 |
 |
 |
| |
Bidirectional Passage of HIV Between T cells and Renal Epithelial cells through Virological Synapse Formation
|
| |
| |
Reported by Jules Levin
CROI 2013
CROI: The Kidney at CROI 2013 - Christina M. Wyatt, MD Assistant Professor, Medicine/ Nephrology Mount Sinai School of Medicine New York, NY - (03/21/13)
local HIV infection of the kidney has an established role in the pathogenesis of HIV-associated nephropathy (HIVAN), and previous in vitro studies have supported cell-cell transfer of replication-competent virus from T-cells to renal epithelial cells through virologic synapses. In a series of co-culture experiments, Blasi et al (abstract 195) confirmed the ability of T-cells to transfer infectious virus to renal tubular epithelial cells. Co-culture of infected tubular cells with uninfected T-cells further demonstrated transfer of infectious virus to T-cells. These data suggest bidirectional transfer of HIV between T-cells and renal epithelial cells, an important step in establishing the kidney as a potential viral reservoir.
Maria Blasi1 *, Ping Chen2, Benjamin Chen2, Bala Balakumaran1, Donatella Negri1, Andrea Cara1 and Mary Klotman1 1Department of Medicine, Duke University Medical Center, Durham, NC 27710. 2Mount Sinai School of Medicine, New York, New York,USA;* Presenting Author
CONCLUSIONS
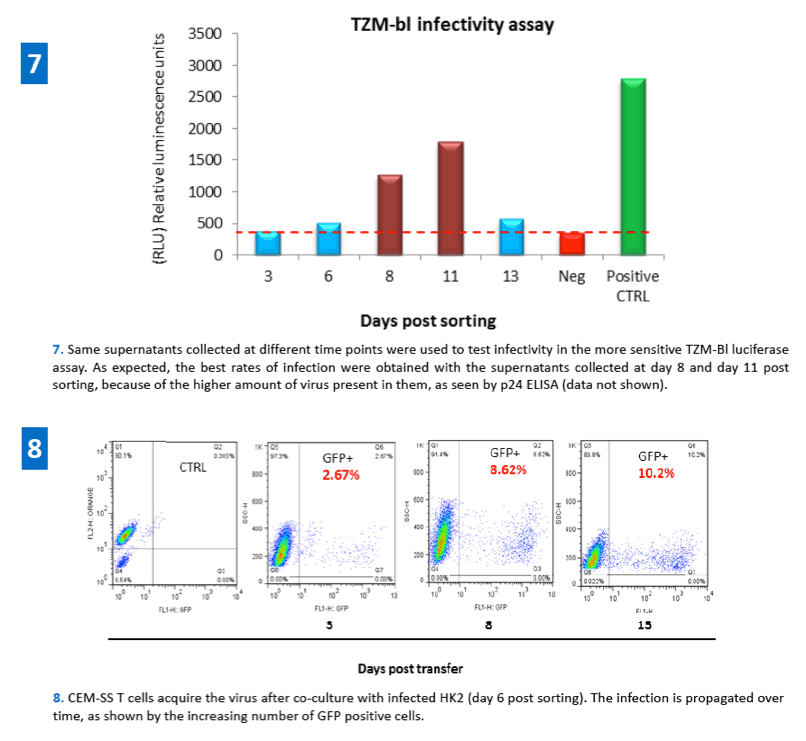
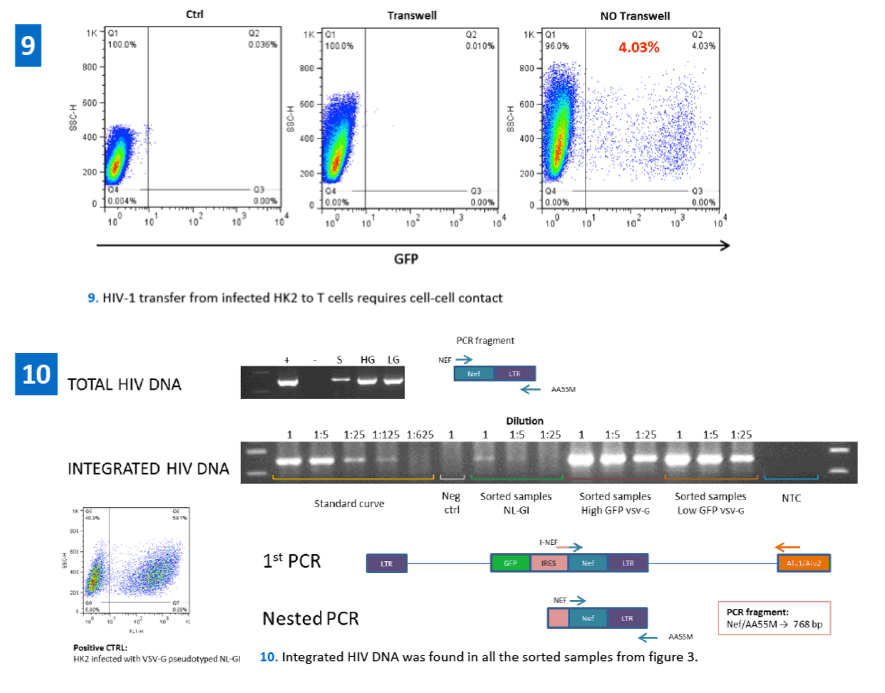
In this work we demonstrated that following cellular interaction between infected T cells and uninfected kidney cells, HIV-1 is efficiently transferred to the epithelial cells. Renal epithelial cells appear to support stable replicative integration of HIV as shown by Alu-PCR, p24 production, TZM-bl infectivity and virus transfer to T cells following cell-cell contact. These results better define the role of HIV-1 infection in kidney-derived cells, underlying the importance of HIV-1 in the kidney and its possible function as a viral reservoir.
BACKGROUND
