 |
 |
 |
| |
Pharmacokinetic study of raltegravir in HIV-infected patients with end stage liver disease:
LIVERAL ANRS 148 study
|
| |
| |
Reported by jules Levin
CROI 2013
Caroline Barau1, Josephine Braun2, Corine Vincent2, Stephanie Haïm-Boukobza3, Jean-Michel Molina4, Patrick Miailhes5, Jean-Pierre Aboulker2, Jean-Charles Duclos-Vallee6, Anne-Marie Taburet1, Elina Teicher1 and the ANRS 148 Study group
1AP-HP, Bicetre Hospital, Kremlin-Bicetre ~ 2INSERM SC10-US019, Villejuif ~ 3AP-HP, INSERM U785, University of Paris XI, Paul Brousse Hospital, Villejuif ~ 4AP-HP, University of Paris VII Denis Diderot, Saint-Louis Hospital, Paris
5Hospices Civils de Lyon, INSERM U1052, Croix-Rousse Hospital, Lyon ~ 6AP-HP, Centre Hepato

ABSTRACT
Background: Raltegravir (RAL), metabolized via UDP glucuronosyltransferase (UGT1A1), takes part to antiretroviral therapy. Because of its good tolerance, studying its relevance in
the context of severe liver disease is crucial. The aim of this study was to evaluate pharmacokinetic parameters of RAL in HIV infected patients with end stage liver disease (ESLD).
Methods: All patients included were virologically controlled (HIV-RNA <50 cps/mL) under antiretroviral therapy (ART) for at least 6 months and were eligible for liver transplantation.
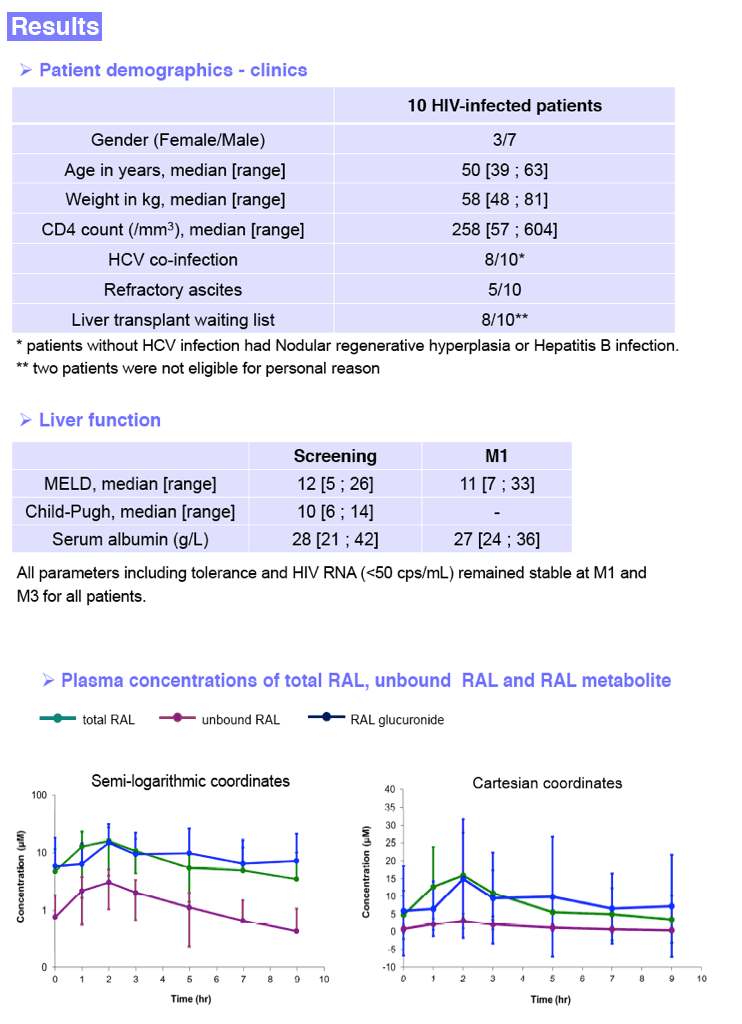
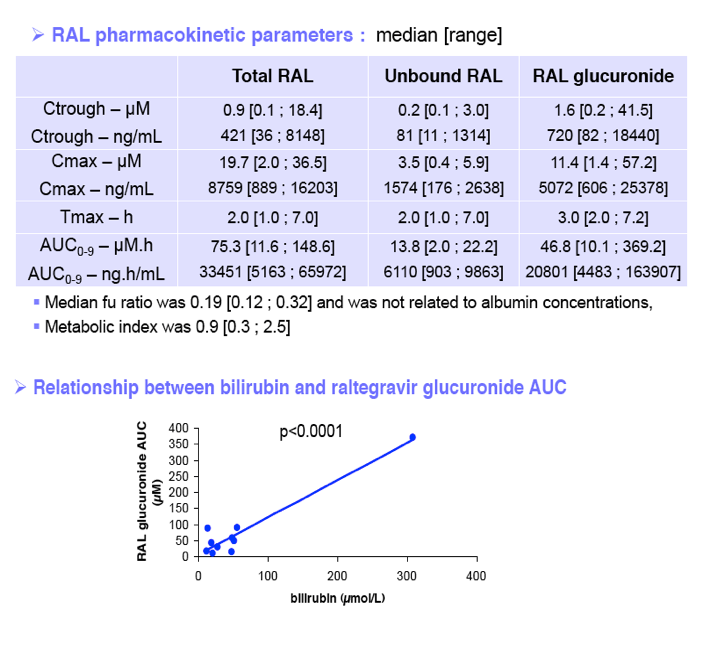
ART was switched to raltegravir 400 mg bid plus a once-a-day fixed dose tenofovir/emtricitabine or abacavir/lamivudine combination. Liver function assessed by MELD and Child-Pugh (CP) scores and immuno-virological parameters were checked at screening and up to M3 after RAL initiation. Serial blood samples were drawn at M1 after RAL initiation over a dosing interval up to 9 hours post RAL intake. Patients had to fast up to 1hr post RAL intake. Plasma concentration of total RAL, protein unbound RAL (RALu) and inactive RAL glucuronide (RALg) were measured by LC/MS/MS. RAL unbound fraction (fu) is the ratio of RALu to RAL. Pharmacokinetic (PK) parameters were analyzed by a model independent method. Metabolic ratio (MR) was calculated from the AUC ratio of RALg to RAL. Results are presented as median [range].
Results: 10 patients (70% males, 80% VHC+) were included in this study: age: 50 [39; 63] years, weight: 58 [48; 81] kg, CD4: 258 [57; 604] cells/mm3. MELD and CP scores at screening were 12 [5; 26] and 10 [6; 14], respectively. Median MELD at M1 was 11 [7; 33]. Albumin concentration at M1 was 27 [24; 36] g/L. All parameters including tolerance and HIV RNA (<50 cps/mL) remained stable at M1 and M3. RAL PK parameters were measured as follows.
Fu ratio: 0.19 [0.12; 0.32] was not related to albumin concentrations. MR ratio was 0.9 [0.3; 2.5]. All the parameters analyzed did not differ from those concerning patients without liver disorders.
Conclusions: RAL was well tolerated in HIV-infected patients with ESLD. There was a huge variability of raltegravir pharmacokinetic parameters which remained however in the ranges reported in patients or volunteers with normal liver functions.

|
| |
|
 |
 |
|
|