 |
 |
 |
| |
Treatment of Chronic Hepatitis C: The Revolution!
|
| |
| |
Reported by Jules Levin
EACs - European AIDS Conference - Oct 16-19 Brussels, Belgium
Heiner Wedemeyer
Hannover Medical School
Germany
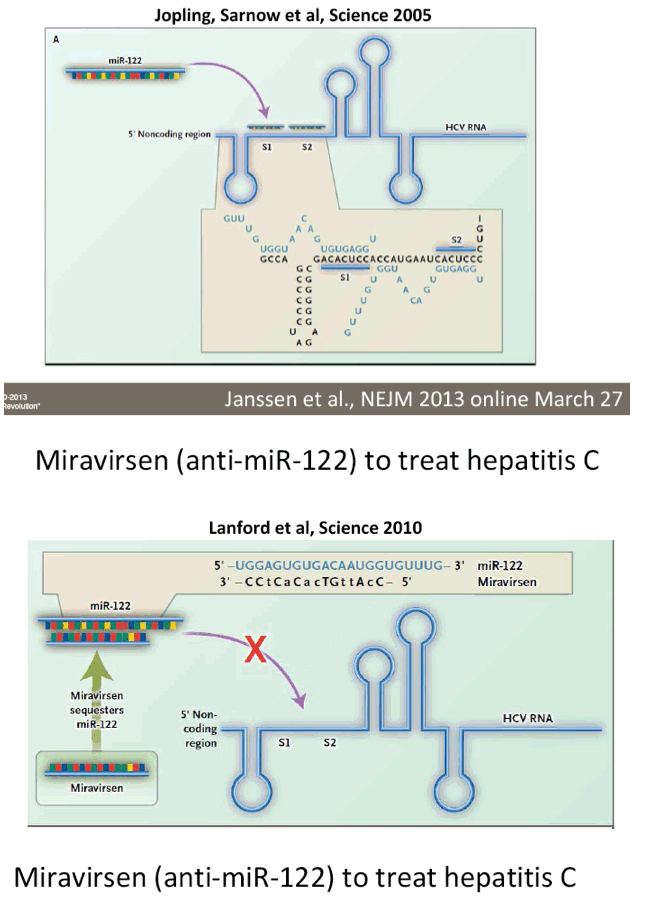
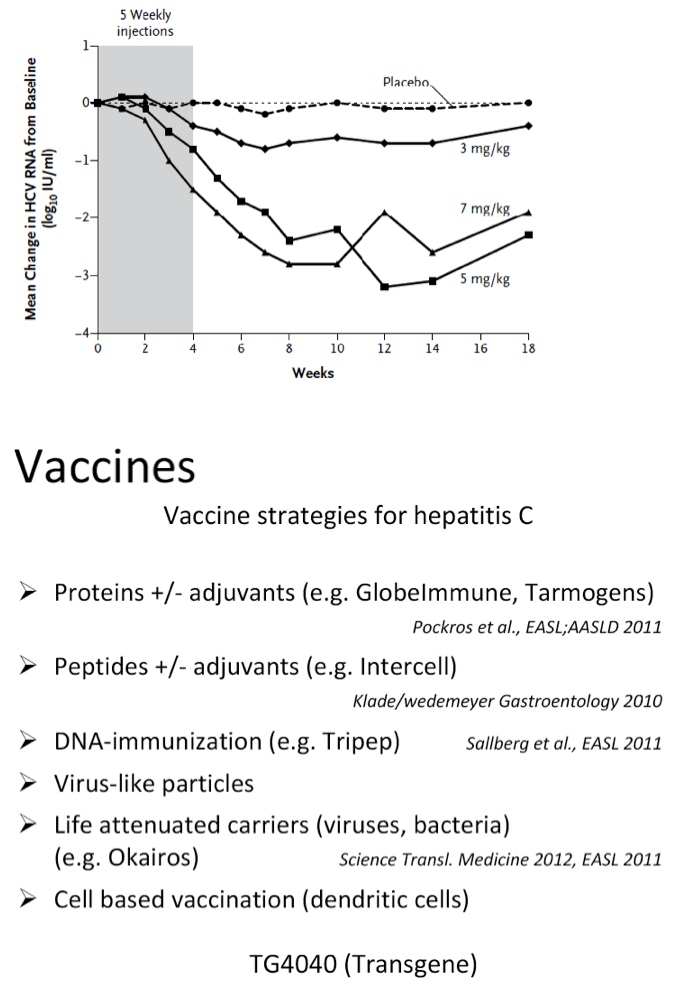
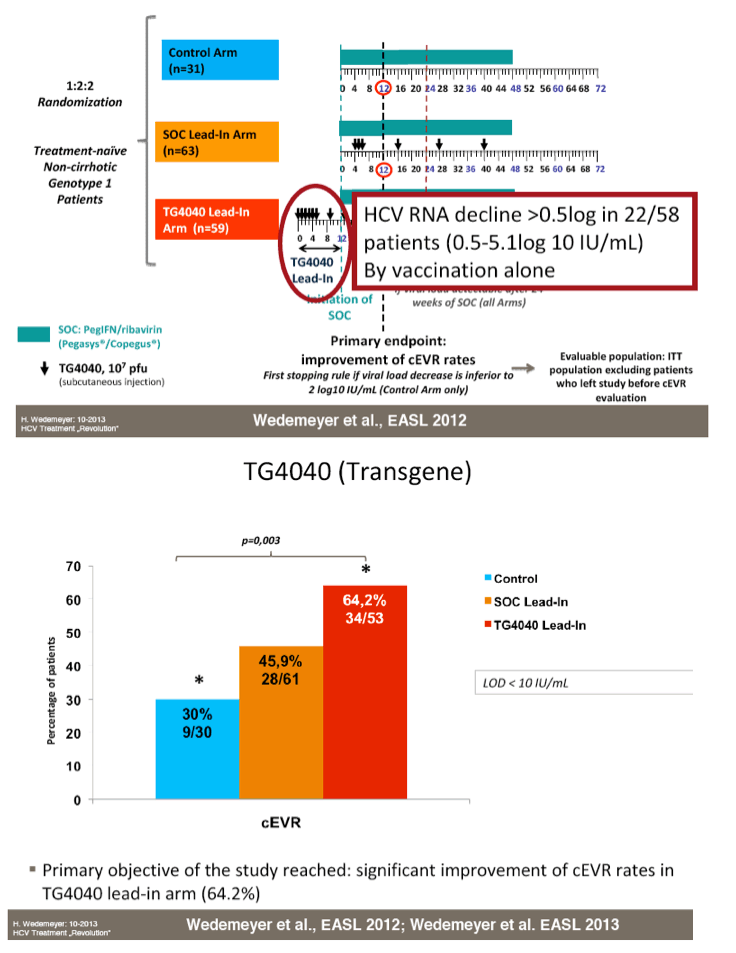
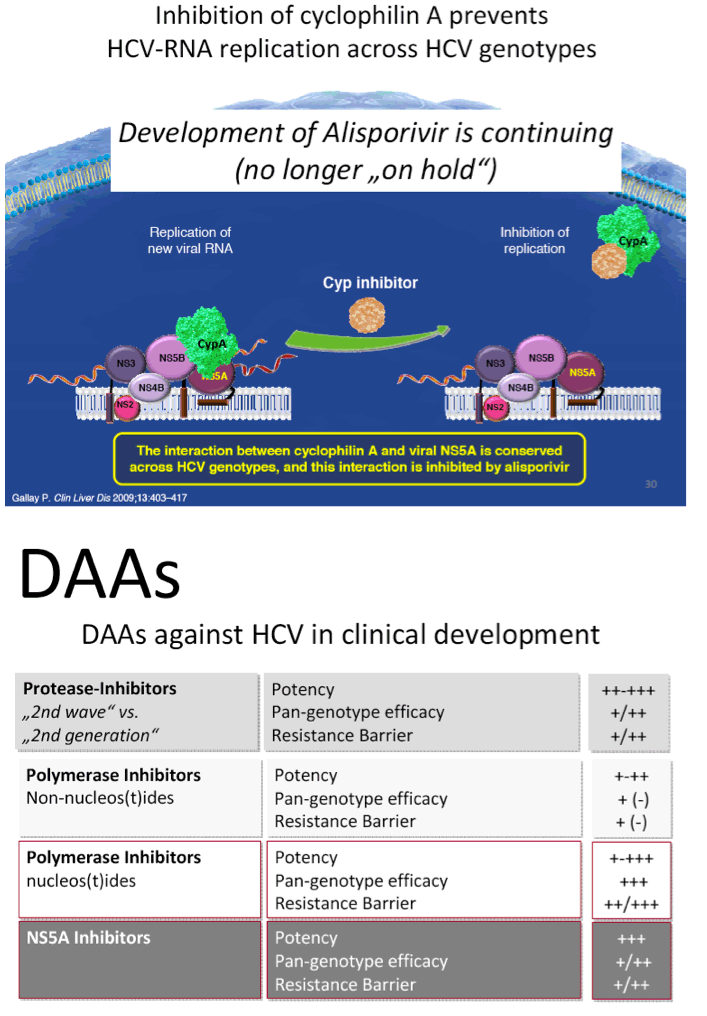
from Jules: here is Dr Wedemeyer's slide presentation where he does a long review including vaccines, immune-based new therapies including Alisporivir, DAAs, direct