 |
 |
 |
| |
Hepatitis B Virus Screening and Prevalence in US Veterans in Department of Veterans Affairs Care
|
| |
| |
Reported by Jules Levin
AASLD Nov 1-5 Wash DC
Lisa Backus* MD PhD1,2, Pamela Belperio PharmD1,3, Timothy Loomis PhD1, James Halloran RN1, Steven-Huy Han MD3,4, and Larry Mole PharmD1
1Office of Public Health/Population Health, 2 Veterans Health Administration Palo Alto, 3 Veterans Health Administration Los Angeles, 4Pfleger Liver Institute, David Geffen School of Medicine at UCLA

PROGRAM ABSTRACT: Background: Little information exists about hepatitis B virus (HBV) screening rates and infection prevalence in a national population-based United States cohort. We assessed the extent to which Veterans in Department of Veterans Affairs (VA) care were screened for HBV and estimated the prevalence in this large, racially diverse national population.
Methods: We used VA's Corporate Data Warehouse to identify birth dates, sex, race/ethnicity and VA laboratory tests for all Veterans with at least one VA outpatient visit in 2012. For these individuals, we determined if VA HBV testing occurred at any time from October 1999 through December 2012. We accepted HBV surface antigen (HBsAg), surface antibody (anti-HBs), e antigen (HBeAg), e antibody (anti-HBe), core antibody (anti-HBc), and HBV DNA tests as evidence of "any" HBV screening. "Recommended" screening required a HBsAg and anti-HBs test. Additionally, individuals with a negative anti-HBc test were considered to have met "alternative" screening according to the American Association for the Study of Liver Disease guidelines. For patients who were HBsAg+, suggested supplementary testing included anti-HBe, HBeAg and HBV DNA. We calculated HBV screening rates, supplementary HBV testing rates, and HBV infection prevalence.
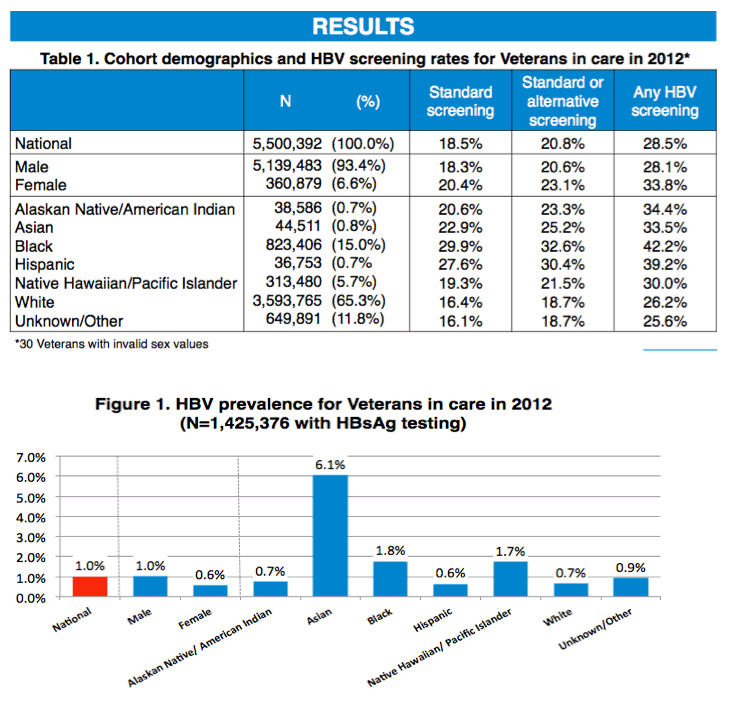
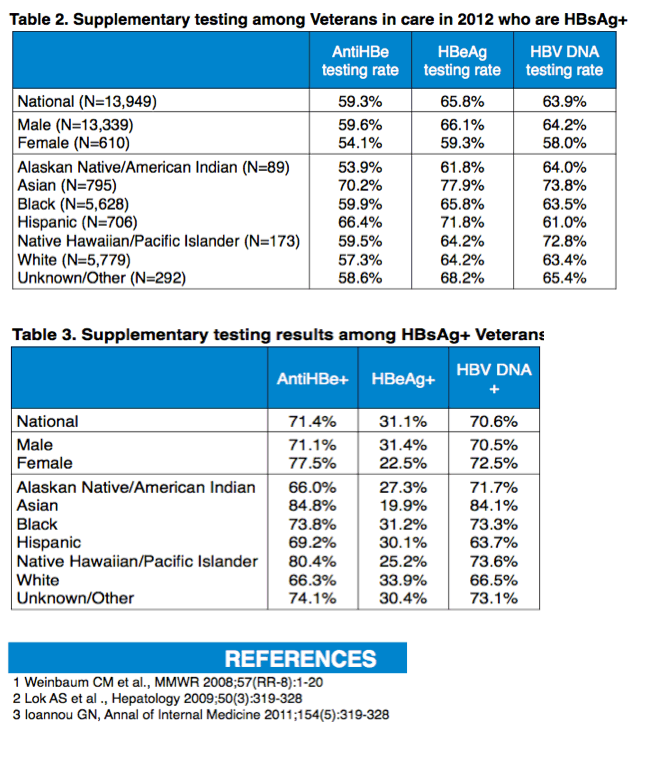
Results: Among 5,500,392 Veterans in care in 2012, 28.5% had VA HBV screening with any HBV-related laboratory test, 18.5% had recommended HBV screening, and an additional 2.3% had alternative screening. In the 1,425,376 Veterans with HBsAg testing, HBV infection prevalence was 1.0%. HBV screening and prevalence by race/ethnicity are shown in the table. Among HBsAg+ individuals (n=13,949), supplementary testing for anti-HBe, HBeAg and HBV DNA occurred in 59.3%, 65.8% and 63.9%, respectively; rates of positivity were 71.4%, 31.1% and 70.6%, respectively.

|
| |
|
 |
 |
|
|