 |
 |
 |
| |
PRECLINICAL CHARACTERISTICS OF ACH-3102:
A NOVEL HCV NS5A INHIBITOR WITH IMPROVED POTENCY AGAINST GENOTYPE-1A VIRUS AND VARIANTS RESISTANT TO 1ST GENERATION NS5A INHIBITORS
|
| |
| |
Reported by Jules Levin
EASL 2012
Yongsen Zhao, Jason Wiles, Guangwei Yang, Dharaben Patel,
Joanne Fabrycki, Samantha Weinheimer, Christopher Marlor,
Jose Rivera, Qiuping Wang, Venkat Gadhachanda,
Akihiro Hashimoto, Dawei Chen, Godwin Pais, Xiangzhu Wang,
Milind Deshpande, Kathe Stauber, Mingjun Huang and
Avinash Phadke
CONCLUSIONS
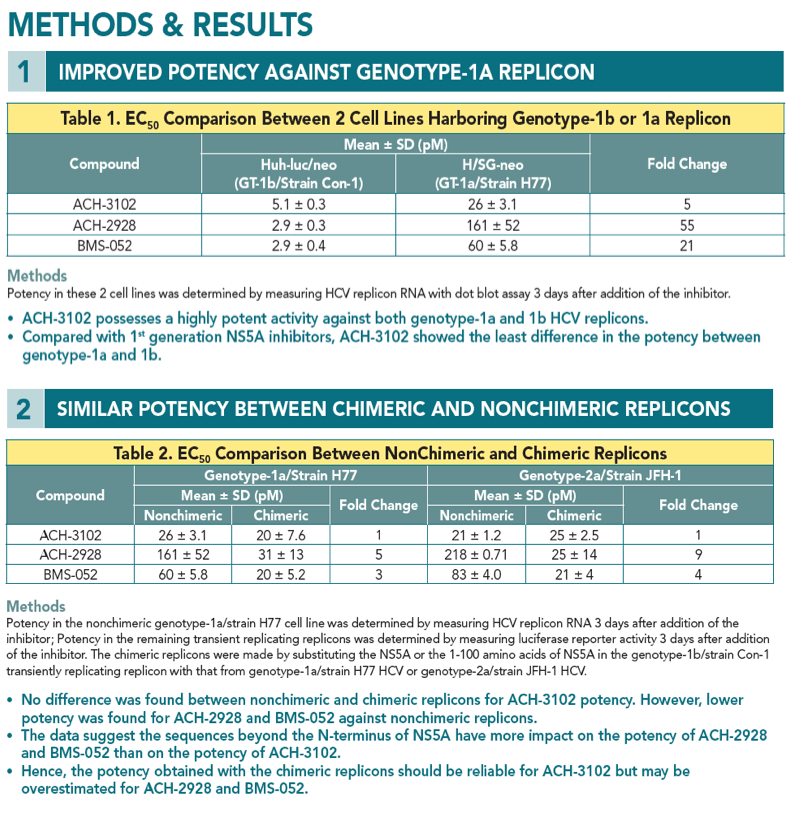
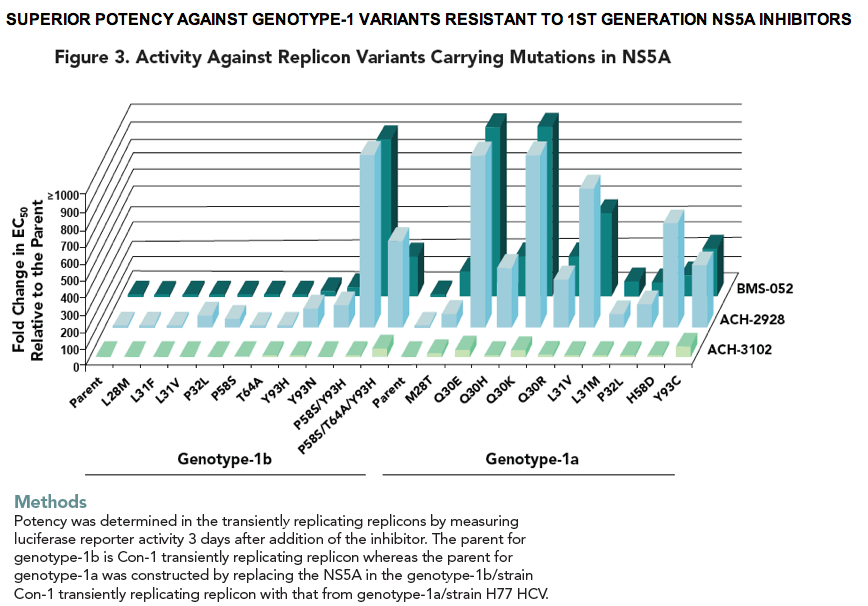
· ACH-3102 displays improved potency against genotypes 1-6 HCV compared with 1st generation NS5A inhibitors.
· The majority of mutations that confer high resistance to 1st generation NS5A inhibitors do not confer resistance to ACH-3102.