acting activirals, new oral drugs, and reviews many of the reported studies from phase 2 & 3.
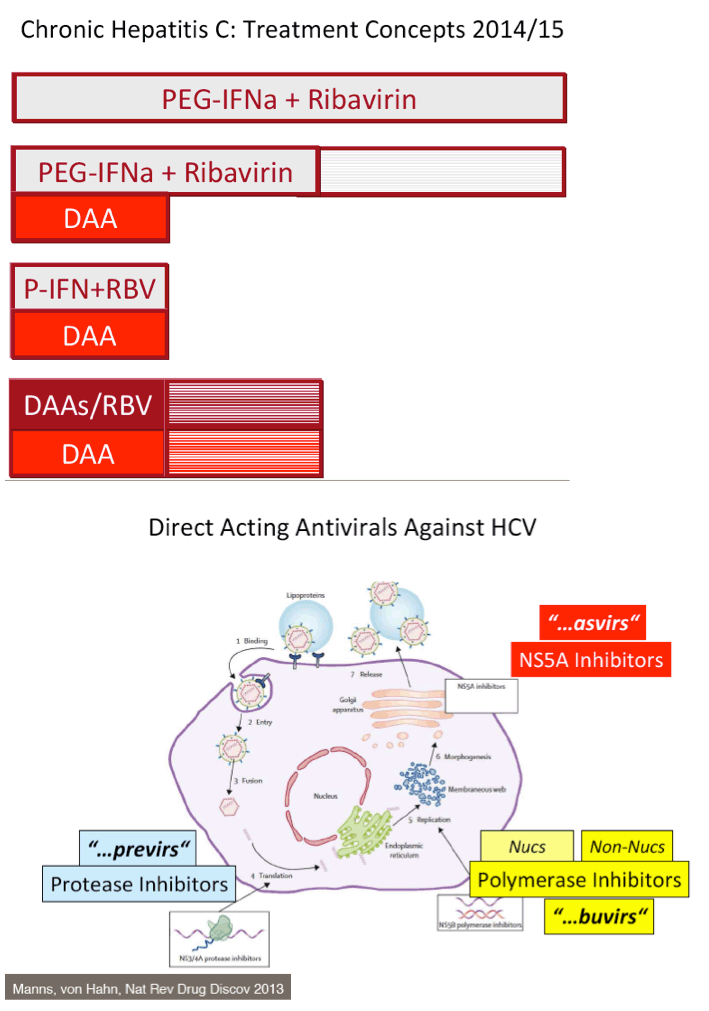
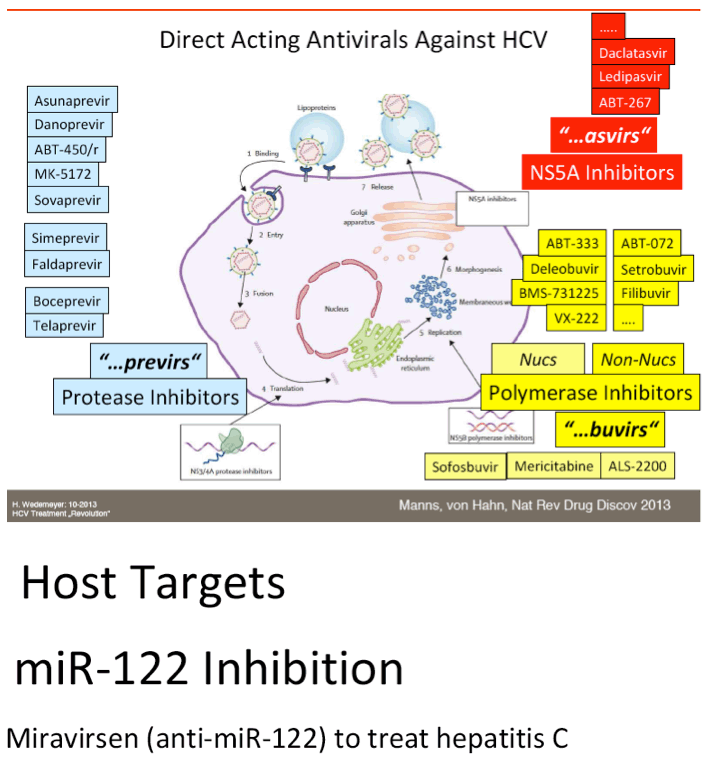
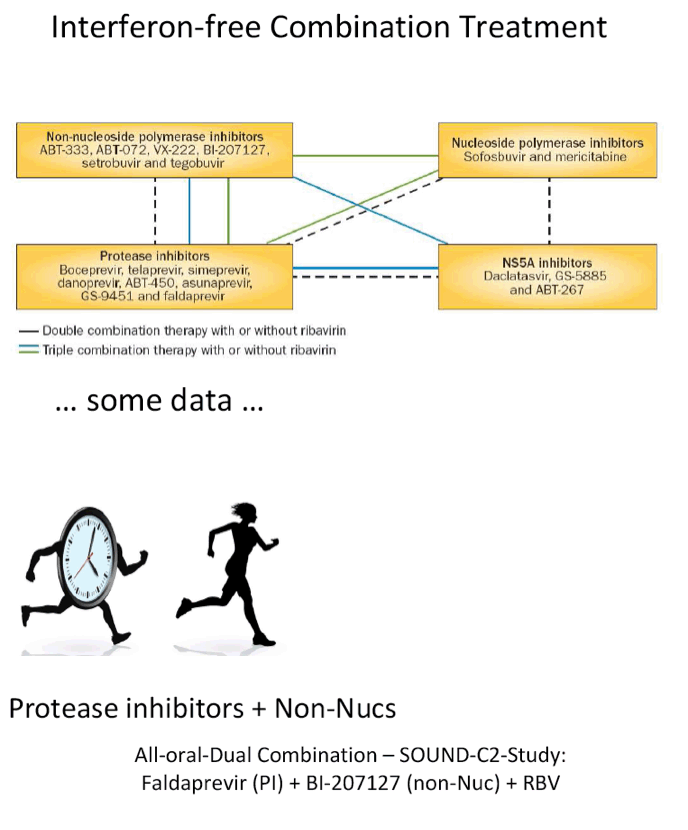
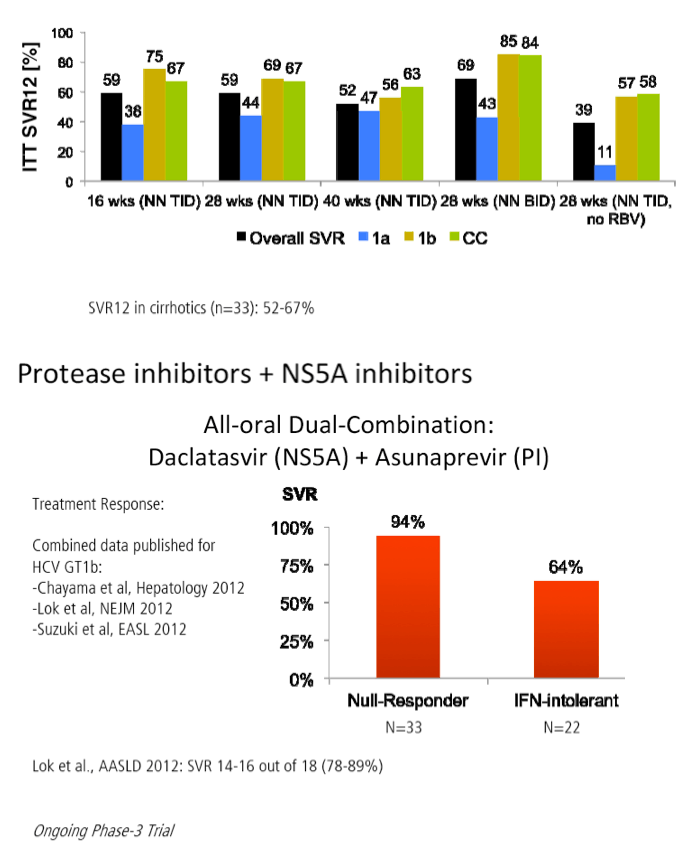
The FDA hearings on Oct 24/25 reviewed Simeprevir+PR for 24 weeks and Sofosbuvir +PR for 12 weeks, and as expected the panels voted unanimously to approve both 19-0 & 15-0. This is just the prelude to next year when the first 2 IFN-free regimens will be considered