 |
 |
 |
| |
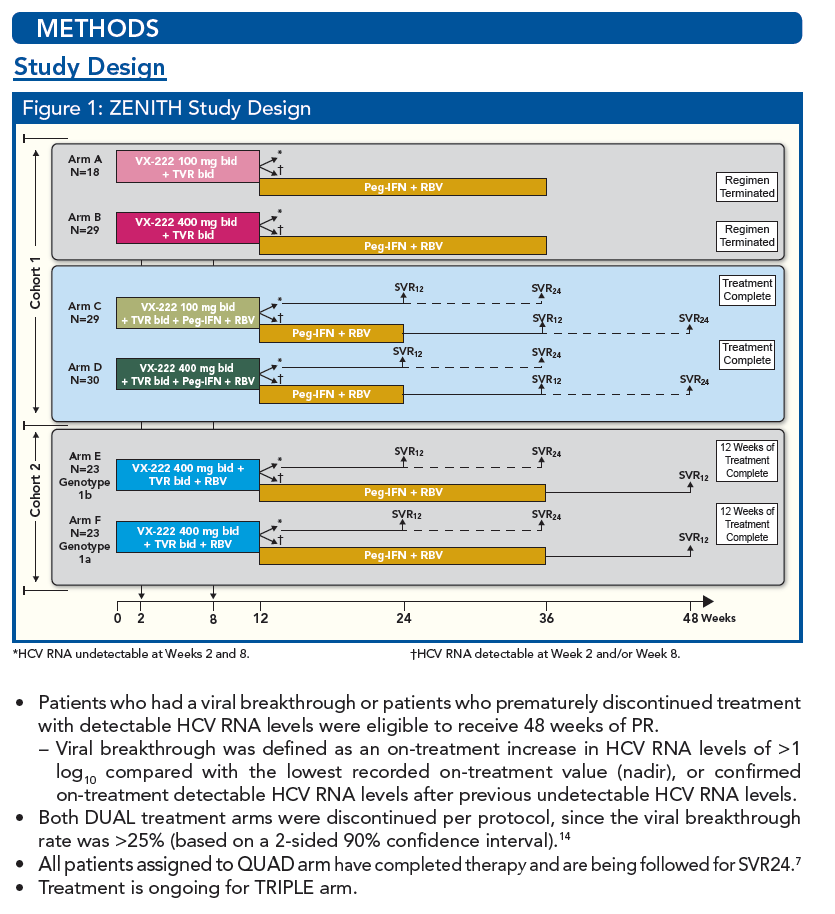
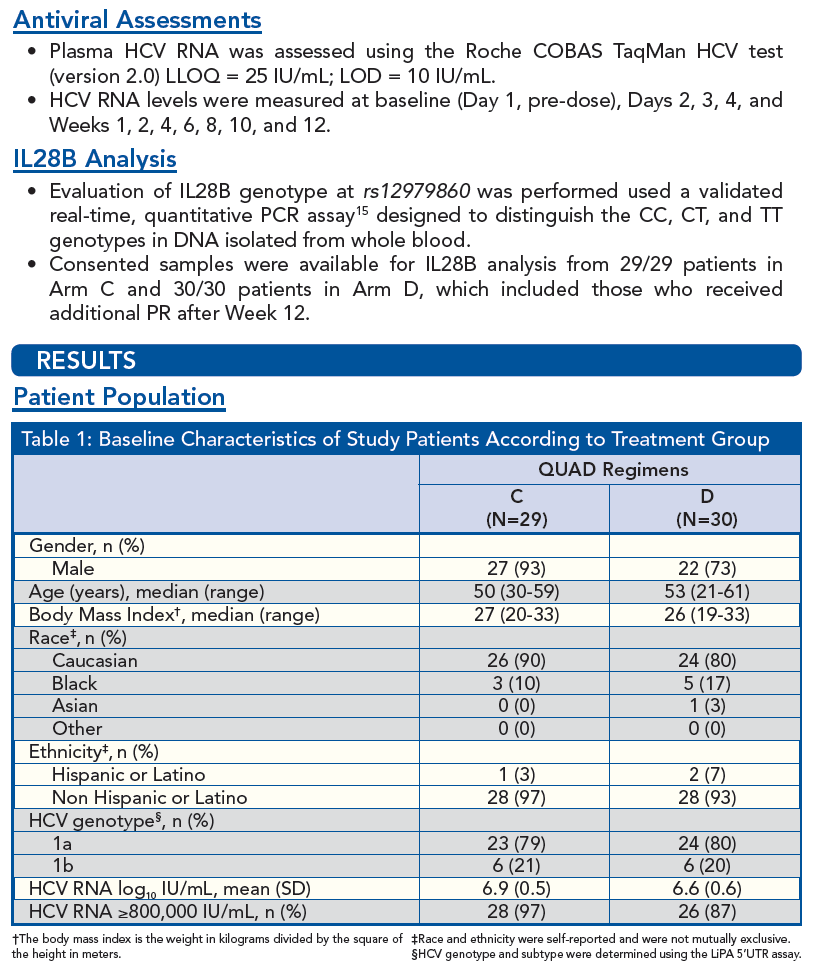
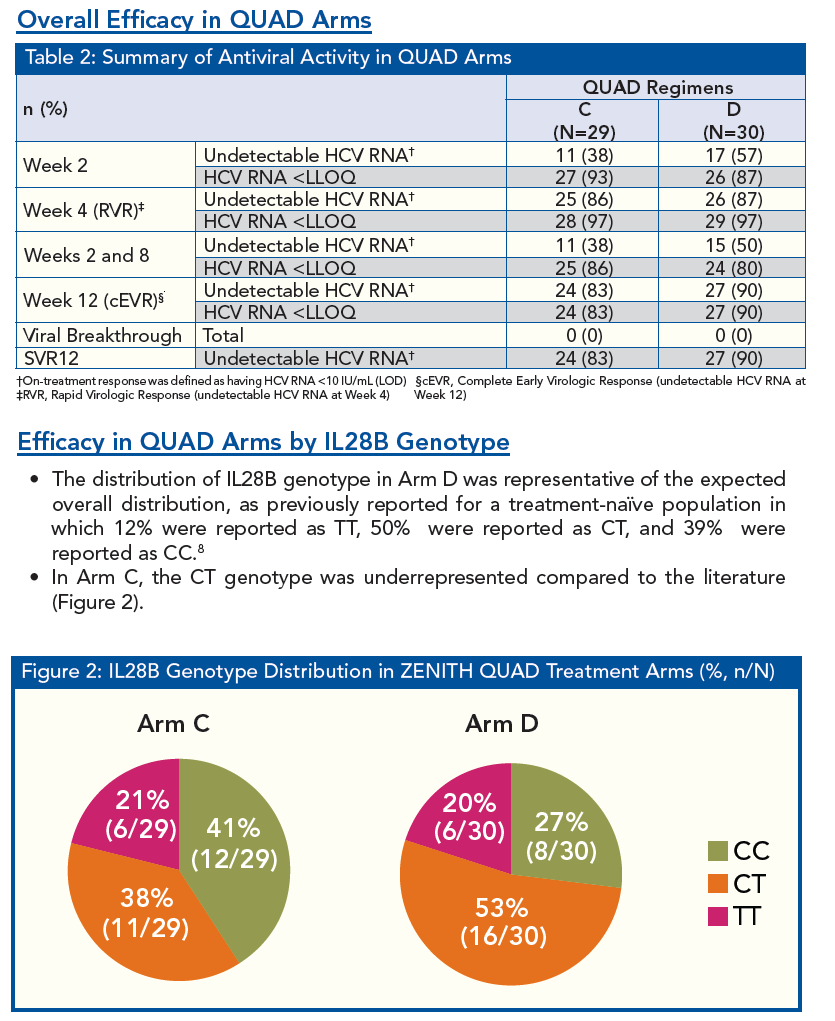
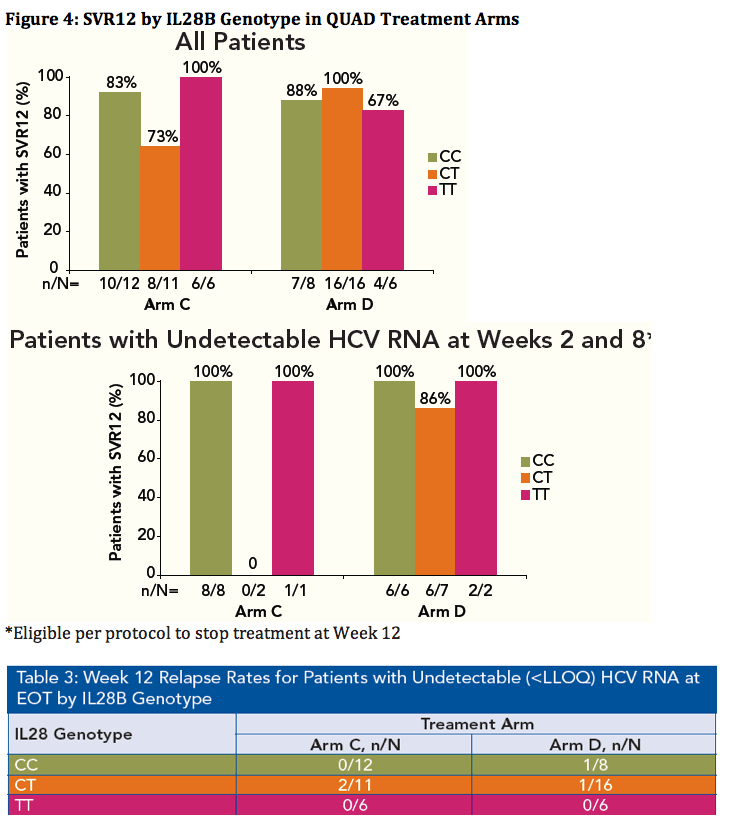
Patients of all IL28B Genotypes have High SVR Rates when Treated with VX-222 in Combination with Telaprevir/Peginterferon/Ribavirin in the ZENITH Study
|
| |
| |
Reported by Jules Levin
47th Annual Meeting of the European Association for the Study of the Liver (EASL), The International Liver Congress, Barcelona, Spain, 18-22 April 2012
