 |
 |
 |
| |
Once-Daily Raltegravir for 48 Weeks as Maintenance Therapy in Paris
|
| |
| |
14th European AIDS Conference. October 16-19, 2013. Brussels
Mark Mascolini
Sixty-eight of 71 people who switched to once-daily raltegravir with a viral load below 50 copies maintained virologic suppression for 48 weeks in an observational study at a Paris hospital [1]. Virologic failure occurred in 3 people taking nucleos(t)ides with raltegravir who had a previous nucleos(t)ide failure.
Raltegravir is licensed at a dose of 400 mg twice daily, but its long half-life and long binding to the HIV integration complex suggest it may be effective in a once-daily dose for some people. The double-blind QDMRK trial determined that once-daily raltegravir was not noninferior to twice-daily raltegravir in previously untreated people when both were given with tenofovir/emtricitabine [2]. "Despite high response rates with both regimens," the QDMRK investigators concluded, "once-daily raltegravir cannot be recommended in place of twice-daily dosing." But a team at the Hopital Pitie-Salpetriere in Paris noted that once-daily raltegravir was noninferior to twice-daily raltegravir among QDMRK patients with a pretreatment viral load below 100,000 copies.
The ODIS trial in Spain enrolled patients with a viral load below 50 copies for more than 24 weeks on a protease inhibitor regimen and randomized them to once- or twice-daily raltegravir [3]. These investigators found a trend toward a greater risk of virologic failure in 24 weeks with once-daily raltegravir than twice-daily raltegravir in people with nucleoside-associated resistance mutations when they started raltegravir, but only 1 of 148 people without nucleoside resistance had virologic failure on once-daily raltegravir.
The Paris team conducted their observational study in adults with a viral load below 50 copies at least twice in the past 6 months while taking a stable antiretroviral regimen. No one had taken an integrase inhibitor before or had taken raltegravir at the standard dose of 400 mg twice daily without virologic failure. The investigators defined virologic failure as a viral load above 400 copies or two viral loads above 50 copies at any point in the study.
Of the 71 study participants, 47 (66%) were men and age averaged 46 (range 26 to 73). They had taken antiretrovirals for an average 14 years (range 1 to 22) and had tried an average 5 regimens (range 1 to 15). Viral suppression when switching to once-daily raltegravir averaged 62 months (range 6 to 179). Seventeen people (24%) had taken twice-daily raltegravir for an average 8 months.
Most people took once-daily raltegravir with a nucleos(t)ide backbone, either tenofovir/emtricitabine (TDF/FTC) (56%) or abacavir/lamivudine (ABC/3TC) (18%). The others took once-daily raltegravir with etravirine, nevirapine, efavirenz, atazanavir/ritonavir, or unboosted atazanavir. Four people switched to once-daily raltegravir to simplify their regimen, 52 switched because of toxicity, and 15 switched for unreported reasons.
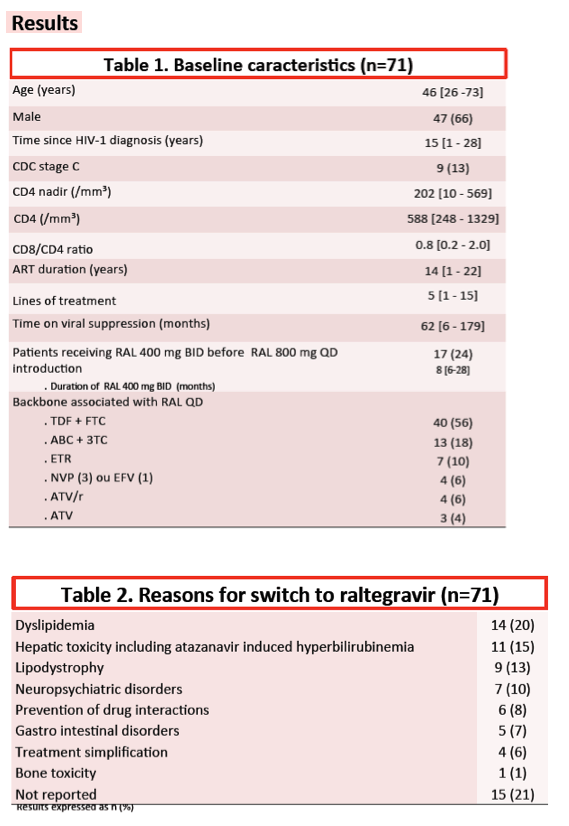
After 24 weeks of once-daily raltegravir, 70 of 71 people (99%) maintained a viral load below 50 copies. In the following 24 weeks 2 more people had virologic failure to bring the virologic success rate of 96%. All 3 people with virologic failure took raltegravir with two nucleos(t)ides, 2 taking TDF/FTC and 1 taking ABC/3TC.
