 |
 |
 |
| |
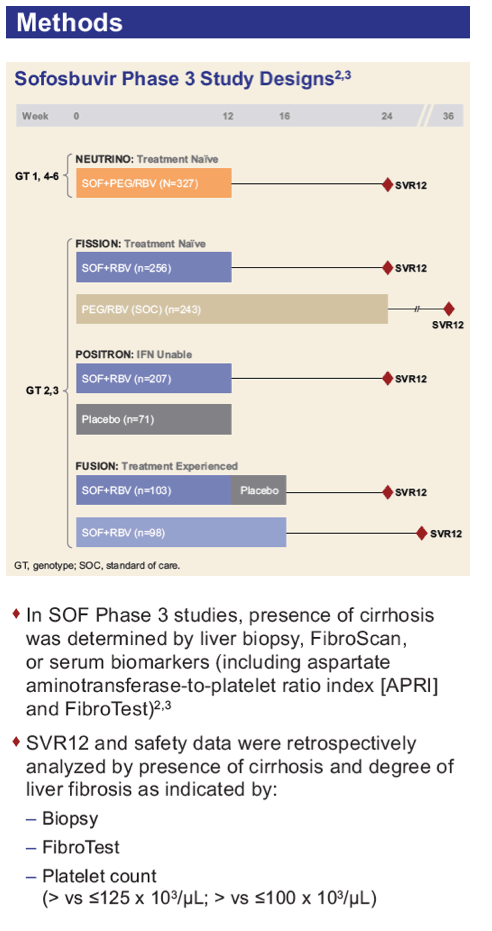
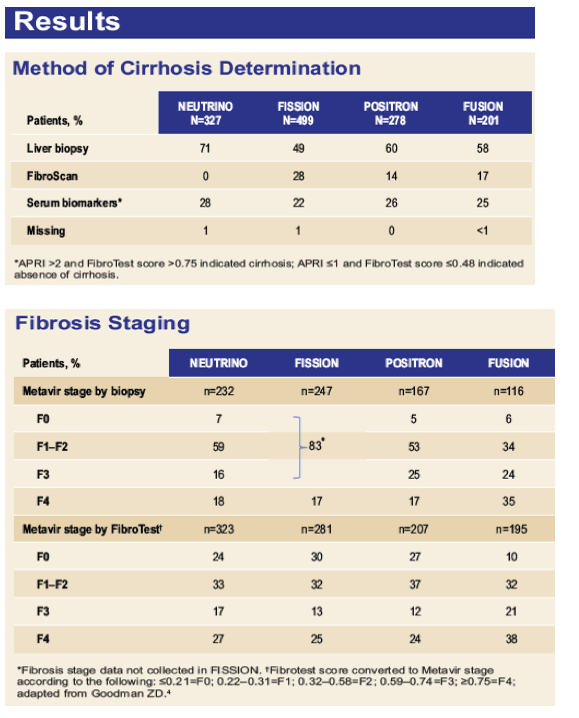
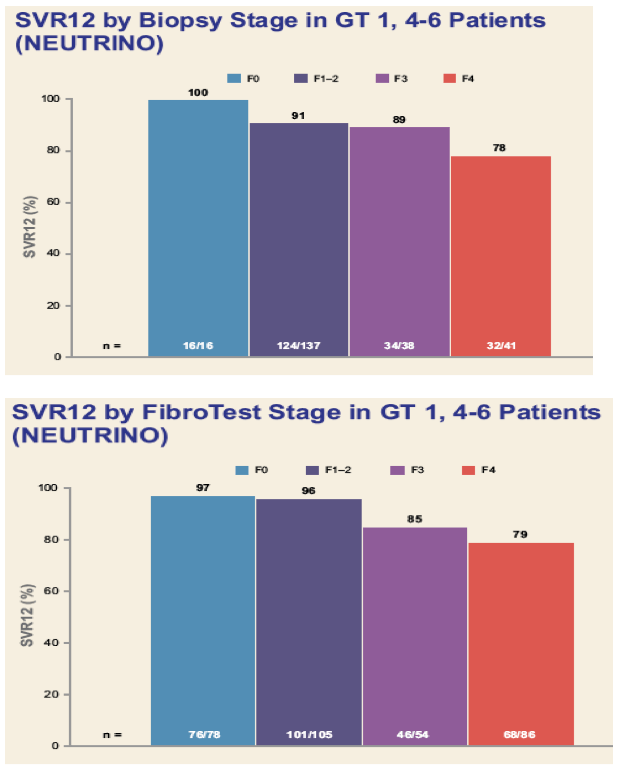
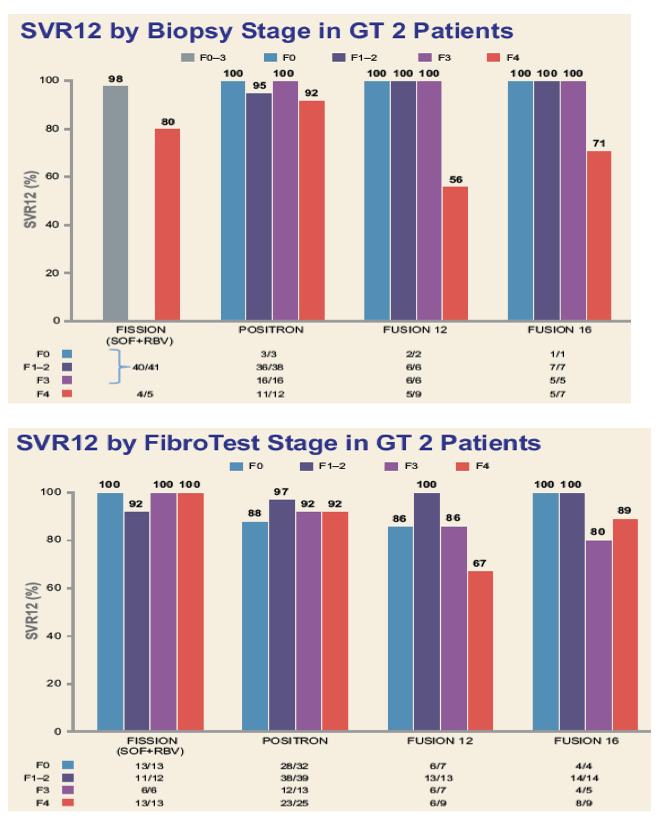
Efficacy and Safety of Sofosbuvir in Patients According to Fibrosis Stage: An Analysis of Phase 3 Data
|
| |
| |
Reported by Jules Levin
AASLD Nov 1-4 2013 Wash DC
Keyur Patel,1 Stuart C. Gordon,2 Aasim M. Sheikh,3 Ziad Younes,4 Di An,5 Shu-Min Chuang,5 Julie Ma,5 John McNally,5 Luisa M. Stamm,5
