 |
 |
 |
| |
CHARACTERIZATION AND IDENTIFICATION OF PPI-437, PPI-668 AND PPI-833 AS POTENT AND SELECTIVE HCV NS5A INHIBITORS WITH ACTIVITY AGAINST ALL HCV GENOTYPES
|
| |
| |
Reported by Jules Levin EASL 2011 Berlin Germany March 30-Apr 2
R.J. Colonno, N. Huang, Q. Huang, E. Peng, A. Huq, M. Lau, M. Bencsik, M. Zhong and L. Li
Presidio Pharmaceuticals, San Francisco, CA, USA
EASL: Presidio Pharmaceuticals, Inc. Announces Positive Results in a Phase 1b Clinical Trial of PPI-461, a Novel, Potent Broadly-Active Hepatitis C Virus (HCV) Inhibitor - (03/30/11)
The study objectives are to assess the safety, tolerability, pharmacokinetics, and initial antiviral effects of PPI-461 during once-daily (QD) dosing at three dosing levels (50, 100, and 200 mg/day) for three days. The 50 mg dosing group achieved a mean maximal HCV RNA reduction of 3.1 log10 IU/mL (5 of 6 patients), while the 100 mg group achieved a mean maximal HCV RNA reduction of 3.7 log10 IU/mL (6 of 6 patients).

ABSTRACT
Background: Inhibitors of the HCV NS5A protein are distinguished by their picomolar potency and broad HCV genotype coverage. Here we report the discovery and preclinical profile of three new NS5A inhibitors that are advancing toward clinical studies.
Methods: Antiviral potency, combination and resistance studies utilized standard HCV replicon systems (genotype 1a and 1b). HCV spectrum studies employed stable replicon cell assays with insertion of NS5A gene segments from other HCV genotypes (2a, 3a, 4a, 5a and 6a) into a HCV 1b backbone. Pharmacokinetic studies utilized IV and PO administration in rats, monkeys and dogs. Extensive ADME and toxicology profiling was performed in vitro and in multiple animal species.
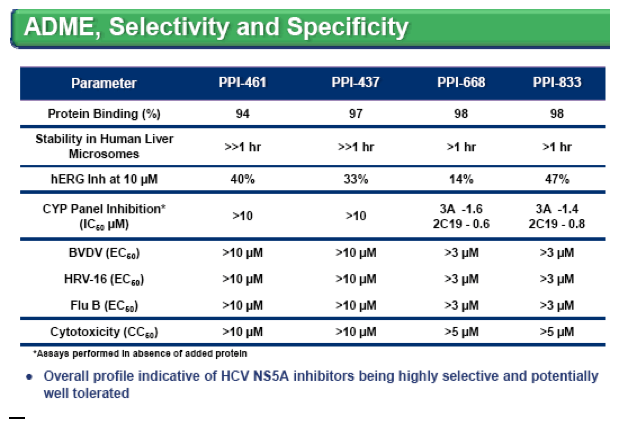
Results: PPI-437, PPI-668 and PPI-833 belong to distinct chemical series and exhibit EC50s of 0.07-0.26 and 0.01-0.02 nM in HCV 1a and 1b replicon assays, respectively. Antiviral activity (EC50) below 1 nM was exhibited against all major HCV genotypes for all three compounds, except for PPI-437 and PPI-668 which have EC s against genotype 3a of 15 and 1 3 nM respectively. Cellular cytotoxicity levels CC50) >5 μM in several cell lines and no activity was observed against several other viruses (including BVDV) at >3 μM. Additive to synergistic effects were observed for all three compounds in combination studies that
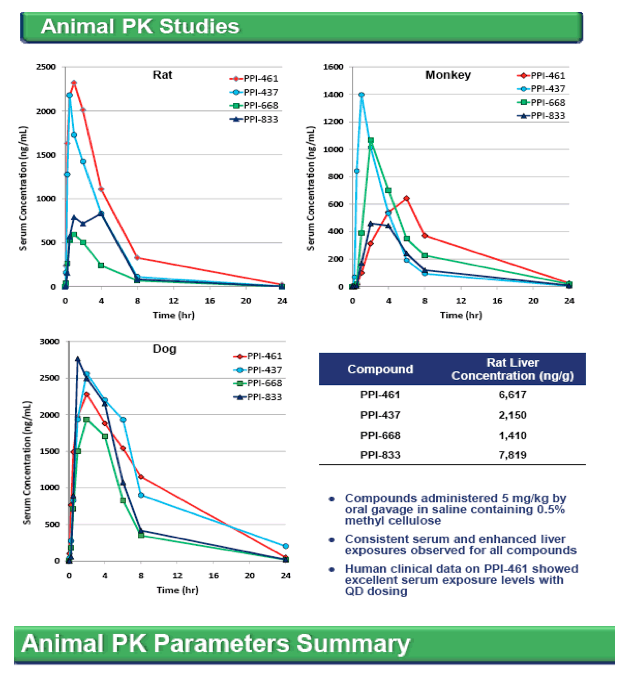
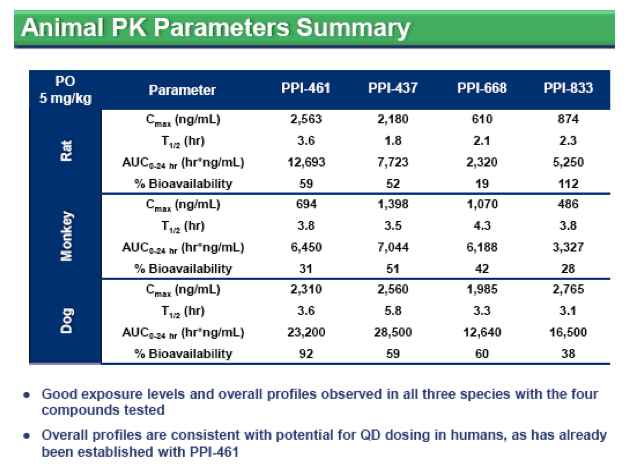
included either IFNα, HCV protease or polymerase inhibitors. Similar to other NS5A inhibitors, resistant variants readily emerge in vitro against all genotypes, although the level of resistance varied among the three compounds and usually required multiple NS5A substitutions. All three inhibitors demonstrated good stability in human liver microsomes and exhibit promising oral bioavailability in rats, monkeys and dogs, with enhanced liver concentrations and plasma half-lives suggestive of once daily dosing in humans. In animal toxicology studies, all three compounds were well tolerated at exposure levels that are orders of magnitude higher than their HCV 1a EC50 levels.
