 |
 |
 |
| |
Factors Associated with Tenofovir-related Adverse Events and Drug Exposure in HIV-positive Patients
|
| |
| |
Reported by Jules Levin
CROI 2013
Cristina Gervasoni1, Paola Meraviglia1, Simona Landonio1, Sara Baldelli2, Serena Fucile2, Laura Castagnoli1, Emilio Clementi2, Massimo Galli1, Agostino Riva1, Dario Cattaneo2
1Department of Infectious Diseases, Luigi Sacco University Hospital, Milan, Italy; 2Unit of Clinical Pharmacology, Luigi Sacco University Hospital, Milan, Italy
Renal Impairment in Patients Receiving a Tenofovir-cART Regimen: Impact of Tenofovir Trough Concentration
JAIDS Journal of Acquired Immune Deficiency Syndromes:
1 April 201
http://www.natap.org/2013/HIV/022713_01.htm

Background
Although tenofovir is relatively well tolerated as first-line treatment of HIV, episodes of kidney and bone diseases have been consistently reported in patients on long-term tenofovir exposure. Factors associated with tenofovir-related drug exposure and adverse events are, however, poorly characterized.
Methods
One-hundred HIV-positive patients with basal creatinine clearance >80 mL/min treated with tenofovir for at least 6 months and with at least one assessment of tenofovir plasma trough concentrations were considered. Tenofovir-associated adverse events were defined as the appearance of pathologic proteinuria, worsening of renal function or bone toxicity. Tenofovir concentrations were measured by LC-MS/MS.
Results
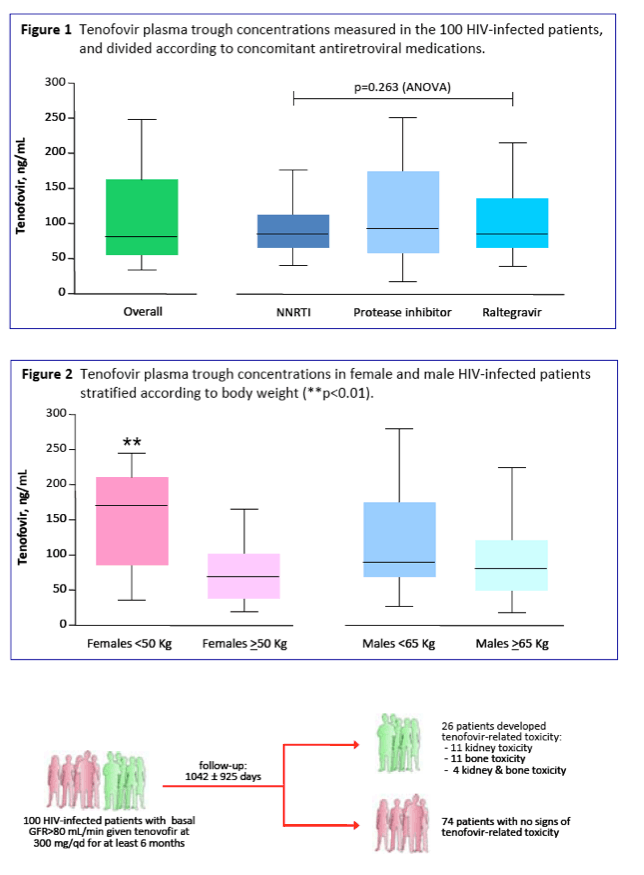
A wide distribution in the tenofovir plasma concentrations was observed, ranging from 20 to 452 ng/mL.
By multivariate regression analysis, the factors independently associated with plasma tenofovir concentrations were serum creatinine (p=0.003) and body weight (p=0.002).
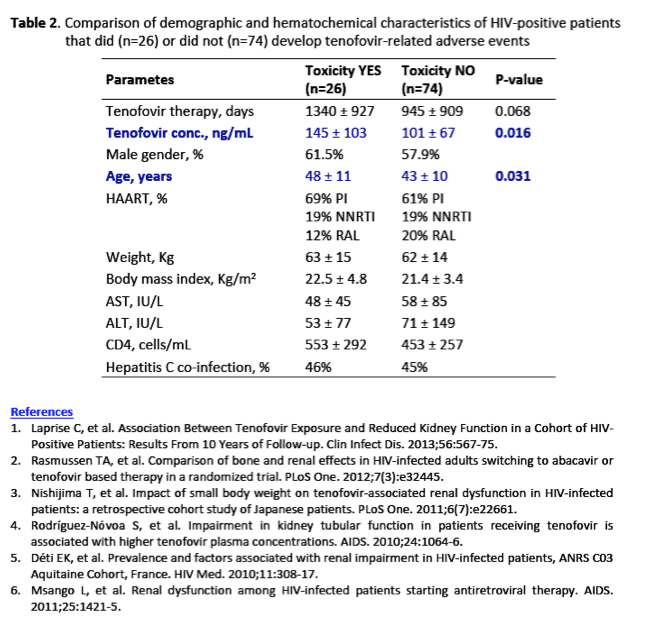
Notably, women with body weight<50 kg had significantly higher plasma tenofovir concentrations than those weighting >50 Kg (160±93 vs.71±52 ng/mL, p<0.001).
No impact of age or concomitant antiretroviral medication on tenofovir concentrations was found.
Twenty-six out of the 100 HIV-positive patients experienced kidney (n=11), bone diseases (n=11) or both complications (n=4).
Patients experiencing tenofovir-related toxicity had significantly higher plasma drug concentrations (145±103 vs. 101±67 ng/mL, p=0.016), were significantly older than those with no tenofovir- related toxicity (48±11 vs. 43±10 years, p=0.031).
The independent association between tenofovir plasma trough concentrations and drug-related toxicity was confirmed by multivariate logistic regression analysis (p=0.008).
Thirty-two percent and 18% of women with body weight < or >50 Kg, respectively, experienced tenofovir-related toxicity.
Women experiencing the events had tenofovir plasma concentrations significantly higher than those with no event (173±131 vs. 106±65 ng/mL, p=0.042).
Conclusions
HIV-positive patients (mainly women) with low body weight are at risk to be exposed to high tenofovir plasma trough concentrations, ultimately resulting in a significant hazard to develop drug-related complications. This could be eventually handled by routine therapeutic monitoring of tenofovir plasma concentrations.

|
| |
|
 |
 |
|
|