 |
 |
 |
| |
Alisporivir (ALV) plus Peg-interferon/Ribavirin (PR) in HCV G1 Treatment-experienced Patients Achieves
Primary Endpoint with Superior Efficacy at Treatment Week 12 Compared to Retreatment with PR
|
| |
| |
Reported by Jules Levin
EASL 2012 Apr 18-22 Barcelona Spain
Alfredo Alberti1, Wan-Long Chuang2, Robert Flisiak3, Giusseppe Mazzella4, Andrzej Horban5, Tobias Goeser6, Pietro Calistru7, Maria Buti8, Gary Davis9, Yankun Gong10, Claudio Avila11, Jia-Horng Kao12
1Azienda Ospedaliera Università di Padova Univ.degli Studi, Padova, Italy; 2Kaohsiung Medical University Hospital, Kaohsiung Medical University, Kaohsiung, Taiwan; 3Medical University, Bialystok, Poland; 4Az.Osp. di Bologna Policl. S.Orsola-Malpighi, Bologna, Italy; 5Klinika Obserwacyjno-Zakazna, SP ZOZ Wojewodzki Szpital Zakazny, Warsaw, Poland; 6Klinik für Gastroenterologie und Hepatologie Universitätsklinik Köln, Germany; 7Centrul de Diagnostic si Tratament