for approval by the FDA in the early part of 2014 & of course approval is expected, they are Gilead's Sofosbuvir+Ledipasvir & Abbvie's ABT450+ABT267+ABT333 both with and/or without RBV, phase 3 studies are ongoing, fully enrolled, phase 2 studied reported 95-100% SVR rates with 12 weeks therapy. FDA approval is expected around end of November & beginning of Decmber 2013. In phase 2 studies, Sofosbuvir+Simprevir has yielded 96% SVR rates in hard-to-treat null responders & patients with advanced disease, and Sofosbuvir+Daclatasvir has yielded 100% SVR rates in naives & patients who previously failed telaprevir or boceprevir. Earlier in research is Merck's 2nd generation drugs, MK8472, their NS5A & MK5172, their protease, phase 2 data will be reported at AASLD next week, phase 3 is planned. Boerhinger Ingelheim is studying a 3-drug oral combination for which they reported early results a few weeks ago with 90-100% SVR rates, which includes their protease Faldaprevir, their non-nuc & the Presidio NS5A. BMS is planning phase 3 now for their 3-drug IFN/Rbv free regimen which yielded a 94% SVR rate in phase 2. Vertex is studying their nucleotide in combination with other DAAs from other companies. Janssen istudying their own 3-drug oral combination, last week they announced acquisition of the GSK NS5A inhibitor to combine with their protease Simeprevir and their non-nuc. Roche is reporting results at this AASLD for their ANNAPR+URNA Study which looked at their 3-4 drug oral regimen which included their protease Danoprevir, Mericitabine, their nuc, and their non-nuc.
