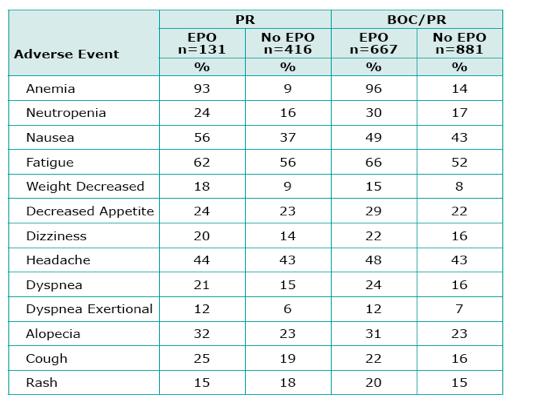
 |
 |
 |
| |
Anemia During Treatment With Peginterferon Alfa-2b/Ribavirin With or Without Boceprevir is Associated With Higher SVR Rates: Analysis of Previously Untreated and Previous Treatment-Failure Patients
|
| |
| |
Reported by Jules Levin EASL 2011 Berlin Germany March 30-Apr 2
Mark S. Sulkowski1*, Fred Poordad2, Michael P. Manns3, Jean-Pierre Bronowicki4, K. Rajender Reddy5, Stephen A. Harrison6, Nezam H. Afdhal7, Navdeep Boparai8, Kenneth Koury8, Vilma Sniukiene8,
Margaret Burroughs8, Janice K. Albrecht8, Clifford A. Brass8, Ira M. Jacobson9
1Johns Hopkins University School of Medicine, Baltimore, MD, USA; 2Cedars-Sinai Medical Center, Los Angeles, CA, USA; 3Medical School of Hannover, Hannover, Germany; 4University Henri Poincaré of Nancy, Vandoeuvre lès Nancy, France; 5University of Pennsylvania, Philadelphia, PA, USA;
6Brooke Army Medical Center, San Antonio, TX, USA; 7Beth Israel Deaconess Medical Center, Boston, MA, USA; 8Merck Sharp & Dohme Corp., Whitehouse Station, NJ, USA; 9Weill Cornell Medical College, New York, NY, USA, *msulkowski@jhmi.edu
AUTHOR CONCLUSIONS
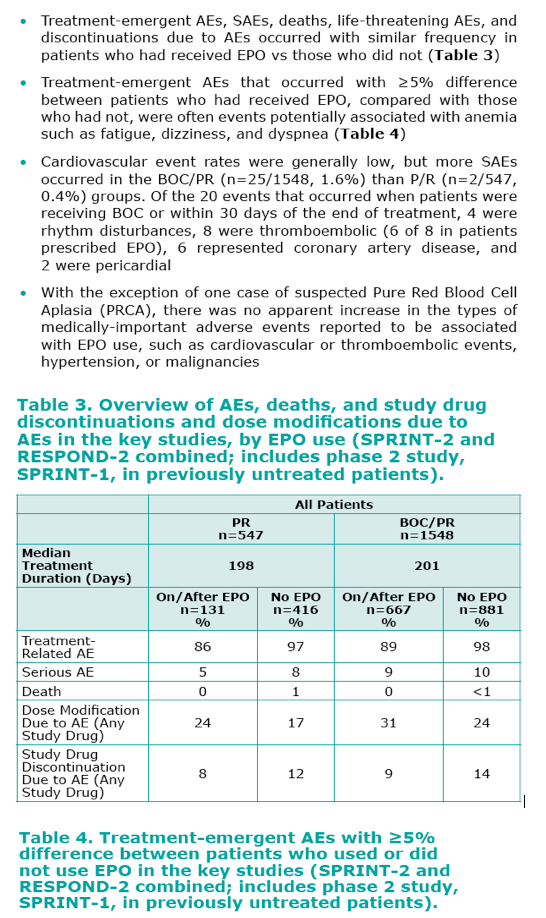
· The incidence of anemia was significantly greater in patients treated with BOC compared to those who received standard
of care P/R.
- In addition to BOC, female gender, lower baseline Hb, and older age were associated with a higher incidence of anemia.
· Anemia can be managed with R dose reduction and/or use of EPO and appropriate use of EPO in patients receiving BOC/
PR appears to have acceptable tolerability.
