 |
 |
 |
| |
RITONAVIR SUBSTANTIALLY REDUCES REACTIVE METABOLITE FORMATION OF THE HCV PROTEASE INHIBITOR DANOPREVIR BOTH IN VITRO AND IN VIVO
|
| |
| |
Reported by Jules Levin
EASL 2012 Barcelona Spain Apr 18-22
P. Goelzer,1 P. Morcos,1 J. Tran,2 B. Wen,1 N. Shulman,3 P. Smith,1 T. Singer,4 B. Brennan,1 J. Hammond1
1Roche, Nutley, NJ, USA; 2Roche, Palo Alto, CA, USA; 3Genentech Inc., South San Francisco, USA; 4F. Hoffmann-La Roche Ltd, Basel, Switzerland
SUMMARY
Ritonavir inhibits DNV reactive metabolite formation in vitro through inhibition of CYP3A metabolism.
Human plasma samples showed numerous metabolites when DNV was administered alone, while no oxidative metabolites were identified when DNV was administered in combination with ritonavir.
· Ritonavir coadministration resulted in significantly higher levels of DNV in plasma with lower levels of oxidative metabolites in faeces.Oxidative metabolites reduced from ~25% of total radioactivity for DNV alone to ~2% for DNVr
· When coadministered with ritonavir, DNV is probably predominantly excreted unchanged into bile
· Ritonavir significantly decreases the proportion of metabolites relative to unchanged drug in plasma.
These results indicate a significant reduction of CYP3A-mediated metabolism in vivo.
INTRODUCTION
Danoprevir (DNV) is a potent, macrocylic, second-generation HCV protease inhibitor that has similar in vitro activity against HCV genotypes 1, 4 and 6.[1,2]
In the phase II ATLAS trial, the highest DNV dose arm (900 mg q12h) was prematurely discontinued due to reversible Grade 4 alanine aminotransferase (ALT) elevations in three patients.[3]
Considering the role of reactive metabolites in drug-induced liver injury[4] the potential for DNV to form reactive metabolites was evaluated and the effect of low-dose ritonavir on the metabolic profile of DNV was investigated.
OBJECTIVES
To explore the potential association between the formation of DNV reactive metabolites and ALT elevations in patients receiving DNV.
To explore the potential role of ritonavir boosting in reducing reactive metabolite formation.
DATA SOURCES
Data from four sources were evaluated:
1: Human liver microsomes
2: Circulating metabolites in vivo
3: Human mass balance study
4: PK comparison of DNV exposure in the ATLAS[3] (with DNV) and DAUPHINE[5]
(with ritonavir-boosted DNV, DNVr) studies
STUDY 1: HUMAN LIVER MICROSOMES
METHODS
Human liver microsomes were incubated with 14C-labelled DNV (10 μM) in the absence and presence of ritonavir (1, 5 and 25 μM). Measurements of radioactivity indicated the amount of DNV that was covalently bound to microsomal proteins.
RESULTS
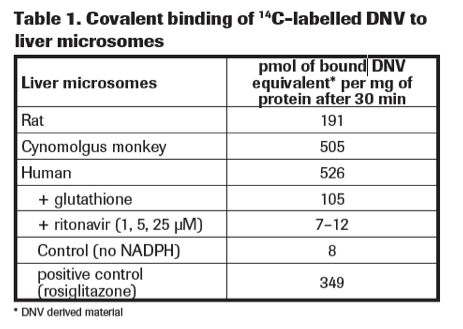
High covalent binding of DNV to rat, monkey and human liver microsomes was observed (Table 1).
Binding in cynomolgus monkey liver microsomes and human liver microsomes was similar to the positive control (rosiglitazone). Addition of glutathione reduces covalent binding by trapping reactive metabolites (Table 1).
Addition of ritonavir completely blocked covalent binding to levels similar to control experiments without NADPH (Table 1).
STUDY 2: CIRCULATING METABOLITES IN VIVO
METHODS
Plasma samples from patients receiving DNV (900 mg q12h) or DNV plus low-dose ritonavir (DNVr: 200 mg DNV plus 100 mg ritonavir q12h) were qualitatively assessed for metabolites using liquid chromatography-tandem mass spectrometry (LC-MS/MS).
RESULTS
Among patients receiving DNV (900 mg q12h), up to 20 different DNV metabolites were qualitatively detected.In the phase II ATLAS study the 900 mg q12h regimen was associated with the highest incidence of ALT increases.[3]
Among patients receiving DNVr (200 mg DNV plus 100 mg ritonavir q12h), no circulating DNV metabolites were detected in plasma samples.