Dr. Viktor Babes, Bucharest, Romania; 8Medicina Interna Hospital Vall D'Hebron, Barcelona, Spain; 9Baylor Health, Dallas, Texas, USA; 10Beijing Novartis Pharma Co. Ltd., Beijing, China; 11Novartis Pharma AG, Basel, Switzerland; 12National Taiwan University Hospital, Taipei, Taiwan


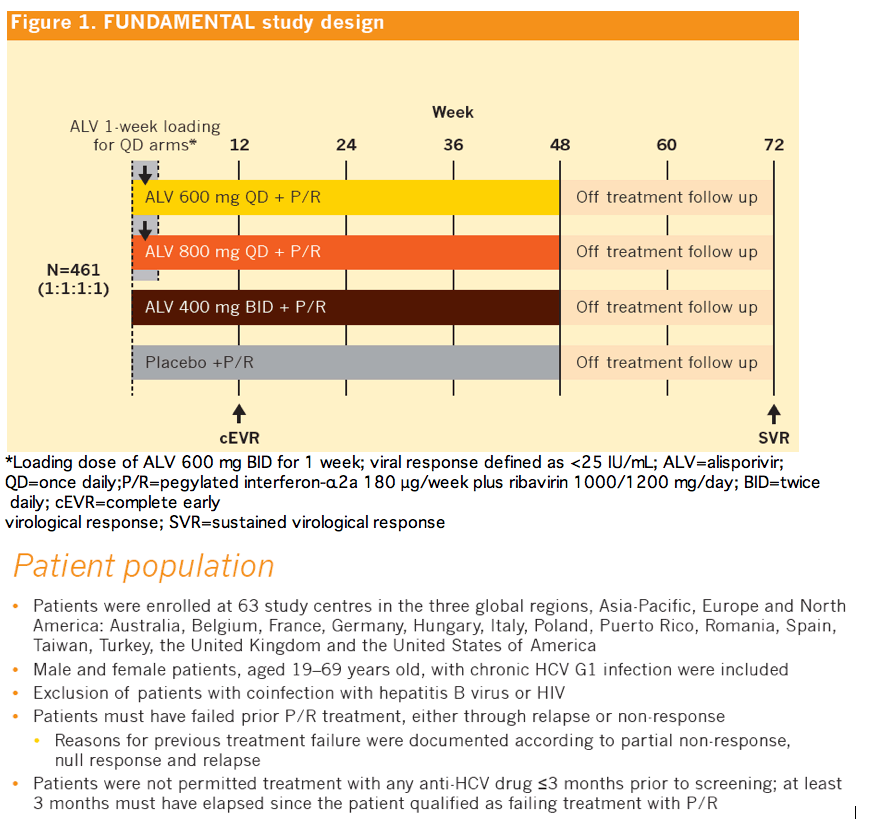

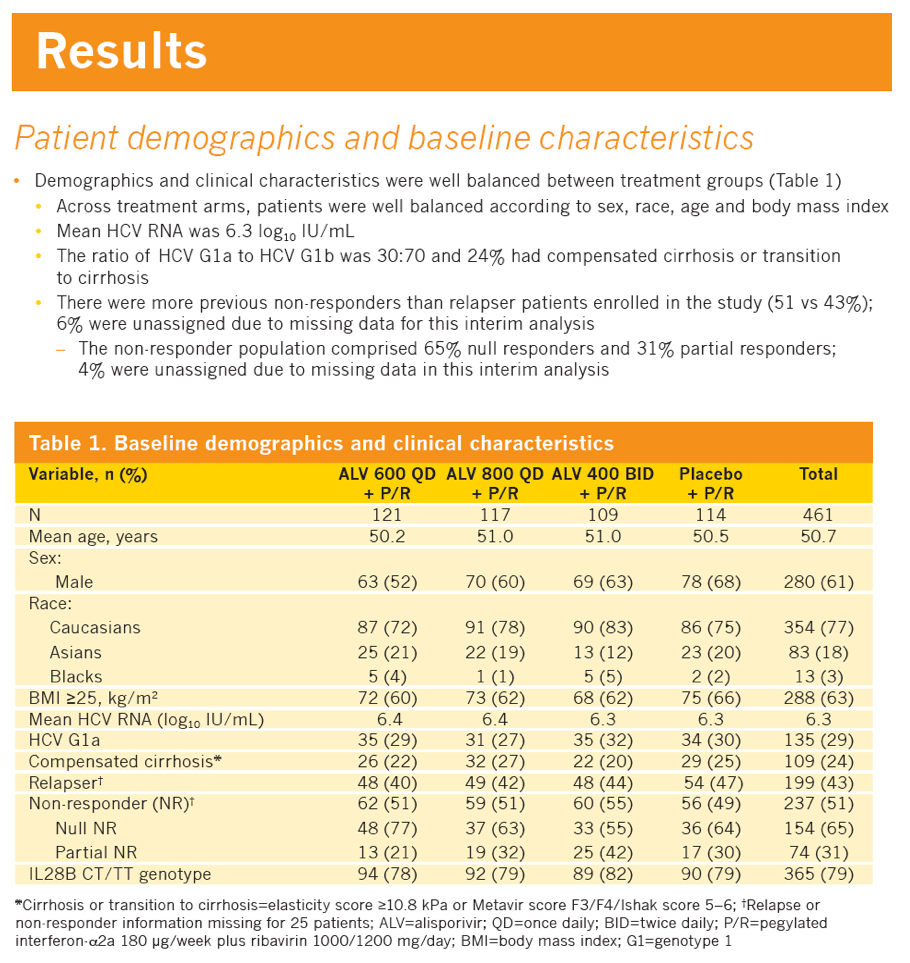

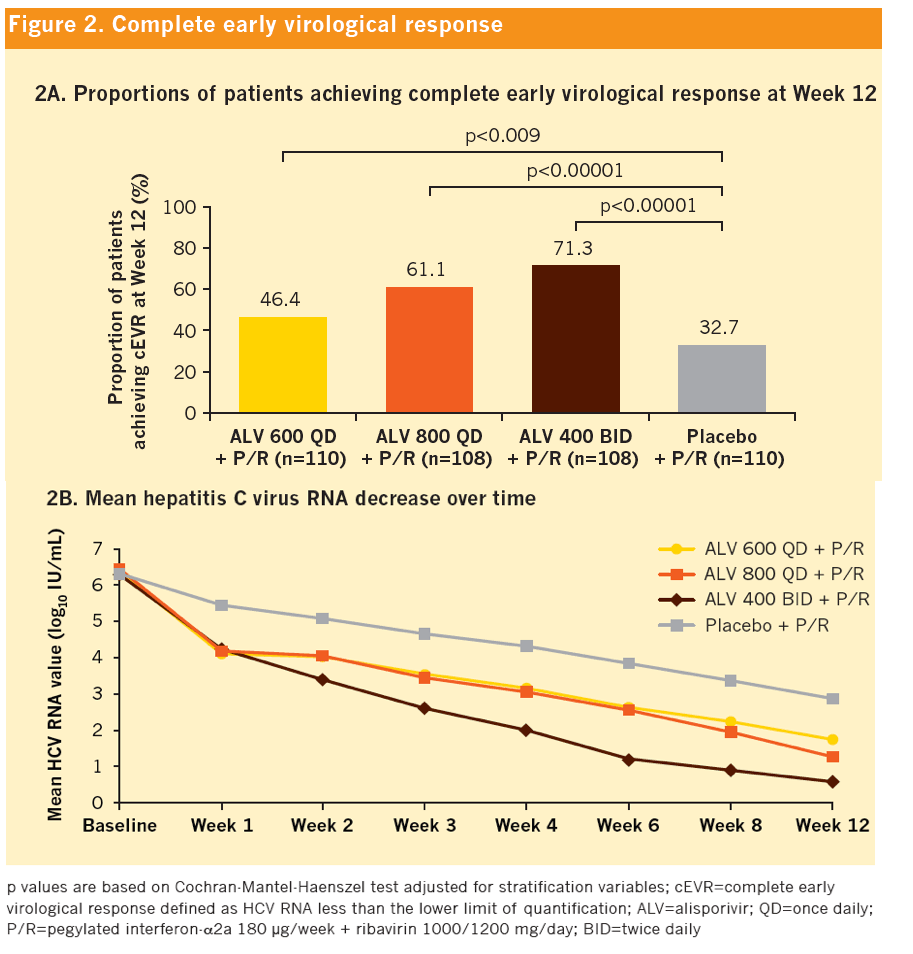
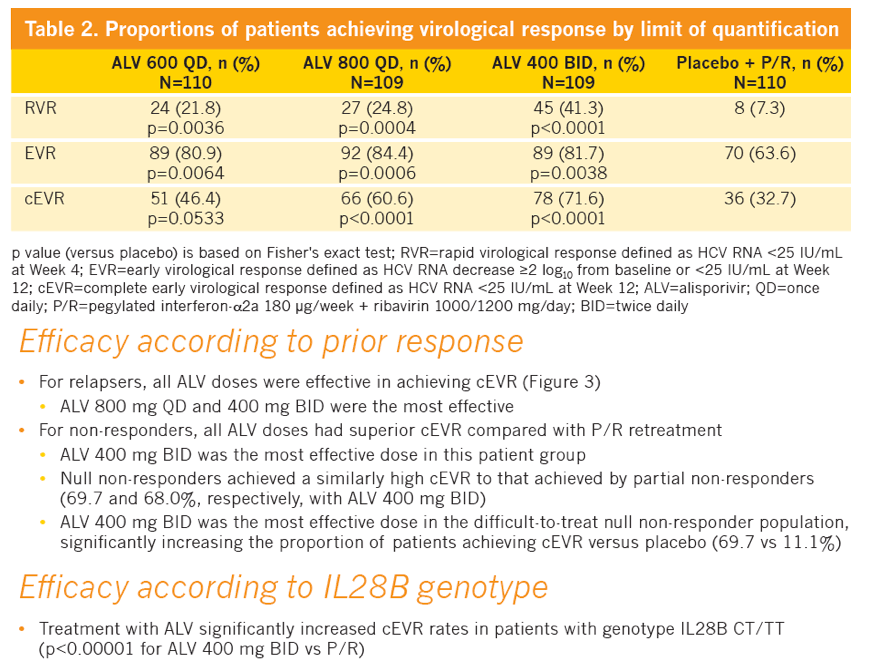
|
| |
|
 |
 |
|
|