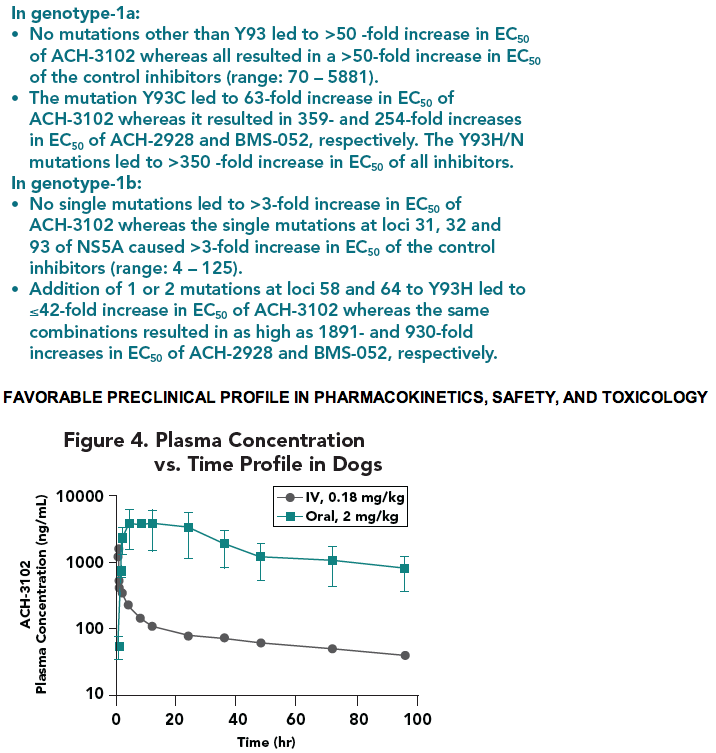
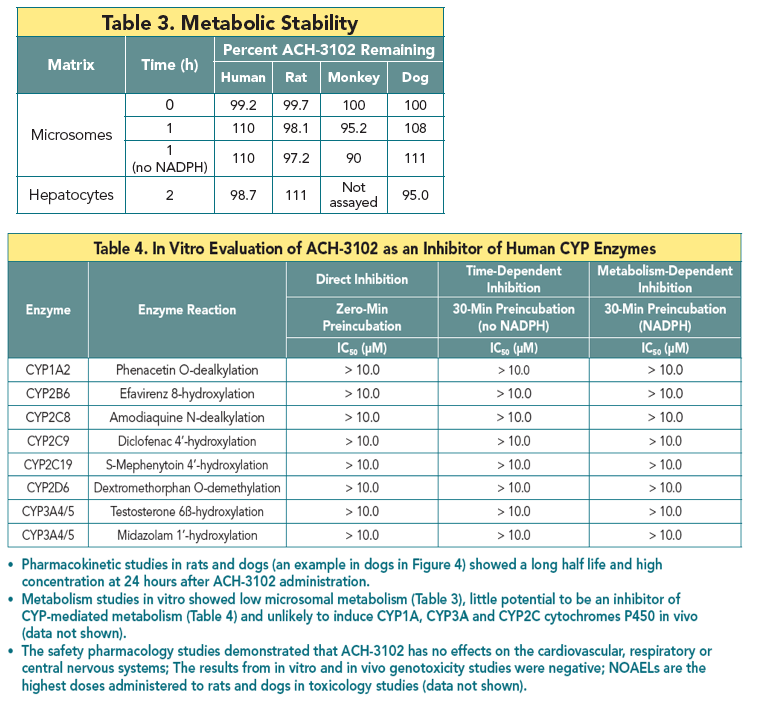
· ADME studies demonstrated that ACH-3012 is a once a day drug and is unlikely to impose drug-drug interactions.
· The results of the genetic toxicology studies, the safety pharmacology studies, and the general toxicity studies in rats and dogs
support the use of ACH-3102 in humans for chronic hepatitis C infection.
· Clinical investigation of ACH-3102 is on-going.
· Clinical investigation of ACH-3102 in combination with ACH-1625 (NS3 protease inhibitor currently in phase 2a) is planned.
ABSTRACT
Background The 1st generation NS5A inhibitors display picomolar potency against genotype-1b replicons but are less active against genotype-1a replicons. Consequently, the clinical efficacy of the 1st generation NS5A inhibitors is lower in genotype-1a patients than in genotype-1b patients. Here, we report antiviral activity, specifi city, safety, and pharmacokinetic characteristics of a novel NS5A inhibitor ACH-3102 that support once-daily dosing in genotype-1 to 6 hepatitis C patients.
Methods Optimization of NS5A inhibitors was achieved through evaluation of potency in cell lines harboring wild-type or resistant genotype-1a and 1b HCV replicons. Various chimeric HCV replicons were constructed to assess the anti-HCV spectrum of the inhibitors. ADME characteristics, pharmacokinetic properties, and off-target activities were examined by standard procedures. In vivo safety was assessed after repeat dosing in rodent and non-rodent species.