STUDY 3: HUMAN MASS BALANCE STUDY
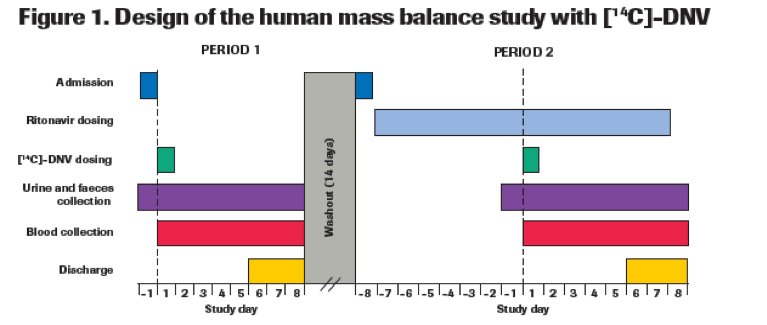
METHODS
The study enrolled 10 healthy volunteers, and included two single doses of [14C]-DNV (100 mg DNV containing 100 μCi [14C]-DNV) separated by a washout period of at least 14 days (Figure 1).Period 1: Subjects received a single 100 mg oral dose of [14C]-DNV alone.
Period 2: Ritonavir 100 mg was administered twice daily from 7 days before and until up to day 7 after receiving a single oral dose of 100 mg [14C]-DNV.
ANALYSIS
· Plasma concentrations of DNV and ritonavir were measured using LC-MS/MS.
· Radioactivity in urine and faeces was measured using liquid scintillation counting.
· Radioactivity in blood and plasma was measured by accelerator mass spectroscopy (AMS).
· Metabolite profiles in urine and faeces were generated by LC-MS/MS.
· Metabolite profiles in plasma were generated by high-performance liquid chromatography (HPLC) fraction collection and AMS.
RESULTS
EXCRETION
· The majority of [14C]-DNV-related radioactivity was excreted via the faeces (89-96%). This is likely to be a combination of unabsorbed and biliary-excreted radioactivity.
· Only 0.1-0.2% of radioactivity was excreted via the urine.
METABOLISM
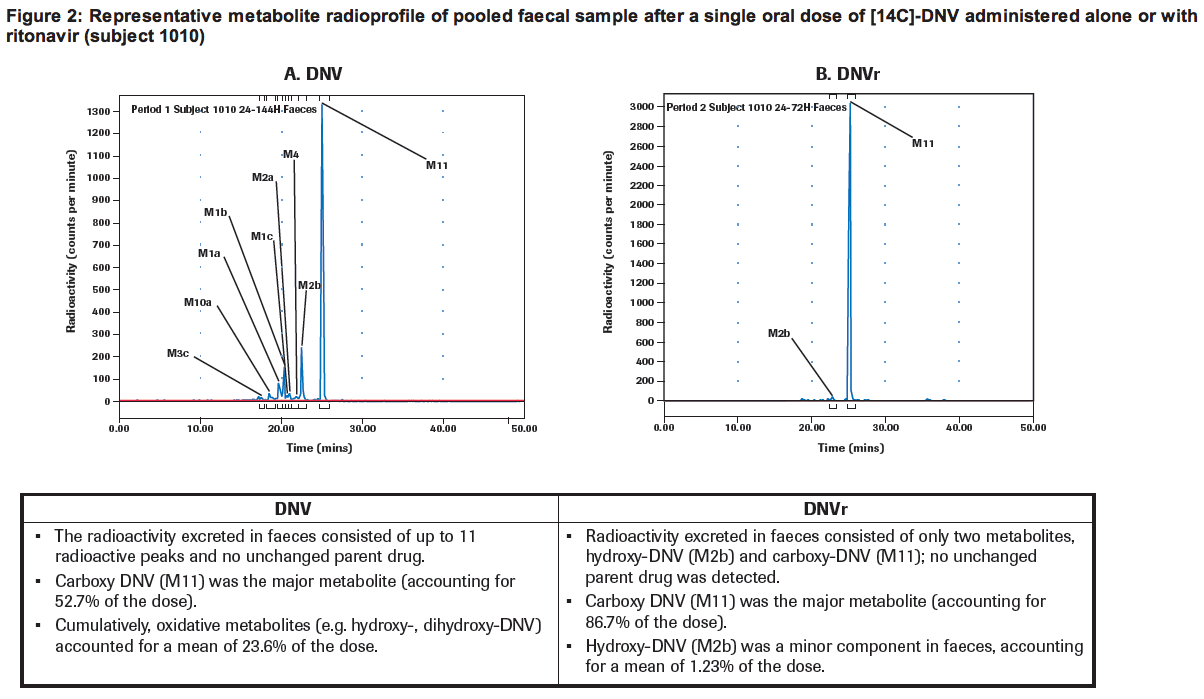
· The metabolism of [14C]-DNV involved oxidation to numerous metabolites and hydrolysis to the metabolite M11 (Figure 2A).
· The main oxidative metabolic pathway of absorbed DNV is likely to be via the cytochrome P450 family member CYP3A, while hydrolysis occurs probably mainly in the faeces (no parent drug was found in faeces).
· The addition of ritonavir led to substantially decreased levels of oxidative metabolites in faeces (Figure 2B), suggesting that DNV is predominantly excreted unchanged via the bile when administered with ritonavir.
· The proportion of oxidative metabolites was higher for DNV alone compared with ritonavir-boosted DNV, being ~25% and ~2% of total radioactivity, respectively (Figure 2).
Figure 2: Representative metabolite radioprofile of pooled faecal sample after a single oral dose of [14C]-DNV administered alone or with ritonavir (subject 1010)
|
| |
|
 |
 |
|
|