotype 1-infected treatment-naïve patients who achieved extended rapid virologic response (eRVR, undetectable HCV RNA at weeks 4 and 12). In the pooled analysis 903 patients treated with telaprevir 12 weeks/PEG/RBV were compared to 361 treated with PEG/RBV (ADVANCE only). The participants of the pooled telaprevir for 12 weeks/PEG/RBV study group were mainly male (60%) and Caucasian (83%) with a mean age of 48 years; 80% of patients had baseline HCV RNA ≥800,000 IU/mL, 25% had advanced fibrosis (F3/F4). At weeks 1, 2, and 4, 6% (58 of 903), 28% (251 of 903), and 70% (635 of 903) of T12PR patients had undetectable HCV RNA vs 2% (7 of 361), 5% (17 of 361), and 9% (34 of 361) in the PEG/RBV group. HCV RNA undetectability at weeks 1, 2, and 4 were associated with extended rapid virologic response and sustained virologic response. 70%, 63% and 73% of telaprevir/PEG/RBV patients achieved RVR, eRVR, and SVR compared to 9%, 8%, and 44% of PEG/RBV patients, respectively. The most common adverse events during overall treatment phase were fatigue, pruritus, and nausea. Overall discontinuation rates of all study drugs due to adverse events during telaprevir treatment phase were 7% vs 4% in the PEG/RBV treated patients. Clearly the results highlight that the addition of the HCV protease inhibitor telaprevir led to an eight-fold higher HCV-RNA negativation at week 4 compared to PEG/RBV therapy. The rate of study discontinuations due to adverse events was low. Another important message from the ADVANCE trial was that 12 weeks of telaprevir/PEG/RBV was associated with a significantly higher SVR rate than the shorter duration of only 8 weeks of telaprevir exposure. [from Jules: In ADVANCE SVR rate was 75% for patients who received 12 weeks of telaprevir vs 69% for those receiving 8 weeks].
Interim results of a phase 2a double-blind Study of TVR in Combination with pegIFN-a2a and RBV in HIV/HCV Co-infected Patients
From the standpoint of an HIV/HCV coinfection care provider the most exciting presentation at this year CROI were the first results from a 12 week interim analysis around the efficacy and safety of telaprevir use in HIV/HCV coinfected individuals (5). Within this pilot study patients without HIV treatment indication (part A) were randomized to either TVR 750 mg every 8 hours + pegIFN-α2a 180 mg/week + RBV 800 mg/day for 12 weeks followed by 36 weeks of pegIFN-α2a + RBV (TVR/PEG/RBV group) or placebo + pegIFN-α2a/RBV for 48 weeks. In part B patients on antiretroviral therapy (either Efavirenz or boosted atazanavir plus tenofovir + 3TC or FTC) were again randomized 2:1 to either TVR 750 mg every 8 hours + pegIFN-α2a 180 mg/week + RBV 800 mg/day for 12 weeks followed by 36 weeks of pegIFN-α2a + RBV (TVR/PEG/RBV group) or placebo + pegIFN-α2a/RBV for 48 weeks. The TVR dose was increased to 1125 mg every 8 hours when the ART regimen included EFV. This interim analysis was performed on 59 of the 60 dosed patients (part A, 13; part B, 46; 24 on an Efavirenz containing ART regimen, 22 on an atazanavir/r containing regimen). Overall, 88% of patients were male, 69% white, mean age was 46 years, 68% had genotype 1a, 83% had a baseline HCV RNA ≥800,000 IU/mL, and 10% revealed advanced liver fibrosis based on liver biopsy. The primary objective of the trial was to determine the proportion of patients with HCV RNA undetectable after 12 weeks of telaprevir, peginterferon, and ribavirin as well as to study the safety of the triple regimen in HIV/HCV coinfected individuals. Rate of undetectable HCV-RNA at week 4 (rapid virological response) was significantly higher in the telaprevir containing arms with 71% (no ART arm), 75 (Efavirenz containing arm) and 64% (boosted atazanavir containing arm) compared to only 5% in all the patients just receiving PEG/RBV. Similarly at week 12 rate of undetectable HCV RNA again was substantially higher in all telaprevir arms with 71% (no ART), 75 (Efavirenz containing arm), and 57 % (atazanavir/r containing arm) compared to 14% in all patients on PEG/RBV. Two patients in part B experienced HCV viral breakthrough. Discontinuations due to adverse events occurred in 2 patients (3%) in the TVR/PR groups vs 0 in the placebo group. Pruritus, nausea, vomiting, fever, anorexia and dizziness were more frequent in patients who received TVR/PR than in controls. No case of severe rash was seen. No significant changes in CD4 decrease or in HIV RNA level were observed in part B patients who received either ART regimen compared to controls.