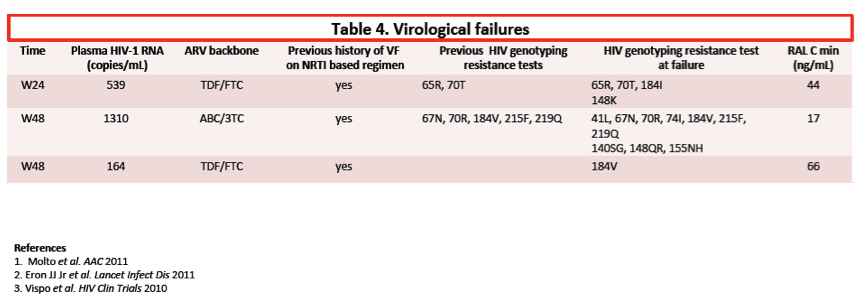
Viral loads at failure in these 3 people were 164, 539, and 1310 copies. All 3 people had a previous virologic failure while taking two nucleos(t)ides. Two people with previous genotyping available had standard nucleos(t)ide mutations at failure. Genotyping at failure of once-daily raltegravir revealed the same mutations plus others, including integrase mutations. One person taking raltegravir with TDF/FTC and without a previously recorded genotype had only the M184V mutation detectable upon failure of the raltegravir regimen.
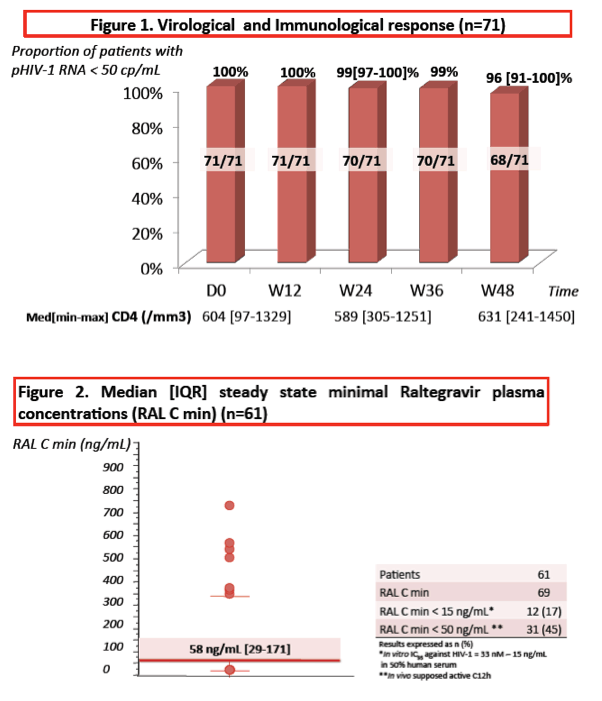
Median steady-state raltegravir minimum concentration measured 58 ng/mL (interquartile range 29 to 171) in 61 people tested. Of 69 minimum concentrations recorded, 12 (17%) were below 15 ng/mL (the 95% inhibitory concentration) and 31 (45%) were below 50 ng/mL (the proposed cutoff for activity in vivo). In the 3 people with virologic failure, raltegravir minimum concentrations were 17, 44, and 66 ng/mL.
The Paris team proposed that switching virologically suppressed people to once-daily raltegravir is effective through 48 weeks as long as raltegravir is taken "with a fully active backbone regimen." Switching patients to either once- or twice-daily raltegravir, they cautioned, requires "access to the entire antiretroviral history and all previous HIV genotyping test results." Even with these precautions, the high proportion of people with low raltegravir minimum concentrations may worry some clinicians.
References
1. Caby F, Bonmarchand M, Soulie C, et al. Efficacy of raltegravir once daily in switching strategies in HIV-1 infected patients with suppressed viraemia. 14th European AIDS Conference. October 16-19, 2013. Brussels. Abstract BPD1/7.
2. Eron JJ Jr, Rockstroh JK, Reynes J, et al. Raltegravir once daily or twice daily in previously untreated patients with HIV-1: a randomised, active-controlled, phase 3 non-inferiority trial. Lancet Infect Dis. 2011;11:907-915. Erratum in Lancet Infect Dis. 2011;11:895. Dosage error in article text.
3. Vispo E, Barreiro P, Maida I, et al. Simplification from protease inhibitors to once- or twice-daily raltegravir: the ODIS trial. HIV Clin Trials. 2010;11:197-204.
------------------------------------



|
| |
|
 |
 |
|
|