Conclusion: The HCV NS5A protein is a promising target for antiviral intervention. PPI-437, PPI-668 and PPI-833 were derived from distinct chemical series and exhibit selective and potent inhibition against all genotypes of HCV with favorable preclinical profiles supportive of advancement into clinical trials.
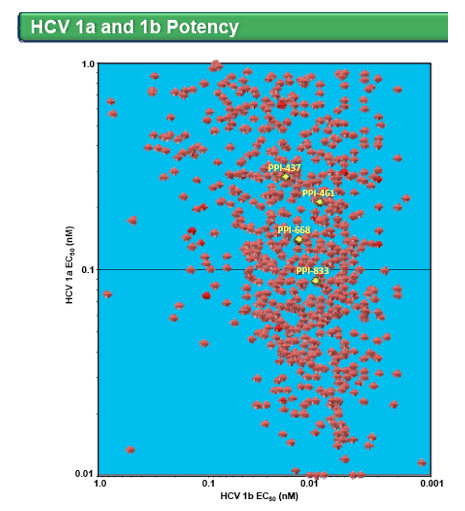
· 734 of >1,700 molecules synthesized had EC50s ≤1 nM against both HCV genotypes 1a and 1b in replicon assays
· Many proprietary compounds derived from 3 distinct chemical series met our established profiling criteria, the 4 highlighted compounds were selected for further development
· PPI-461 is currently undergoing evaluation in a Phase 1b clinical trial
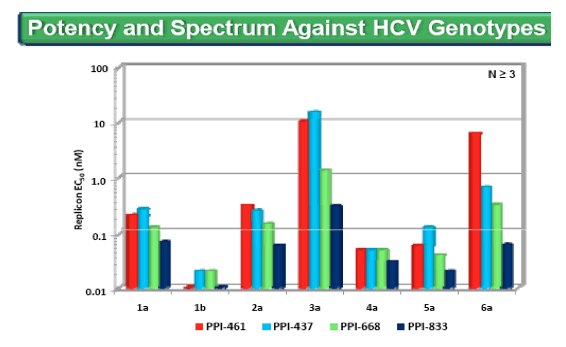
· HCV 1a replicon (1a-LucNeo replicon, luciferase) and HCV 1b (1b-LucNeo replicon, luciferase) stable cell lines were generated in Huh7-Naïve cells
· All other stable replicon cell lines containing NS5A gene segments (175-424 aa) from other HCV genotype were generated in Huh7-Naïve cells using the 1b-LucNeo replicon, except 1b NS5A (5a) was generated using Huh7-Lunet cells (EASL 2011 Poster 1199)
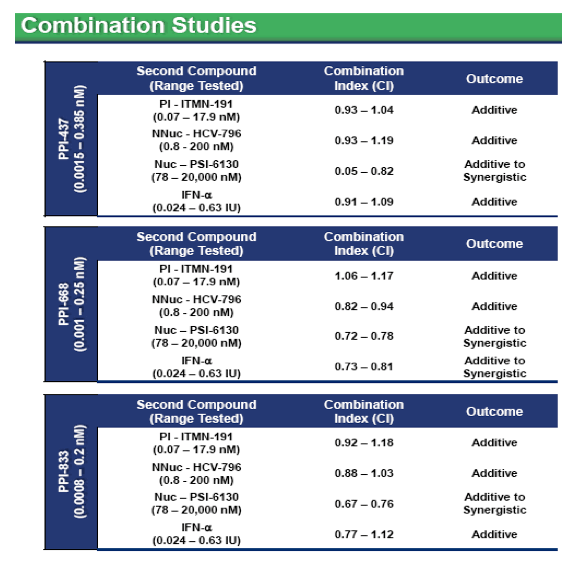
· Study conducted using matrix of indicated concentrations
· CI values calculated using CalcuSyn software, values <0.8 indicate synergy, >1.2 indicate antagonism
· NS5A inhibitors are predicted to be additive when combined with other classes of HCV antivirals, no evidence of antagonism
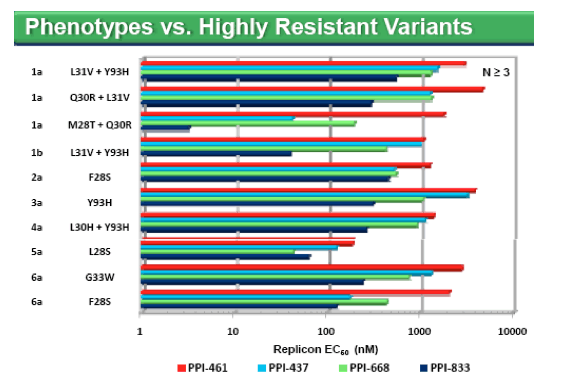
· Cross resistance observed among all NS5A inhibitors tested
· For this panel of chemically distinct NS5A inhibitors, the genotypic patterns of resistance were conserved, while the magnitude of susceptibility (phenotype) was more variable
|
| |
|
 |
 |
|
|