In summary in this first pilot trial on efficacy and safety of an oral HCV protease inhibitor in combination with PEG/RBV significantly higher early virological response rates could be observed in HIV/HCV coinfected patients which were almost comparable with the results obtained at similar time points in HCV monoinfected patients. Obviously SVR rates need to be awaited to be able to completely determine efficacy of telaprevir in HIV/HCV coinfected individuals. Nevertheless under consideration of the strong association of rapid as well as early undetectability of HCV RNA and overall SVR rates also in coinfection it appears very likely that cure rates for HCV under triple therapy will be substantially higher than what has been observed in treatment of chronic HCV in HIV coinfection so far. Surprisingly the adverse event profile was quite favourably with only few study terminations because of adverse events. In particular no discontinuation of study drugs because of rash was observed in this particular patient population.
What drug-drug interactions need to be considered between HIV and HCV compounds?
Under consideration of the fact that frequently used HIV drugs such as NNRTI or protease inhibitors are metabolized by the cytochrome P450 system as well as HCV protease inhibitors the potential for significant drug-drug interactions arises. Indeed for many pharmaceutical companies the potential for such interactions making many expensive drug-drug interactions studies unavoidable in the drug development process has been one of the major obstacles to overcome prior to moving research of these compounds also into the coinfected individual. At this year CROI for the first time relevant drug-drug interaction data was disclosed for the two HCV protease inhibitors furthest in development. In one of the presentations the interactions between TVR and antiretroviral (ARV) agents were evaluated to guide studies of TVR in HIV/hepatitis C virus (HCV) co-infected patients (6). Table 1 summarizes the corresponding findings and shows some significant drug-drug interactions. A 52% reduction in telaprevir cmin concentrations when coadministered with lopinavir/r makes this combination not advisable to use at present until appropriate doses have been established. Somewhat lower AUC and cmin have also been observed when combining TPV and fosamprenavir/r or darunavir/r. More worrisome there is also a significant decline in HIV drug concentrations for darunavir or fosamprenavir potentially leading to subtherapeutic levels of the respective antiretroviral drugs. Therefore at present it appears safest to use either boosted atazanavir or a higher dose of TVR (1125 mg every 8 hours) together with Efavirenz which seems to partly offset the interaction with EFV. Moreover, these ART combinations have at least also been explored in the ongoing pilot trial presented above and been demonstrated to work well and to be safe. Data on the combined administration of TPV and raltegravir (which is not metabolized by the cytochrome P4540 system) is eagerly awaited. In a separate poster presentation low dose ritonavir and telaprevir interactions were studied in healthy volunteers (7). Following multiple doses of telaprevir and ritonavir, no significant boosting of telaprevir exposure by ritonavir was observed.
Table 1:
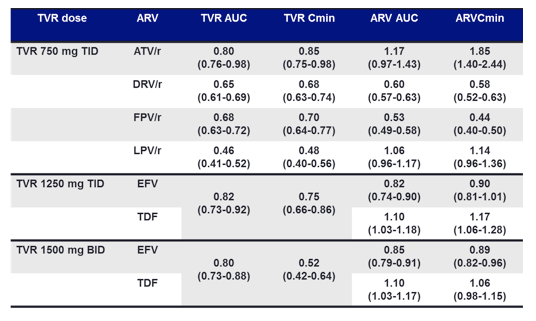
Phase 1 multiple-dose studies of boceprevir were conducted in healthy subjects to study the metabolic pathways used in BOC metabolism/elimination and drug interactions, using probe drugs and medications likely to be co-administered in patients with hepatitis C (8). There were no clinically relevant changes in BOC exposure when co-administered with peginterferon (PEG) alfa-2b, tenofovir (TFV), or drospirenone (DRSP) + ethinyl estradiol (EE). There was a slight reduction in BOC AUC(0-8h) and Cmax (19% and 8%, respectively), and a 44% decrease in BOC Cmin when co-administered with efavirenz (EFV). Ketoconazole (KCZ) increased BOC exposure (131%, AUCtf); however, ritonavir (RTV) and clarithromycin had minimal effects on steady-state BOC exposure. In conclusion, the results of pk-work with boceprevir so far has demonstrated that CYP3A4 and CYP3A5 are capable of producing a large number of oxidative metabolites from boceprevir (8,9). However, the formation of M28 and M31, known to be major circulating plasma metabolites in human is most likely mediated by aldo-keto reductases. Since the biotransformation and clearance of boceprevir involves two different enzymatic pathways, namely, oxidation via CYP3A4/3A5 and keto-reduction via aldo-keto reductase, boceprevir is less likely to be victim of drug interaction with concomitantly administered drugs that are CYP3A4 inhibitors. Nevertheless not all interactions evaluated showed results as planned or modelled suggesting that formal drug-drug interaction studies between boceprevir and commonly used HIV drugs are needed to safely use these drugs in combination in clinical practice. Although little interactions are expected according to the presenters between boceprevir and boosted PIs it would be reassuring to see that data. Efavirenz obviously induces a substantial decrease in boceprevir levels and so as long as appropriate doses are not established efavirenz should not be used in combination with boceprevir until more data becomes available. A recent overview on new directly acting antivirals for hepatitis C and their potential for interaction with antiretrovirals is recommended for further reading (10).
