 |
 |
 |
| |
Maraviroc plus Raltegravir dual Therapy in Aviremic HIV infected Patients with Lipodystrophy : Results from the ROCnRAL ANRS 157 Study.
|
| |
| |
Reported by Jules Levin
CROI 2013
Christine Katlama1,2, Lambert Assoumou2, Marc-Antoine Valantin1,2, Claudine Duvivier3,4, Cathia Soulie1,2, Laetitia Chablais2, Gilles Pialoux5, Patrick Mercie6, Gilles Peytavin7, Anne-Genevieve Marcelin1,2.

ABSTRACT
Background: Novel NRTI and PI sparing strategies are needed in long term treated patients with lipodystrophy. Given their potency, excellent metabolic profile, lack of drug interactions maraviroc (MVC) and raltegravir (RAL) appear as good candidates for such approach.
Methods: RocNral, a single-arm study had enrolled patients with plasma HIV RNA < 50cp/ml, clinical lipodystrophy, CD4 cells nadir > 100 cells/mm3, a subtype B or CRF02 R5 tropic virus who were switched from a suppressive ART to MVC 300 mg bid plus RAL 400mg bid. Viral tropism was assessed on HIV DNA using Geno2pheno test with a 20% false positive rate. Primary endpoint was the proportion of patients with treatment (Rx) failure at W24 (ITT analysis), defined as either virological failure (VF) with 2 consecutive plasma HIV RNA > 50 cp/ml or Rx discontinuation. Patients were monitored monthly up to W24 and then every 2 months until W48. Secondary endpoints