Direct HIV-1 infection and gene expression in tubular and glomerular epithelial cells leads to the development of HIV-associated nephropathy (HIVAN), ultimately leading to the decreased renal function characteristic of HIVAN patients1. Interstitial infiltrating lymphocytes are often found in HIVAN renal biopsies suggesting a role for T cells in HIV-1 spread within the tissue. Several reports indicated that cell-cell contact can mediate transfer of HIV into recipient cells with a much greater efficiency (100 to 1000 times) than cell-free virus2. Epithelial cells use an atypical mechanism to facilitate HIV-1 entry. In permissive cells, which express CD4 and CXCR4/CCR5 co-receptors, HIV-1 efficiently enters cells using gp-120 mediated membrane fusion. Since renal epithelial cells do not express CD4, HIV-1 entry into those cells is expected to be less efficient than other permissive cells. Heparan sulfate proteoglycans (HSPGs) have been suggested to be alternate receptors for HIV-1 entry into CD4 negative cells, enabling HIV-1 entry into an envelope independent manner. In vitro co-cultivation of HIV-infected T cells with non-infected renal tubular epithelial cells results in virus transfer to the renal epithelial cells, while cell free virus infection of these cells is very inefficient3.
AIM
The aim of this study was to better understand HIV-1 infection cycle following transfer to epithelial cells.
METHODS
·Infectious virus was produced in 293T cells by transfection
·T cell lines were infected with MOI 1 of free virus and GFP expression was monitored by flow citometry
·HIV-1 transfer from infected T cells (CEM) to epithelial cells (HK2) was carried out in a co-culture system and evaluated by FACS analysis. Infected T cells were added in a 4:1 ratio to the dye labeled (CMTMR, Invitrogen) target epithelial cells and co-cultured overnight.
·48h post co-culture GFP expressing renal epithelial cells were sorted out by flow cytometry and re-plated. Supernatants from these cells were collected at different time points in order to evaluate virus production (p24 ELISA) and infectivity (TZM-bl luciferase activity assay).
·HIV-1 infected renal epithelial cells were also used as donor cells in a co-culture system to evaluate their ability to transfer the virus back to uninfected T cells.
·Genomic DNA was extracted from HIV-1 infected renal epithelial cells in order to evaluate HIV-1 integration through Alu-PCR.
