 |
 |
 |
| |
The clinical and economic impact of a generic first-line antiretroviral regimen in the U.S.
|
| |
| |
Presented by Rochelle P. Walensky (United States).
FRLBX06
Powerpoint
Webcast
R.P. Walensky1, P.E. Sax2, Y.M. Nakamura3, M.C. Weinstein4, P.P. Pei3, K.A. Freedberg1, A.D. Paltiel5, B.R. Schackman6
1Massachusetts General Hospital, Harvard Medical School, Boston, United States, 2Brigham and Women's Hospital, Harvard Medical School, Boston, United States, 3Massachusetts General Hospital, Boston, United States, 4Harvard School of Public Health, Boston, United States, 5Yale School of Medicine, New Haven, United States, 6Weill Cornell Medical College, New York, United States
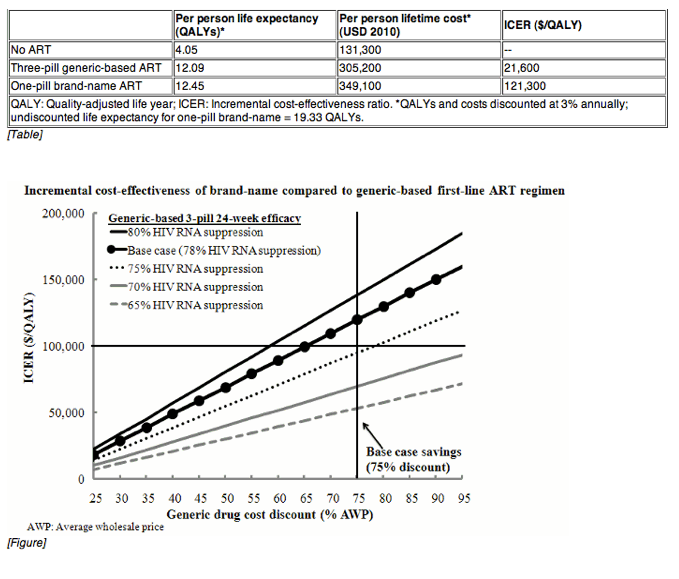
Background: US guidelines recommend once-daily, one-pill efavirenz/emtricitabine/tenofovir as a preferred first-line ART regimen. With the anticipated availability of generic efavirenz in the US, the cost of a once-daily, three-pill alternative (generic efavirenz, generic lamivudine, tenofovir) will decrease, but adherence and virologic suppression may be lower. We project the clinical impact, cost, and cost-effectiveness of the generic-based vs. brand-name options.
Methods: Using an HIV simulation model (CEPAC-US), we examine 3 strategies: 1) No ART (for comparison); 2) three-pill generic-based ART; and 3) one-pill brand-name ART. Inputs are from peer-reviewed data: ART efficacies (24-week suppression: 78% generic-based vs. 85% brand-name); drug costs are 25% of average wholesale price (AWP) for generics and 77% of AWP for others ($8,600/year vs. $15,370/year for the regimen); the cohort is 84% male, mean CD4 317/µl. From the US health system perspective (2010 USD), we report incremental cost-effectiveness ratios (ICERs, $/QALY) compared against a $100,000/QALY threshold. We also project the potential annual savings for those initiating ART (incident diagnoses, ~2,500) and those eligible to switch to the generic-based regimen (prevalent cases, ~147,000)
Results: Compared to No ART, generic-based ART has an ICER of $21,600/QALY. Compared to generic-based ART, brand-name ART increases lifetime costs by $43,900, and per-person survival gains by 0.36 quality-adjusted life years (due to increased efficacy and reduced resistance from emtricitabine compared to lamivudine), for an ICER of $121,300/QALY (Table). ICERs are sensitive to decreases in generic-based ART efficacy and to generic drug costs; most plausible combinations lead to ICERs >$100,000/QALY (Figure). Estimated annual savings if all eligible US incident/prevalent patients switched to the generic-based regimen are $1.01B.
Conclusions: Compared to a slightly-less effective generic-based regimen, the cost-effectiveness of the guideline-recommended brand-name regimen exceeds the $100,000/QALY threshold. Switching to generic-based regimens would yield substantial savings for programs that fund HIV treatment.
|
| |
|
 |
 |
|
|