included tolerability, drug concentration, changes in fat tissue, bone density, metabolic parameters at W48. To ensure at least 80% efficacy rate of the MVC/RAL strategy, a maximum of 10 strategies failures was allowed for 90 patients enrolled.
Results:A total of 44 patients were enrolled from Dec 2011 to Jan 2012. Baseline characteristics (median) were: 86% male, age 55 years (IQR, 50-60), nadir CD4 210 cells/mm3 (IQR, 150-276), ART duration 15 years (IQR, 15-19), duration of HIV RNA suppression 5.2 years (IQR, 4.4-7.9).
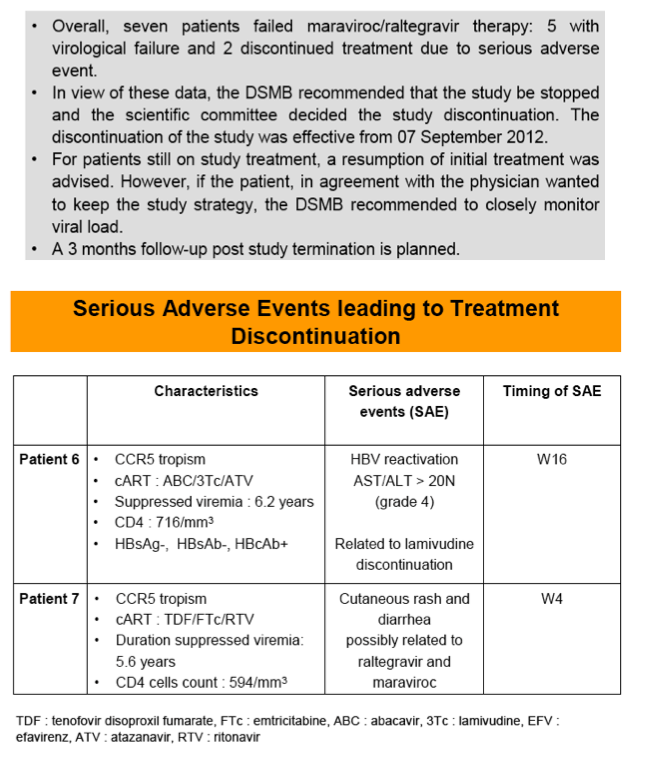
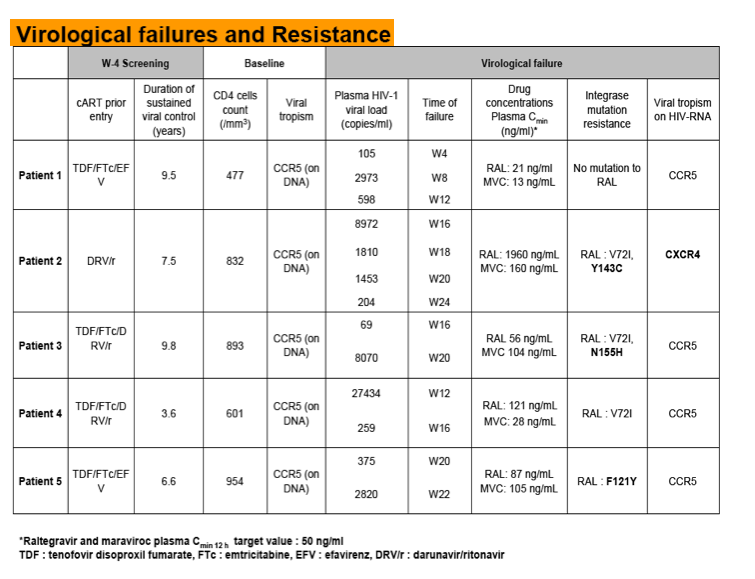
Seven patients failed MVC/RAL therapy: 5 with VF and 2 discontinued Rx due to SAE (HBV rebound in a patient HBcAb+ and HBsAg-, one hypersensitivity syndrome). Resistance mutations emerged in 3/5 VF patients. Considering these data, DSMB approved premature study discontinuation on 28 August 2012.
Conclusion: This pilot study suggests that, in this population of long term treated patients; MVC/RAL dual therapy lacks virological robustness resulting in a high rate of resistance mutations. Several hypotheses are under investigation to understand these results such as lack of sensitivity of tropism test to detect minority variants defect in tropism test or drugs interaction.


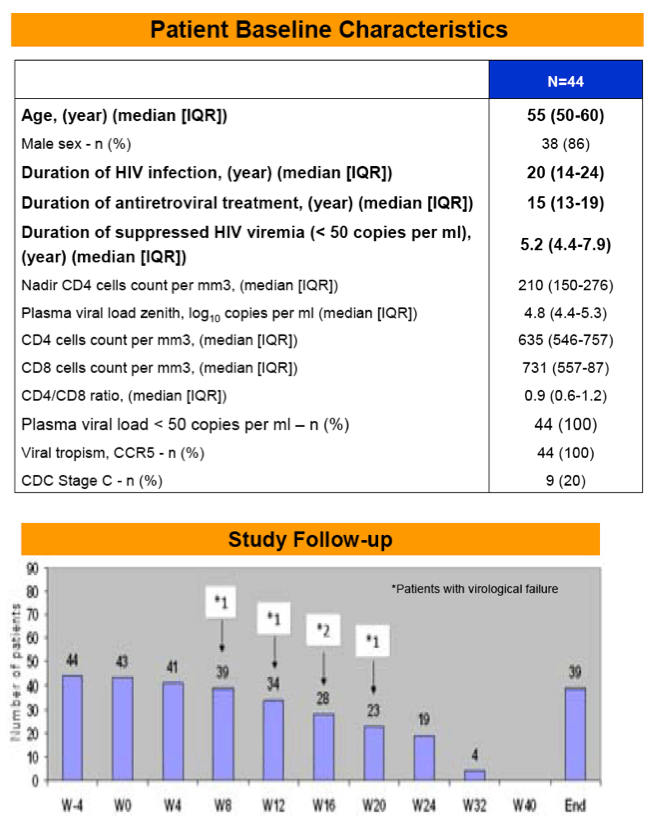
|
| |
|
 |
 |
|
|