M.S. Penney1*, C. DeSouza1, S. Seepersaud1, K. Alves1, M.J. Koziel1, R.S. Kauffman1, A.M. Di
1. Vertex Pharmaceuticals Incorporated, Cambridge, MA; 2. Saint Louis University School of Medicine, Saint Louis, MO, USA.

YOU will note arms E & F are interferon-free, VX222 400mg bid + Telaprevir bid + Ribavirin, in 46 patients for 12 weeks, with an option of adding peg/rbv after 12 weeks of IFN-free. The results are from part of the study are not ready yet for presentation so are not in this poster but we expect it soon. Vertex has 2 nucleotides (acquired from Alios) that they are studying now in phase 1 with results expected soon, within a few months, whereupon based on these results from the phase 1 nucleotide studies new studies will quickly be designed including all of the Vertex orals which in total add up to a potential of 4 oral drugs that can be utilized in such studies (telaprevir, VX222, 2 nucleotides).
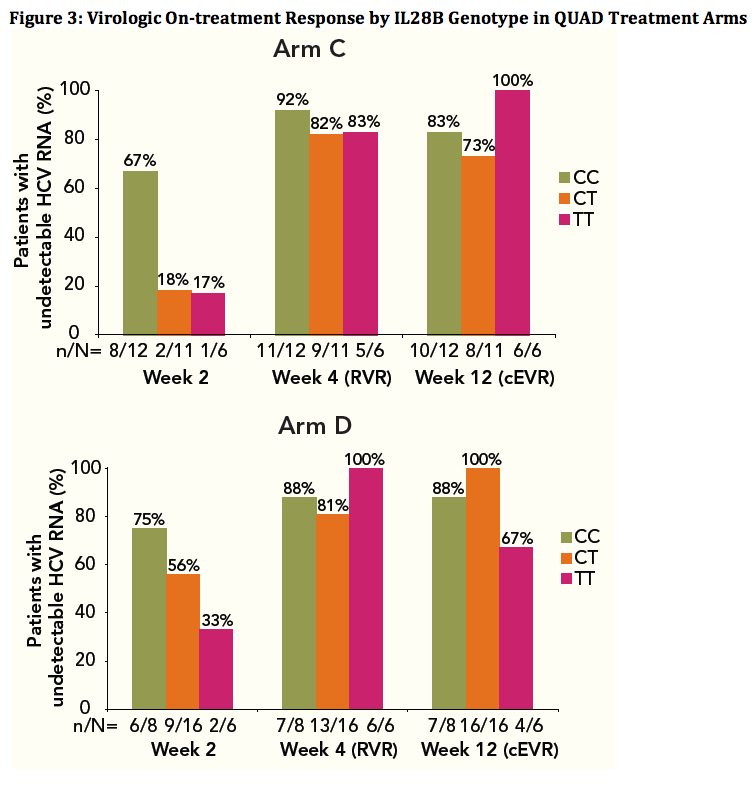

|
| |
|
 |
 |
|
|