Diana M. Brainard,5 William T. Symonds,5 John G. McHutchison,5 Ronald Nahass,6 Eric M. Yoshida,7 Henk Reesink,8 David R. Nelson9
1Duke University Medical Center, Durham, NC; 2Henry Ford Health System, Detroit, MI; 3Gastrointestinal Specialists of Georgia, Marietta, GA; 4Gastro One, Germantown, TN; 5Gilead Sciences, Inc., Foster City, CA;
6ID Care, Hillsborough, NJ; 7University of British Columbia, Vancouver, British Columbia, Canada; 8Academic Medical Center, Amsterdam, the Netherlands; 9University of Florida, Gainesville, FL
AASLD: Gilead at AASLD: Sofosbuvir for GT 3, 2 and 1 - (11/18/13)

AASLD: Sofosbuvir + Ribavirin for 12 or 24 Weeks for Patients With HCV Genotype 2 or 3: the VALENCE Trial - (11/04/13)
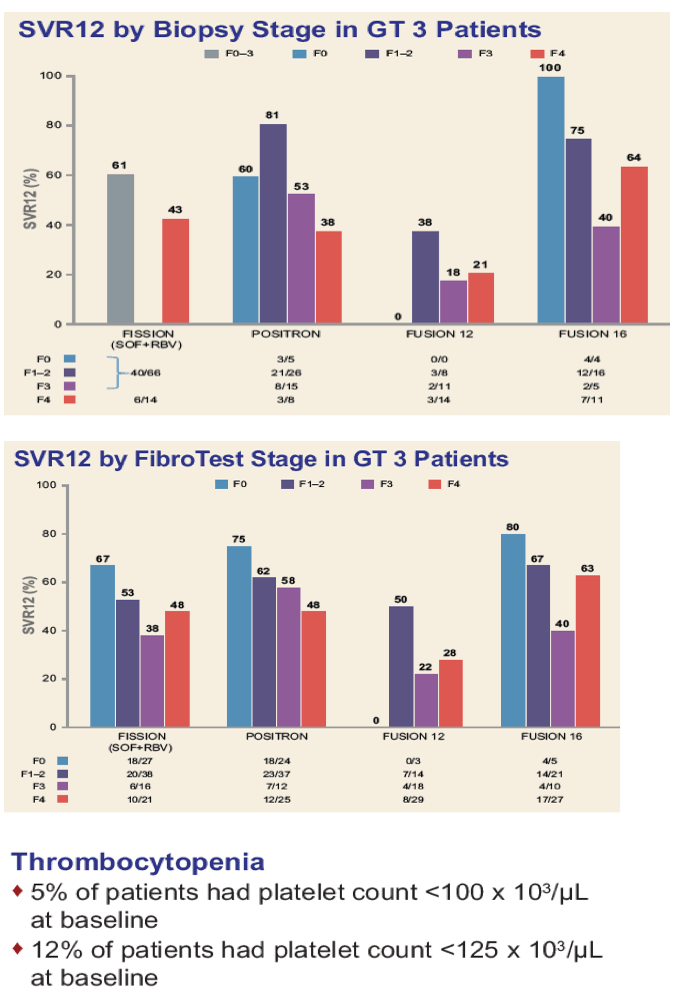
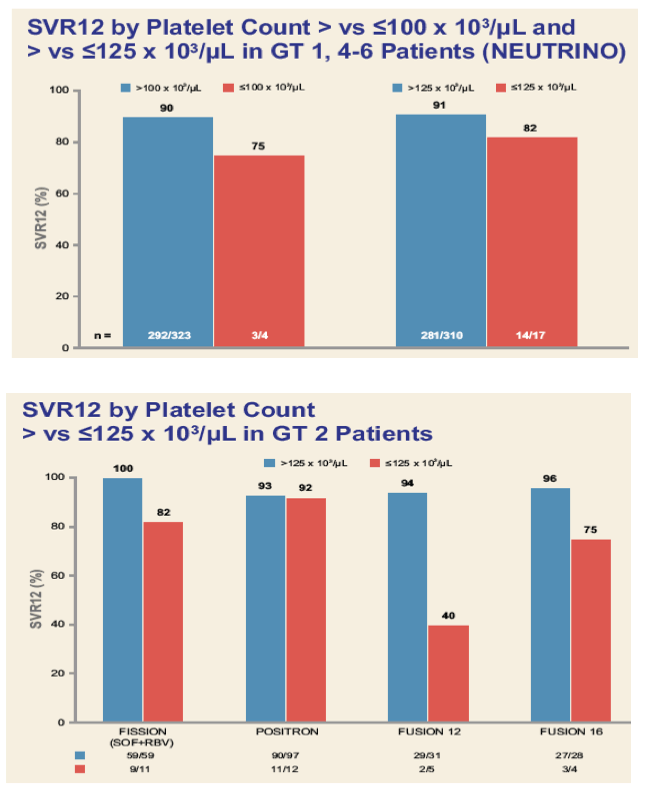
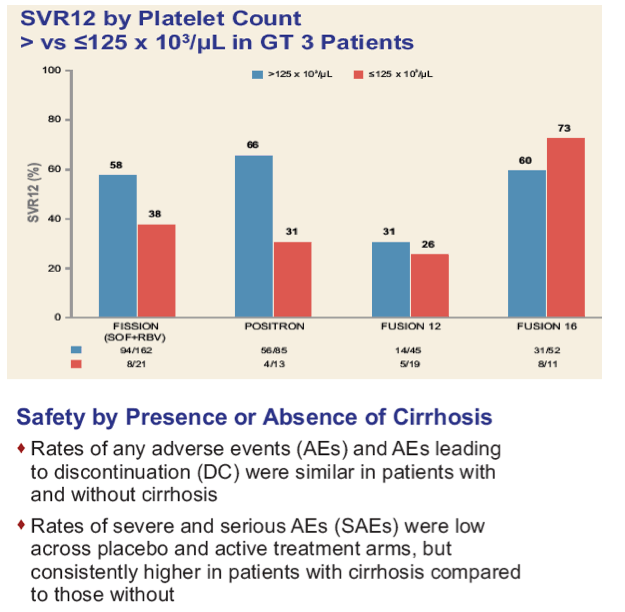
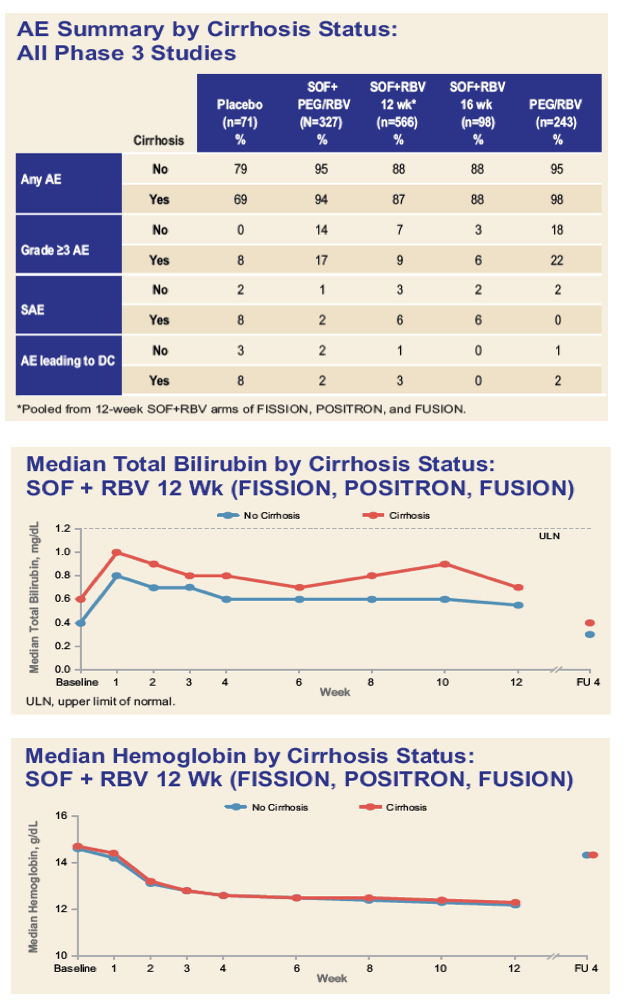

|
| |
|
 |
 |
|
|