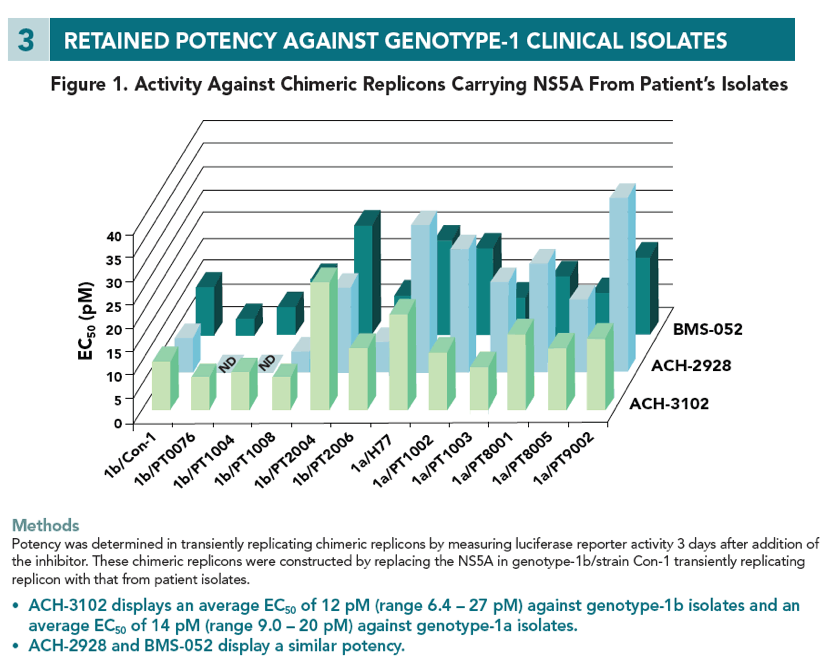
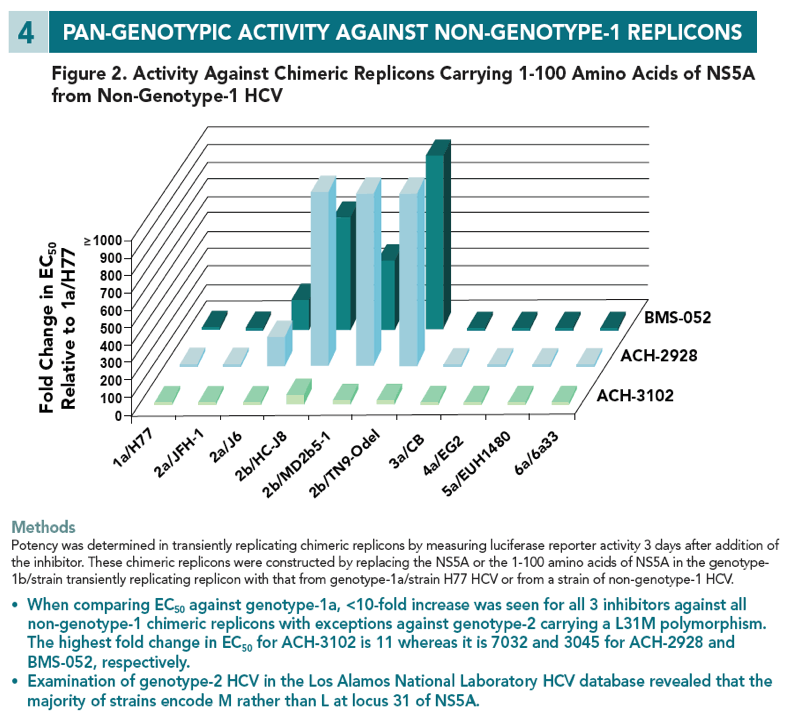
Results ACH-3102 displayed picomolar potency in cell lines harboring a laboratory strain 1b and 1a replicon. High potency was retained against a panel of chimeric replicons carrying NS5A from genotype-1a and 1b-HCV infected patients as well as from non-genotype-1 chimeric replicons and genotype-2 replicons. When evaluated in a panel of replicons carrying mutations at loci 28, 30, 31, 58, and 93 of NS5A, EC50 values were signifi cantly reduced compared with
the 1st generation NS5A inhibitors. ACH-3102 demonstrated excellent metabolic stability and displayed no potential for inhibition or induction of major CYP isoforms. Oral dosing to rats and dogs resulted in good exposure and half-life supporting QD dosing. The safety profi le of ACH-3102 supports progression to fi rst-in-man studies.
Conclusions ACH-3102 is a potent NS5A inhibitor with improved potency against genotype-1a virus, non-genotype-1 virus and variants resistant to the 1st generation of NS5A inhibitors. It shows low potential for off-target activity and metabolic drug-drug interactions. With its profound effect in vitro in preventing the emergence of resistant variants when combined with NS3 protease inhibitor ACH-1625 or ACH-2684, clinical evaluation of ACH-3102 in combination with ACH-2684 or ACH-1625 in hepatitis C patients is warranted.
|
| |
|
 |
 |
|
|