Any news on other interventions or drugs to treat hepatitis C?
Recent studies in HCV mono infection have demonstrated improved virologic response rates in chronic hepatitis C genotype 4 patients treated with nitazoxanide, peginterferon, and ribavirin (11). At this year CROI for the first time the effect of nitazoxanide monotherapy for 4 weeks on HCV viral load was studied in HCV GT 1 HIV coinfected individuals (12). A 30-day course of oral NTZ 500 mg twice daily was well tolerated in this group of HIV patients co-infected with HCV genotype 1. Nevertheless no changes in HCV viral load were observed during treatment in any of the patients evaluated. These data suggest that despite the promising results reported for HCV mono-infected patients with genotype 4, NTZ exhibited poor activity as a monotherapy in HIV/HCV co-infected patients with genotype 1.
What were the highlights in acute hepatitis C research?
Since 2000 outbreaks of acute hepatitis C virus (HCV) among HIV-positive men who have sex with men (MSM) have been reported from Europe, the United States, Canada and Australia. Given the burden of liver disease, in particular HCV, on the morbidity and mortality in HIV patients in the era of combination antiretroviral therapy, the rapid and significant rise in the incidence of HCV in the HIV-infected MSM population in high-income countries is alarming (13). At this year's CROI data on the molecular epidemiology of incident HCV Infection in HIV-infected MSM in the US versus infections in Europe and Australia was presented (14). The limited overlap in genetic relatedness of viral isolates within and between men in US and European cities which was found in this study suggests that, while some mixing has occurred, this epidemic of new HCV infection is occurring essentially de novo in cities with large populations of MSM on three continents. These data further suggest that HCV acquisition in these men is related more to high-risk sexual and other activities or increased susceptibility than to the circulation of a more transmissible HCV clone(s). Another interesting finding from acute hepatitis C research came from the Amsterdam HIV cohort (15). Despite high clearance rates following treatment of acute HCV and a possible decrease in incidence of primary HCV infection among HIV+ MSM in Amsterdam, an alarmingly high incidence of HCV reinfection was found in this group. The rate was about 10 times the HCV incidence described in HIV+ men without a history of HCV infection and should be further studied. This high reinfection rate indicates that prevention measures should be discussed and regular HCV testing should be continued after successful treatment. Finally new data was presented on the treatment of acute HCV and predictive factors on treatment success (16). 238 HIV-infected patients from 4 European countries who had been diagnosed with acute HCV infection and were treated early with pegylated interferon (pegIFN) and ribavirin (RBV) (n = 207) or pegIFN alone (n = 31) were followed prospectively and evaluated for sustained virologic response (i.e. negative HCV RNA 24 weeks after the end of treatment). All patients were male, of median age 39 years. Main routes of transmission were MSM (94%) and injecting drug users (IDU) (3%). In 74% of patients clinical signs of acute HCV infection were missing. 66% of patients were infected with HCV GT 1, 6% with GT 2, 12% with GT 3, and 15% with GT 4. Median baseline HCV RNA was 770,259 IU/mL and median CD4 T cell count 474 cells/mL; 66% of all patients received HAART. By univariate analysis, there were no statistical differences at baseline for HCV or HIV characteristics between patients with GT 1/4 infection (group 1) and patients with GT 2/3 infection (group 2). Median time from diagnosis to treatment start was 9 weeks, median treatment duration 26 weeks, median time to first negative HCV PCR 8 weeks. RBV dose was reduced in 10%. Treatment was stopped in 43 patients because of toxicities. Again, no statistical differences could be observed between the 2 groups. Interestingly, sustained virologic response rates were significantly higher in group 2 than group 1 (81% vs 65%, respectively; p = 0.046). To identify relevant factors for reaching sustained virologic response we performed multivariate analysis. The most important predictors of response were rapid virological response with an odds ratio of 4,600 (95% CI 2,336-9,059) (p �0,0001) and HCV GT 2 or 3 with an odds ratio of 2,945 (95% CI 1,034-8,385) (p = 0,043). In summary early antiviral treatment of acute HCV infection in HIV co-infected individuals results in sustained virologic response rates that are significantly higher than those obtained in treatment of chronic HCV co-infection. Treatment of acute HCV GT 2 and 3 infections is associated with even higher sustained virologic response rates suggesting different cure rates depending on HCV genotype similar to the genotype effects seen in chronic HCV therapy. The current consensus recommendations on how best to diagnose and treat acute hepatitis C have been published last month in AIDS and are recommended for further reading (17).