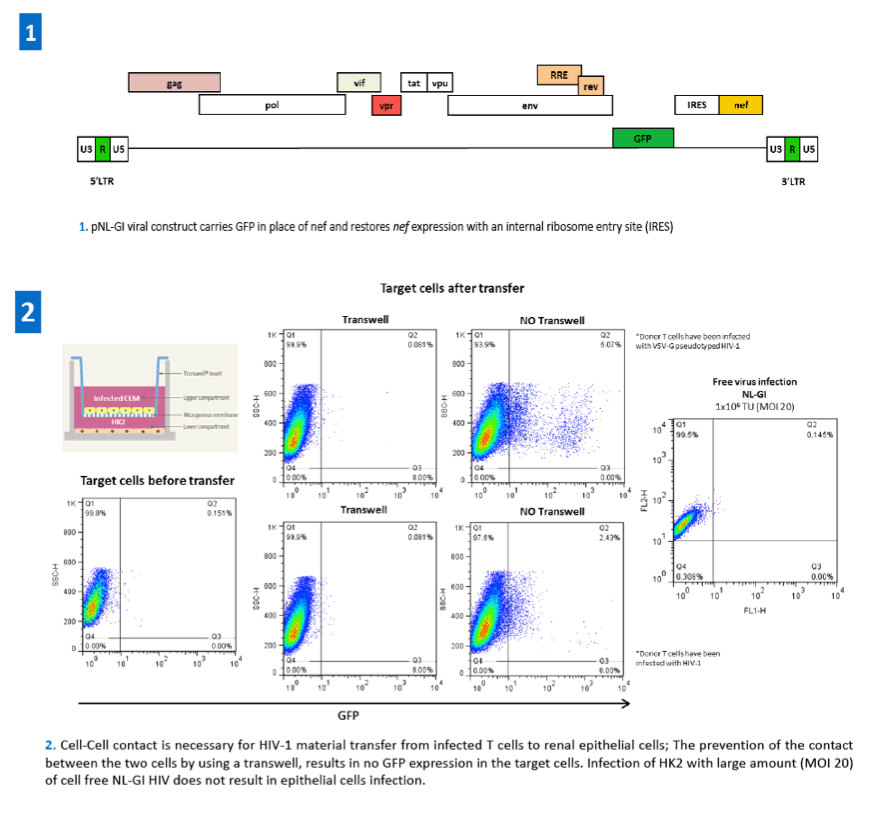
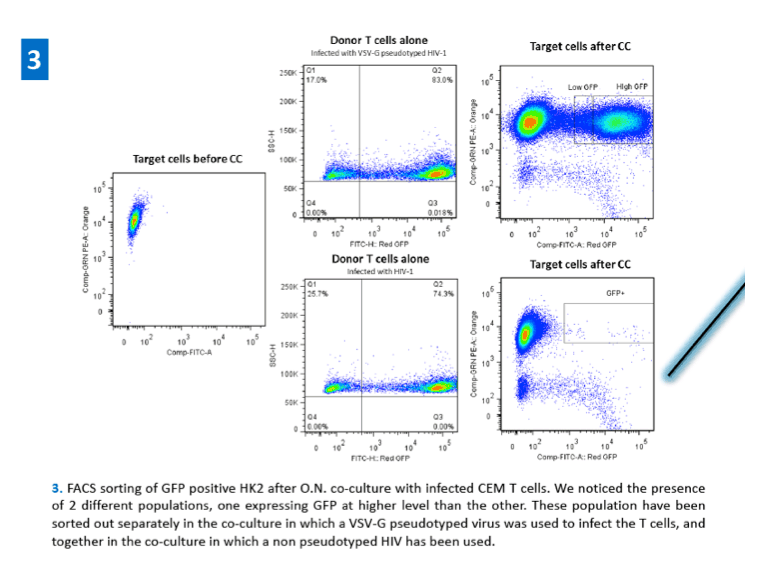
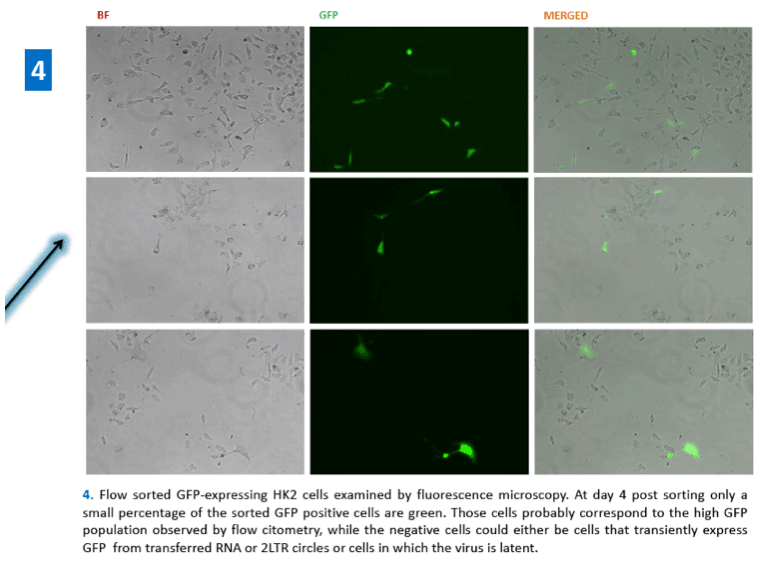
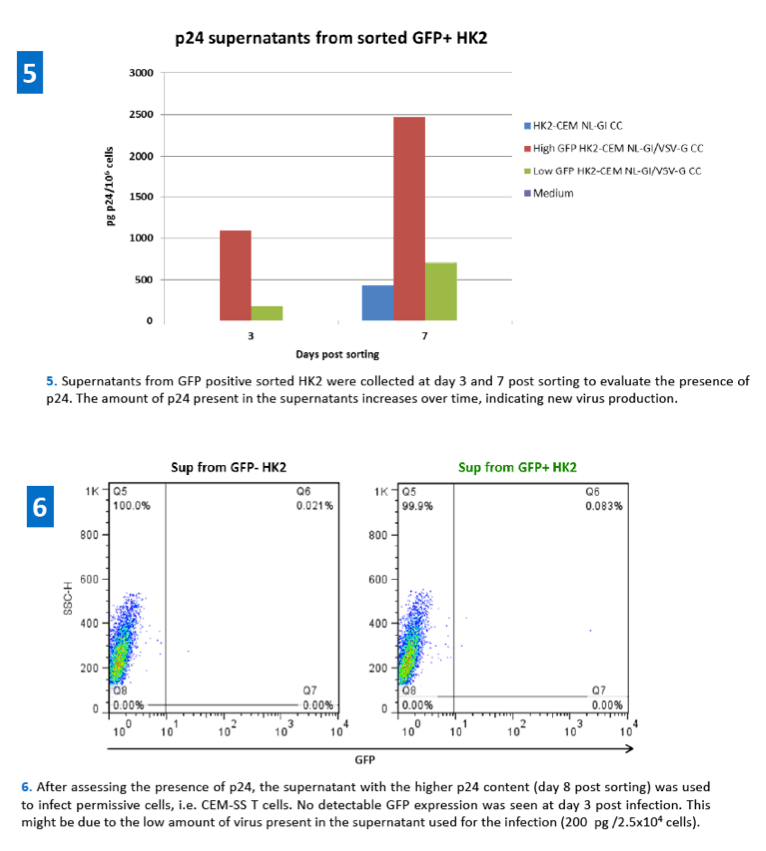
References:
1.Wyatt CM, Rosenstiel PE, Klotman PE. HIV-associated nephropathy. Contrib Nephrol. 2008;159:151-61
2.Dale BM, Alvarez RA, Chen BK. Mechanisms of enhanced HIV spread through T-cell virological synapses. Immunol Rev. 2013 Jan;251(1):113-24.
3.Chen P, Chen BK, Mosoian A, Hays T, Ross MJ, Klotman PE, Klotman ME. Virological synapses allow HIV-1 uptake and gene expression in renal tubular epithelial cells. J Am Soc Nephrol. 2011 Mar;22(3):496-507.
|
| |
|
 |
 |
|
|