- Of the patients who had Hg <10 g/dL, investigators utilized EPO as part of the anemia management strategy in 80%
of patients.
- EPO was provided free of charge and supplied by Merck.
- Most anemic patients also underwent R dose reduction.
· Similar to the observation in the IDEAL study,1 patients who developed treatment-related anemia (Hb <10 g/dL) had a
higher SVR rate compared to patients who did not become anemic.
- No difference in SVR rate was observed in anemic patients managed with or without EPO.
- R dose reduction due to anemia was not associated with a reduction in the SVR rate.
· These data suggest that treatment-related anemia may be a marker of exposure to BOC and R. Further studies are
underway to assess the role of EPO in the management of anemia. In this ongoing trial (NCT# 01023035), patients who
develop anemia during therapy with BOC/PR have been randomized to management with R dose reduction alone or R
dose reduction plus EPO. Results of this trial will be presented at a future meeting.
"patients in the BOC/PR arms who had a >5 g/dL maximum decrease in Hb attained SVR rates of 75- 76%"
BACKGROUND
· More than 170 million people are chronically infected with hepatitis C virus (HCV) worldwide
-- HCV genotype 1 is most common and least responsive to current therapies
-- Leading indication for liver transplantation in Europe and United States
-- Major etiologic factor in hepatocellular carcinoma
· Boceprevir (BOC) - binds to the active site of the HCV NS3 protease
· Phase 3 studies of BOC in previously untreated and previous treatment-failure genotype 1 patients have demonstrated
-- Superior sustained virologic response (SVR) rates compared to standard of care
-- Consistent benefit across all subpopulations, including difficult-to-treat subgroups
· Decreases in hemoglobin (Hb) concentration and concomitant anemia are well recognized as side effects of interferon/ribavirin combination therapy
-- Peginterferon (P)-marrow suppressive