Summary
· The agency's Antiviral Drugs Advisory Committee will review Merck's boceprevir on April 27 and Vertex's telaprevir on April 28 possibly leading to the registration of the first orally available HCV protease inhibitors in the HCV treatment arena soon
· Studies on the efficacy and safety with the HCV protease inhibitors boceprevir or telaprevir in HCV genotype 1 monoinfected individuals have clearly demonstrated substantial higher HCV treatment cure rates under triple therapy compared to just pegylated interferon/ribavirin combination therapy alone allowing for cure of hepatitis c in about two thirds of treated patients; increased toxicities including higher anaemia rates (particularly boceprevir) and higher rash rates /telaprevir) have been reported although overall discontinuation rates due to adverse events remain low in the studies reported so far
· Interim results from a first pilot trial on efficacy and safety of telaprevir in combination with PEGG/RBV in HIV/HCV coinfected patients demonstrated clearly that the use of triple therapy was associated with significantly higher early virological response rates than obtained under PEG/RBV; tolerability of triple therapy so far has been very good with no discontinuation because of rash
· Under consideration of the fact that frequently used HIV drugs such as NNRTI or protease inhibitors are metabolized by the cytochrome P450 system as well as HCV protease inhibitors the potential for significant drug-drug interactions arises and need to be checked prior to combination of the respective agents; this also holds true for other comedications also being metabolized by the cytochrome P450 pathway
· Nitazoxanide exhibited poor activity as a monotherapy in HIV/HCV co-infected patients with genotype 1 infection.
· Early antiviral treatment of acute HCV infection in HIV co-infected individuals results in sustained virologic response rates that are significantly higher than those obtained in treatment of chronic HCV co-infection. Treatment of acute HCV GT 2 and 3 infections is associated with higher sustained virologic response rates than in GT 1 or 4 suggesting different cure rates depending on HCV genotype similar to the genotype effects seen in chronic HCV therapy.
References
1. Sulkowski M, Poordad F, McCone J, Bacon B, Bruno S, Manns M, Jacobson I, Reddy R, Albrecht J, and Bronowicki J-P. BOC Combined with P/R for Treatment-naïve Patients with HCV Genotype-1: SPRINT-2 Final Results. 18th Conference on Retroviruses and Opportunistic Infections, February 27-March 2, 2011; abstract 115
2. Gordon S, Bacon B, Lawitz E, Marcellin P, Vierling J, Zeuzem S, Poordad F, Albrecht J, and Esteban R. HCV RESPOND-2 Final Results: High Sustained Virologic Response among Genotype-1 Previous Non-responders and Relapsers to pegIFN/RBV when Re-treated with BOC + PEGINTRON/RBV18th Conference on Retroviruses and Opportunistic Infections, February 27-March 2, 2011; abstract 116
3. Barnard R, Howe J, Graham D, Ogbert R, Long J, Boparai N, Mendez P, Brass C, Albrecht J, and Ralston R. Durability of Response and Rate of Reemergence of Wild Type at BOC Resistance-associated Variant Loci in Genotype-1a and -1b Patients: Interim Analysis of Long-term Follow-up of Patients Treated with BOC+pegIFN/RBV18th Conference on Retroviruses and Opportunistic Infections, February 27-March 2, 2011; abstract 117
4. Sherman K, Everson G, Jacobson I, Di Bisceglie A, Nelson D, Bengtsson L, Adda N, Kauffman R, Wright C, and Zeuzem S. Early Clearance of HCV RNA in HCV Genotype 1 Treatment-naïve Patients Treated with TVR, pegIFN, and RBV: Pooled Analysis of the Phase 3 Trials ADVANCE and ILLUMINATE. 18th Conference on Retroviruses and Opportunistic Infections, February 27-March 2, 2011; abstract 957
