 |
 |
 |
| |
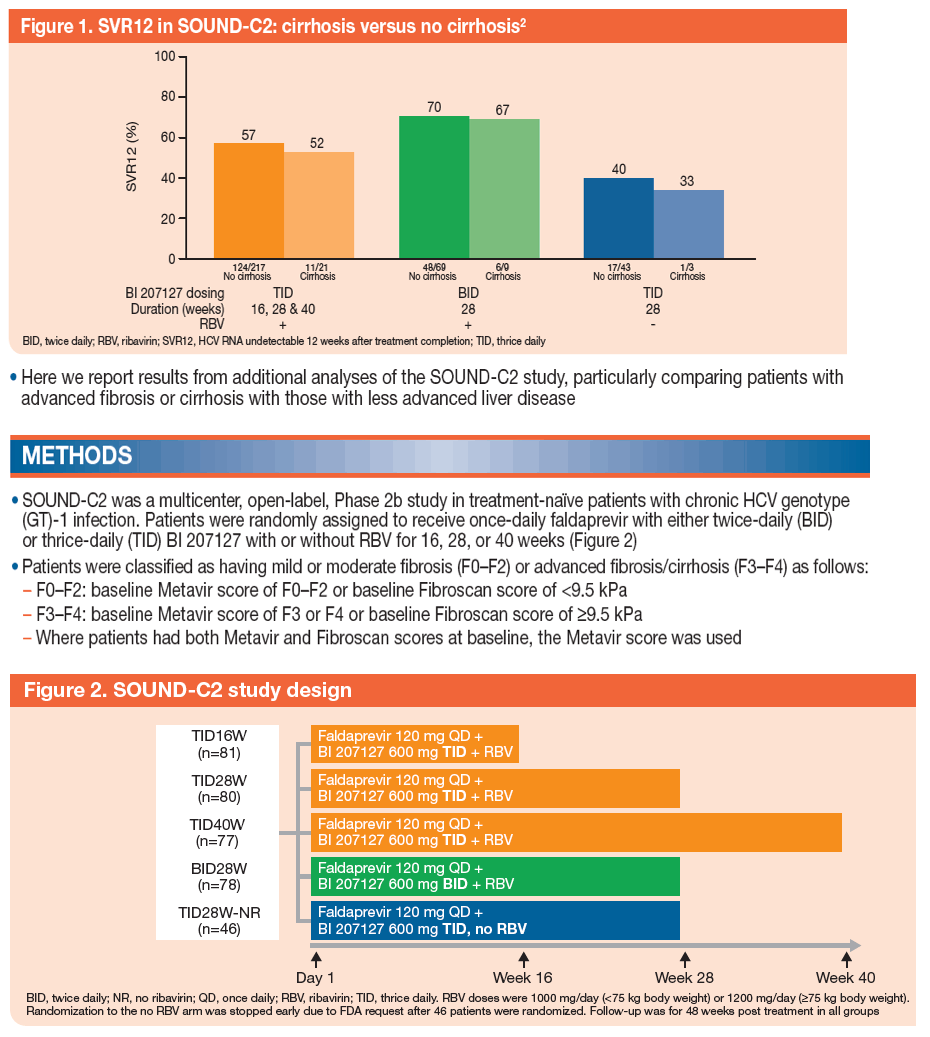
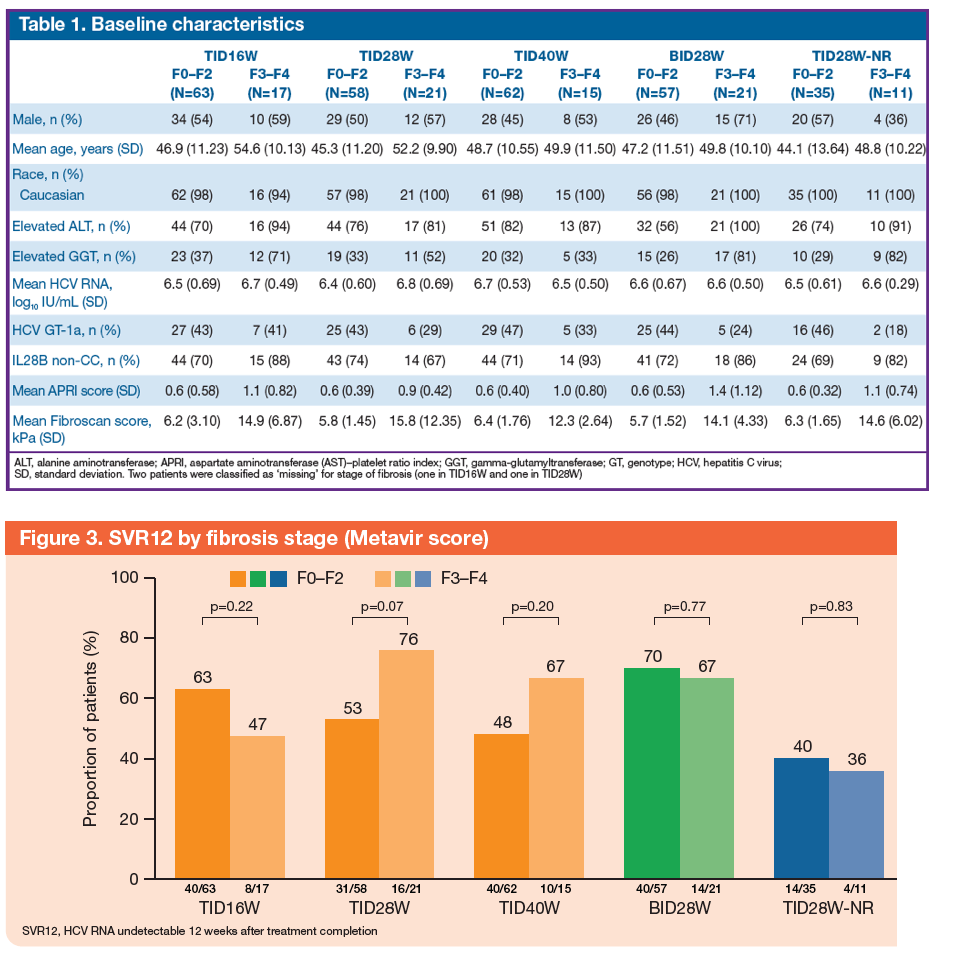
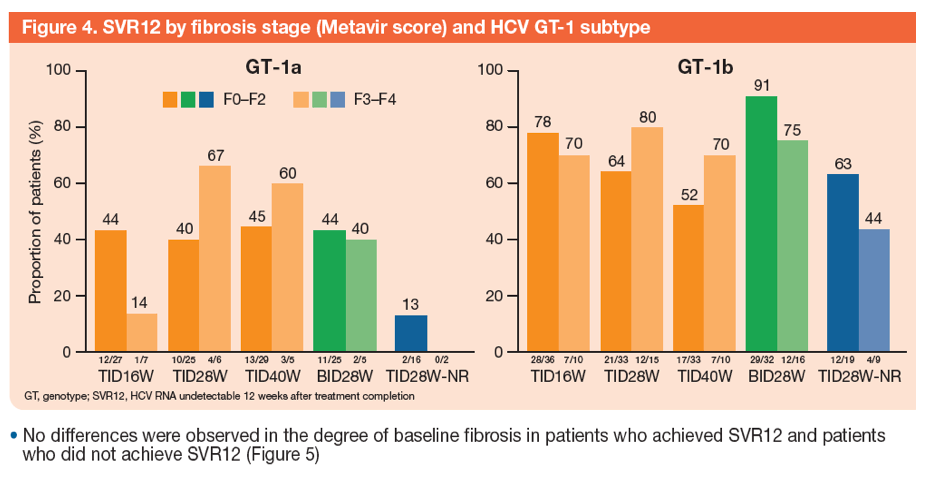
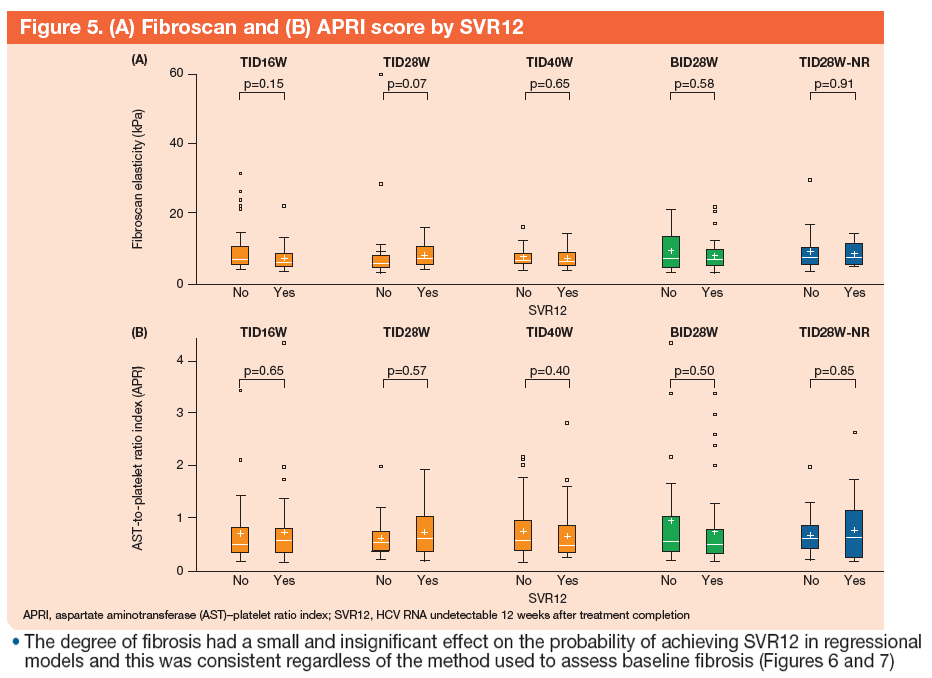
An Analysis of Response Rates by Fibrosis Stage in Patients Treated With Faldaprevir, BI 207127, and Ribavirin in the SOUND-C2 Study
|
| |
| |
Reported by Jules Levin
Digestive Disease Week (DDW), May 18-21, 2013, Orlando, FL
Boehringer announced just before EASL a deal with Presidio to conduct a phase 2a collaborative trial to evaluate Presidio's pan-genotypic HCV NS5A inhibitor (PPI-668) in combination with Boehringer Ingelheim's HCV protease inhibitor faldaprevir (BI201335) and its non-nucleoside HCV polymerase inhibitor (BI207127), with or without ribavirin. At EASL Phase 3 Results were reported for Faldaprevir+Peg/Rbv, soon BI will submit a New Drug Application to the FDA for approval. At CROI BI reported coinfection early virologic response study results & ART PK data showing favorable results, which is expected to be submitted to the FDA for a coinfection indication.