-- Ribavirin (R)-induced hemolytic anemia
-- Erythropoiesis-stimulating agents (ESAs) are commonly used for the management of anemia of patients undergoing current HCV treatment1-4
· ESAs were also used in some trials with first generation HCV protease inhibitors5
-- In a large Phase 3 study of genotype 1-infected patients treated with peginterferon alfa-2a or 2b, patients with anemia had higher rates of
SVR compared with non-anemic patients1
· NS3 protease inhibitors in the ketoamide class have been associated with increased anemia when used in combination with P/R6
-- When healthy male adults were treated with BOC monotherapy for up to 57 days, no effects were demonstrated on red cell production, survival, or destruction, or on markers of anemia. However, when BOC is added to P/R therapy it causes an incremental decline in Hb of ~1 gm/dL.7
OBJECTIVE
To evaluate the relationship of treatment-associated anemia, ESA use, and treatment outcomes in HCV genotype 1-infected patients in the Phase 3 studies of BOC
· Analyses are based on all randomized patients (control arm [PR48] vs. BOC arms combined [BOC/PR]) who underwent Hb measurement at baseline and at least once during the treatment phase
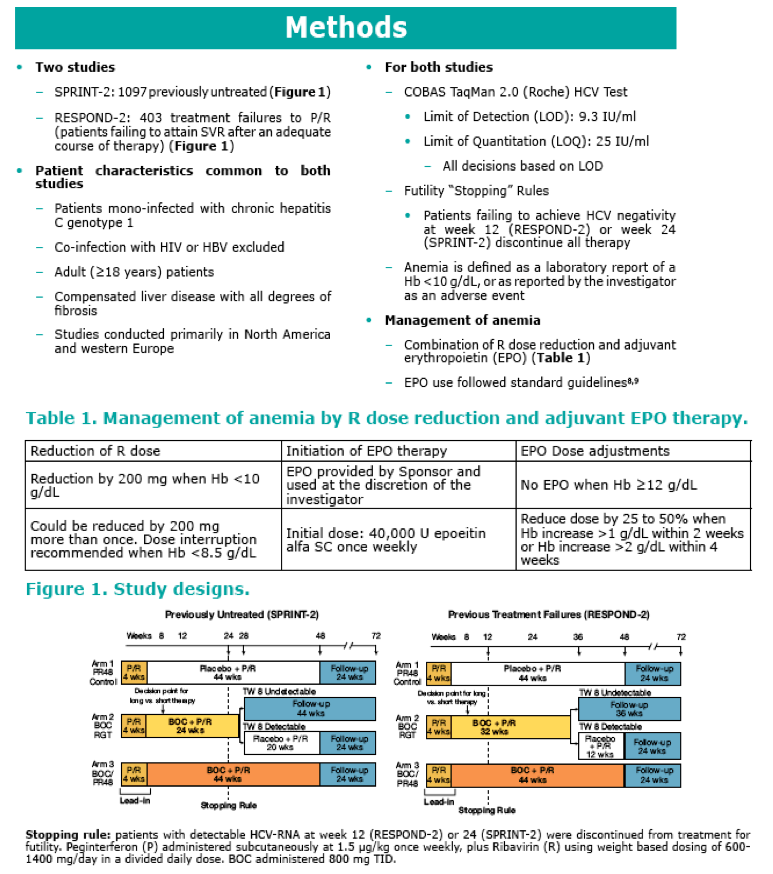
· In both studies, anemia (Hb <10 g/dL) occurred more often in the BOC (49-50%) arms than the control arms (25-30%) (Table 2)
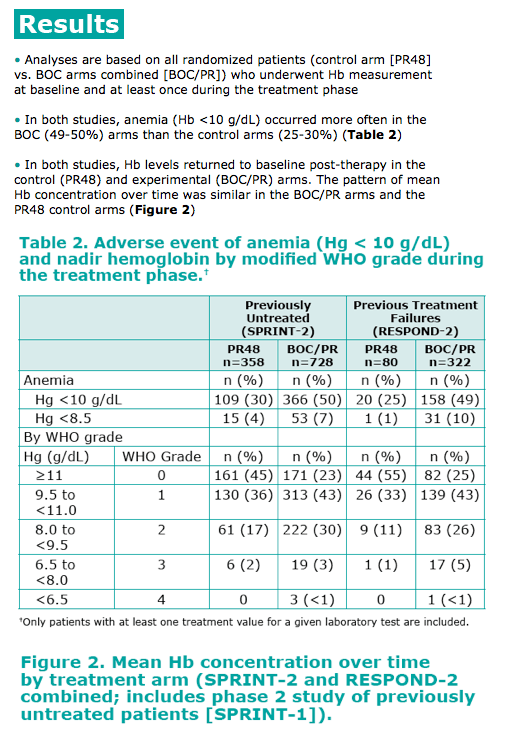
· In both studies, Hb levels returned to baseline post-therapy in the control (PR48) and experimental (BOC/PR) arms. The pattern of mean Hb concentration over time was similar in the BOC/PR arms and the PR48 control arms (Figure 2)

· In both studies, multivariate stepwise logistic regression identified
BOC treatment, baseline Hb (continuous variable), sex, age (≤40
vs >40 years), statin use (SPRINT-2 only), and race (RESPOND-2
only) as significant factors for developing anemia
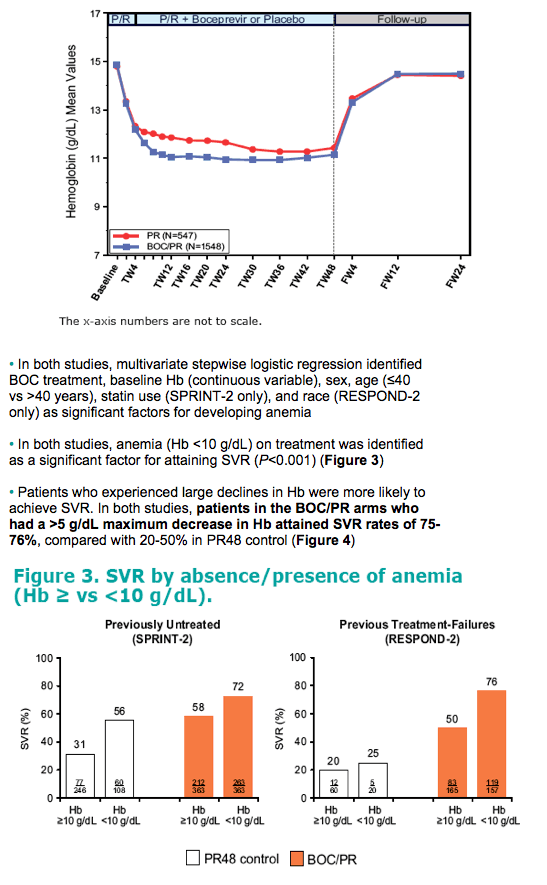
· In both studies, anemia (Hb <10 g/dL) on treatment was identified