5. Sulkowski M, Dieterich D, Sherman K, Rockstroh J, Adda N, Mahnke L,Garg V, Gharakhanian S, McCallister S, and Soriano V. Interim Analysis of a Phase 2a Double-blind Study of TVR in Combination with pegIFN-a2a and RBV in HIV/HCV Co-infected Patients. 18th Conference on Retroviruses and Opportunistic Infections, February 27-March 2, 2011; abstract 146LB
6. Van Heeswijk R, Vandevoorde A, Boogaerts G, Vangeneugden T, de Paepe E, Polo1 R, Van Solingen-Ristea1 R, de Backer K, Garg V, and Beumont M. Pharmacokinetic Interactions between ARV Agents and the Investigational HCV Protease Inhibitor TVR in Healthy Volunteers. 18th Conference on Retroviruses and Opportunistic Infections, February 27-March 2, 2011; abstract 119
7. Garg V, Luo X, McNair L, van Heeswijk R, and Kauffman RS. The Effect of Low-dose Ritonavir on the Pharmacokinetics of the Investigational HCV Protease Inhibitor Telaprevir in Healthy Volunteers. 18th Conference on Retroviruses and Opportunistic Infections, February 27-March 2, 2011; abstract 629
8. Kasserra C, Hughes E, Treitel M, Gupta S, and O'Mara E. Clinical Pharmacology of BOC: Metabolism, Excretion, and Drug-Drug Interactions. 18th Conference on Retroviruses and Opportunistic Infections, February 27-March 2, 2011; abstract 118
9. Ghosal A, Yuan Y, Tong W, Su AD, Gu C, Chowdhury SK, Kishnani NS and Alton KB. Characterization of human liver enzymes involved in the biotransformation of boceprevir, a HCV protease inhibitor. Drug Metab Dispos. 2011 Mar;39(3):510-21. Epub 2010 Dec 1.
10. Seden1 K, Back D and Khoo S. New directly acting antivirals for hepatitis C: potential for interaction with antiretrovirals. J Antimicrob Chemother 2010; 65: 1079-1085
11. Rossignol JF, Elfert A, El-Gohary Y, Keeffe EB. Improved virologic response in chronic hepatitis C genotype 4 treated with nitazoxanide, peginterferon, and ribavirin. Gastroenterology. 2009 Mar;136(3):856-62.
12. Laufer N, Abusamra L, Gun A, Krolewiecki A, Perez H, Salomon H, Quarleri J, and Cahn P. No Reduction of HCV Viral Load in HIV Patients Co-infected with HCV Genotype 1 during a 30-Day Course of NTZ Monotherapy. 18th Conference on Retroviruses and Opportunistic Infections, February 27-March 2, 2011; abstract 952
13. van de Laar TJ, Matthews GV, Prins M, Danta M. Acute hepatitis C in HIV-infected men who have sex with men: an emerging sexually transmitted infection. AIDS. 2010 Jul 31;24(12):1799-812.
14. Fierer D, Khudyakov Y, Hare B, Marks K, Chang K-M, Branch A, van Seggelen W, Danta M, van de Laar T, Holmberg S, and NEAT. Molecular Epidemiology of Incident HCV Infection in HIV-infected MSM in the US vs Infections in Europe and Australia. 18th Conference on Retroviruses and Opportunistic Infections, February 27-March 2, 2011; abstract 112
15. Lambers F, Prins M, Thomas X, Molenkamp R, van der Meer J, and Schinkel J. High Incidence Rate of HCV Reinfection after Treatment of Acute HCV Infection in HIV+ MSM: Amsterdam. 18th Conference on Retroviruses and Opportunistic Infections, February 27-March 2, 2011; abstract 958
16. Boesecke C, Stellbrink H-J, Mauss S, Page E, Nelson M, Bhagani S, Guiguet M, Katlama C, Vogel M, Rockstroh J, and NEAT Study Group. Does Baseline HCV Genotype Have an Impact upon Treatment Outcome of Acute HCV Infection in HIV Co-infected Individuals? 18th Conference on Retroviruses and Opportunistic Infections, February 27-March 2, 2011; abstract 113
17. European AIDS Treatment Network (NEAT) Acute Hepatitis C Infection Consensus Panel. Acute hepatitis C in HIV-infected individuals: recommendations from the European AIDS Treatment Network (NEAT) consensus conference. AIDS. 2011 Feb 20;25(4):399-409.
|
| |
|
 |
 |
|
|