Study Results For PPI668 NS5A, BI335 protease, BI127 non-nuc polymerase - new collaboration between Presidio & Boerhinger Ingelheim.......http://www.natap.org/2013/HCV/031513_01.htm
EASL: FALDAPREVIR PLUS PEGYLATED INTERFERON ALFA-2A AND RIBAVIRIN IN CHRONIC HCV GENOTYPE-1 TREATMENT-NAIVE PATIENTS - (04/27/13)
CROI: STARTVerso 4: High rates of early virologic response in HCV genotype 1/HIV-co-infected patients treated with faldaprevir plus pegIFN and RBV - (03/04/13)
CROI: Pharmacokinetic interactions of darunavir/ritonavir, efavirenz, and tenofovir with the HCV protease inhibitor faldaprevir in healthy volunteers - (03/04/13)
CROI: Boerhinger Ingelheim Announes Interim Results Evaluating Virologic Response Rates in HCV/HIV Coinfected Patients Treated with HCV Protease BI201335, and Drug Drug Interaction Studies with HIV ARTs - (03/04/13)
Tarik Asselah,1 Stefan Zeuzem,2 Vicente Soriano,3 Jean-Pierre Bronowicki,4 Ansgar W Lohse,5 Beat Mullhaupt,6 Marcus Schuchmann,7 Marc Bourliere,8 Maria Buti,9 Stuart Roberts,10 Ed Gane,11 Jerry O Stern,12 John-Paul Gallivan,13 Florian Voss,12 Wulf Böcher,13 Federico J Mensa12
1Hopital Beaujon, Clichy, France; 2Klinikum der J. W. Goethe-Universität, Frankfurt Am Main, Germany; 3Department of Infectious Diseases, Hospital Carlos III, Madrid, Spain; 4Hopital de Brabois, Vandoeuvre, France; 5University Hospital Hamburg-Eppendorf, Hamburg, Germany; 6University Hospital of Zurich, Zurich, Switzerland; 7University Hospital Mainz, Mainz, Germany;
8Hopital Saint Joseph, Marseille, France; 9Hospital Vall d'hebron and Ciberehd, Barcelona, Spain; 10Alfred Hospital, Department of Gastroenterology, Melbourne, Australia; 11Auckland Clinical Studies, Auckland, New Zealand; 12Boehringer Ingelheim Pharmaceuticals, Ridgefield, CT, USA; 13Boehringer Ingelheim Pharma GmbH & Co KG, Ingelheim, Germany


|
| |
|
 |
 |
|
|