as a significant factor for attaining SVR (P<0.001) (Figure 3)
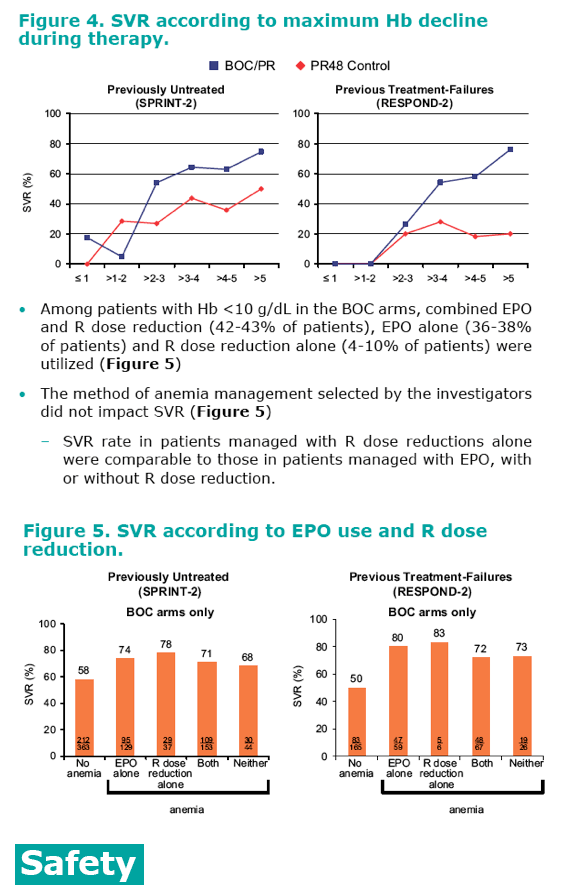
· Patients who experienced large declines in Hb were more likely to achieve SVR. In both studies, patients in the BOC/PR arms who
had a >5 g/dL maximum decrease in Hb attained SVR rates of 75-
76%, compared with 20-50% in PR48 control (Figure 4)
References
1. Sulkowski MS, Shiffman ML, Afdhal NH, et al. Hepatitis C virus treatment-related anemia is associated with higher sustained virologic response rate. Gastroenterology. 2010;139(5):1602-1611.
2. Afdhal NH, Dieterich DT, Pockros PJ, et al. Epoetin alfa maintains ribavirin dose in HCV-infected patients: a prospective, double-blind, randomized controlled study. Gastroenterology. 2004;126(5):1302-1311.
3. Pockros PJ, Shiffman ML, Schiff ER, et al. Epoetin alfa improves quality of life in anemic HCV-infected patients receiving combination therapy. Hepatology. 2004;40(6):1450-1458.
4. Thevenot T, Cadranel JF, Di Martino V, et al. A national French survey on the use of growth factors as adjuvant treatment of chronic hepatitis C. Hepatology. 2007;45(2):377-383.
5. Marcellin P, Forns X, Goeser T, et al. Telaprevir is effective given every 8 or 12 hours with ribavirin and peginterferon alfa-2a or -2b to patients with chronic hepatitis C. Gastroenterology. 2011;140(2):459-468.
6. Reiser M, Timm J. Expert Review of Anti-infective therapy. Expert Reviews. 2009;7(5):537-547.
7. Final Report: Assessment of the effects of boceprevir (SCH 503034) on red blood cells in healthy male volunteers (study report for Protocol No. P05351). Kenilworth (NJ): Schering-Plough Corporation. Clin Document 3723952
2011.
8. AFSSAPS Institut National du Cancer. Epoetine beta. Available at http://www.afssaps.fr/.
9. VHA Pharmacy Benefits Management Strategic Healthcare Group and Medical Advisory Panel. Recombinant Erythropoietin Criteria for Use for Hepatitis C Treatment-Related Anemia. Available at http://vaww.pbm.va.gov. VA 2011.
|
| |
|
 |
 |
|
|