 |
 |
 |
| |
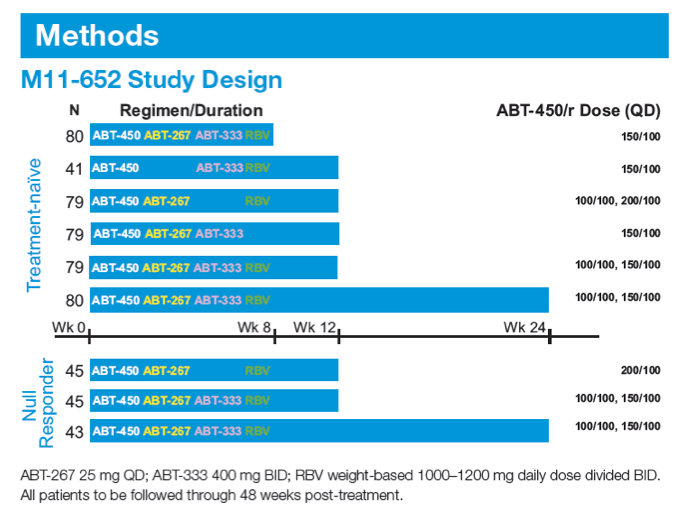
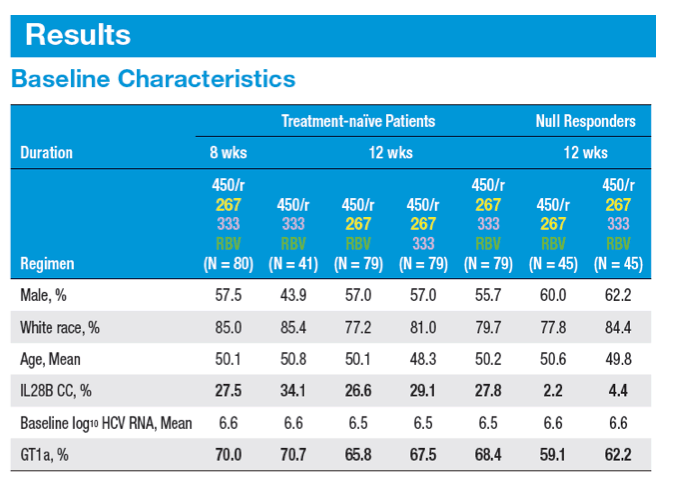
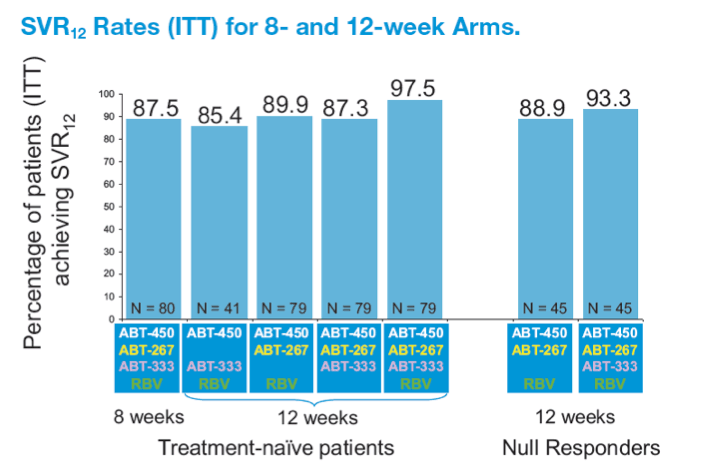
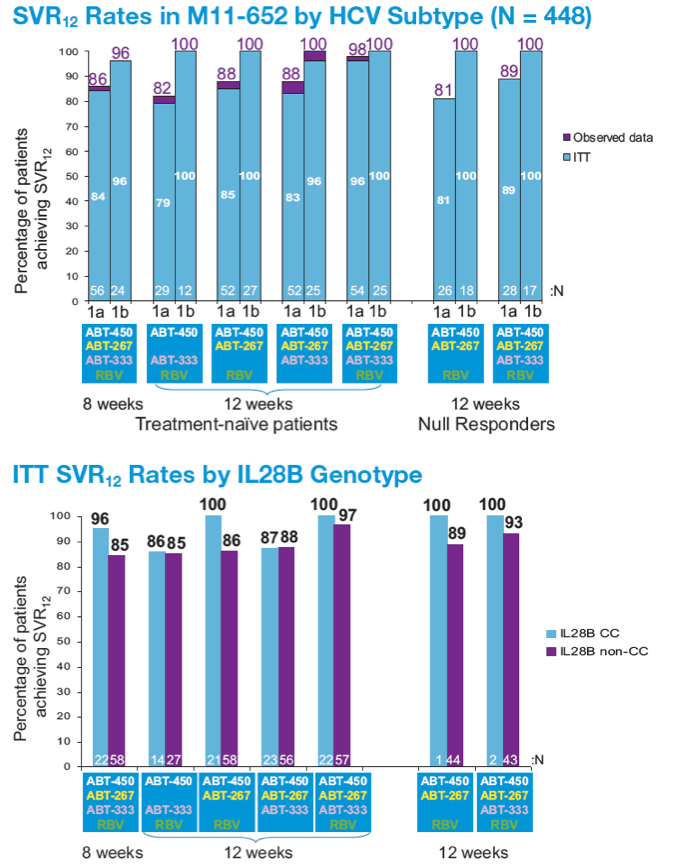
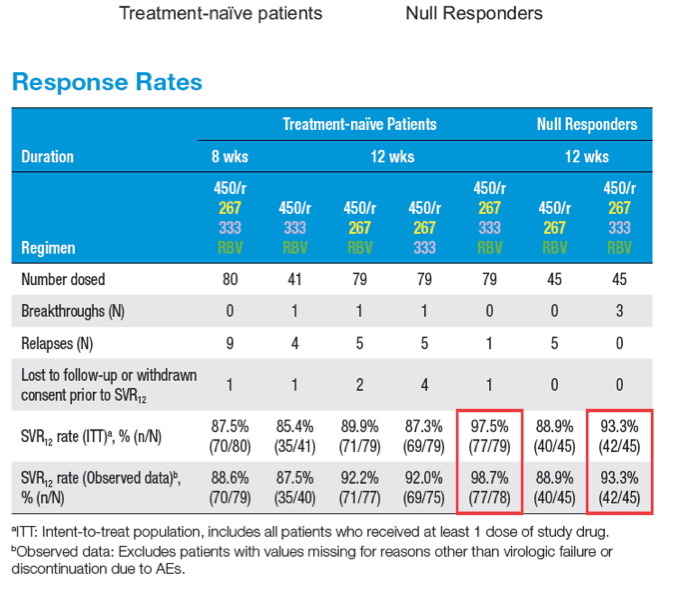
A 12-week Interferon-free Treatment Regimen With ABT-450/r, ABT 267, ABT-333, and Ribavirin Achieves SVR12 Rates (Observed Data) of 99% in Treatment-naïve Patients and 93% in Prior Null Responders With HCV Genotype 1 Infection
|
| |
| |
Reported by Jules Levin
AASLD 2012 Nov 9-13 Boston
Kris V Kowdley1, Eric Lawitz2, Fred Poordad3, Daniel E Cohen4, David Nelson5,
Stefan Zeuzem6, Gregory T Everson7, Paul Kwo8, Graham R Foster9, Mark Sulkowski10,
Wangang Xie4, Tami Pilot-Matias4, George Liossis4, Lois Larsen4, Amit Khatri4,
Thomas Podsadecki4, Barry Bernstein4
1Digestive Disease Institute, Virginia Mason Medical Center, Seattle,
Washington, United States; 2Alamo Medical Research,
San Antonio, Texas, United States; 3University of Texas Health Science Center, San Antonio, Texas, United States; 4Abbott Laboratories, Abbott Park, Illinois, United States; 5University of Florida College of Medicine, Gainesville, Florida, United States; 6J.W. Goethe University, Frankfurt, Germany; 7University of Colorado Denver, Aurora, Colorado, United States; 8Indiana University, Indianapolis, Indiana, United States; 9Queen Marys University of London, Barts Health, London, United Kingdom; 10Johns Hopkins University, Baltimore, Maryland, United States



|
| |
|
 |
 |
|
|