"All oral therapy will lead to a change in treatment paradigm"
"The number of treatable patients will dramatically increase! Interferon-Free"
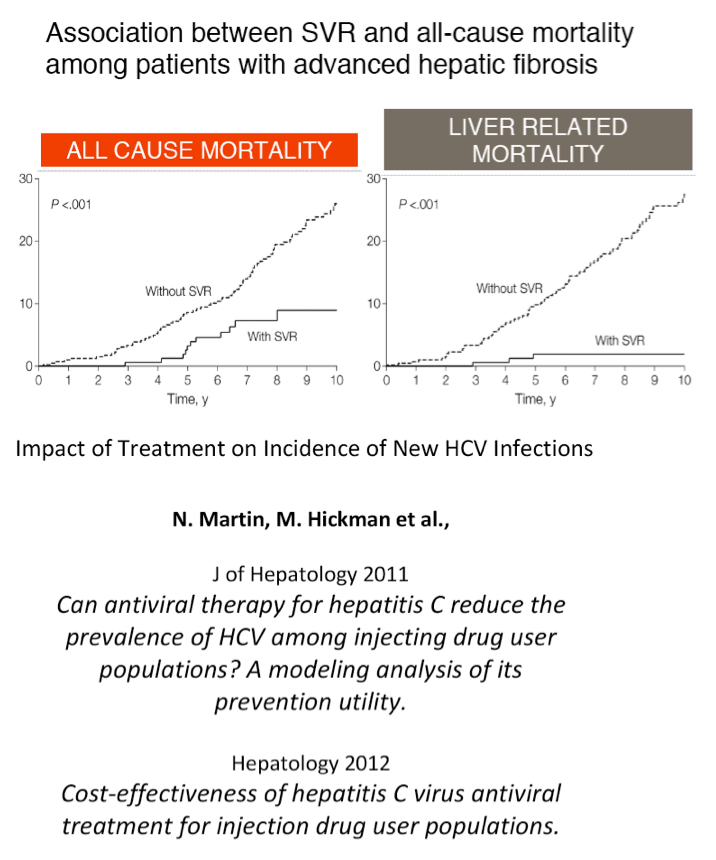
"There is an association between SVR & all-cause mortality & liver-related mortality"
'New HCV oral IFN-free drug regimens will provide with 12-24 weeks convenient therapy an SVR is associated with reduced risk of progression to advanced liver disease, liver cancer and mortality
Will prevent HCV transmission and be cost-effective.....therapies will be once or twice daily, much less side effects than current IFN-based therapy
Drug interactions will be an issue but we can deal with that'

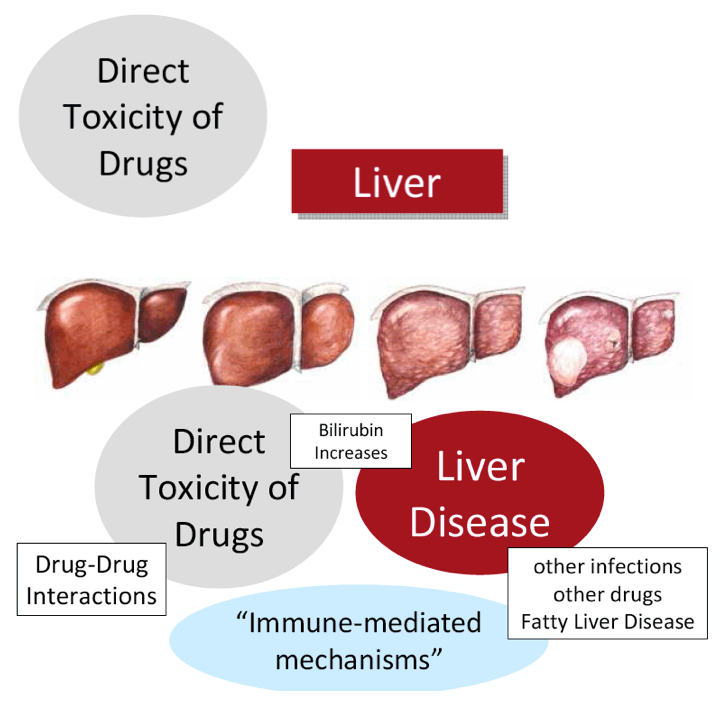

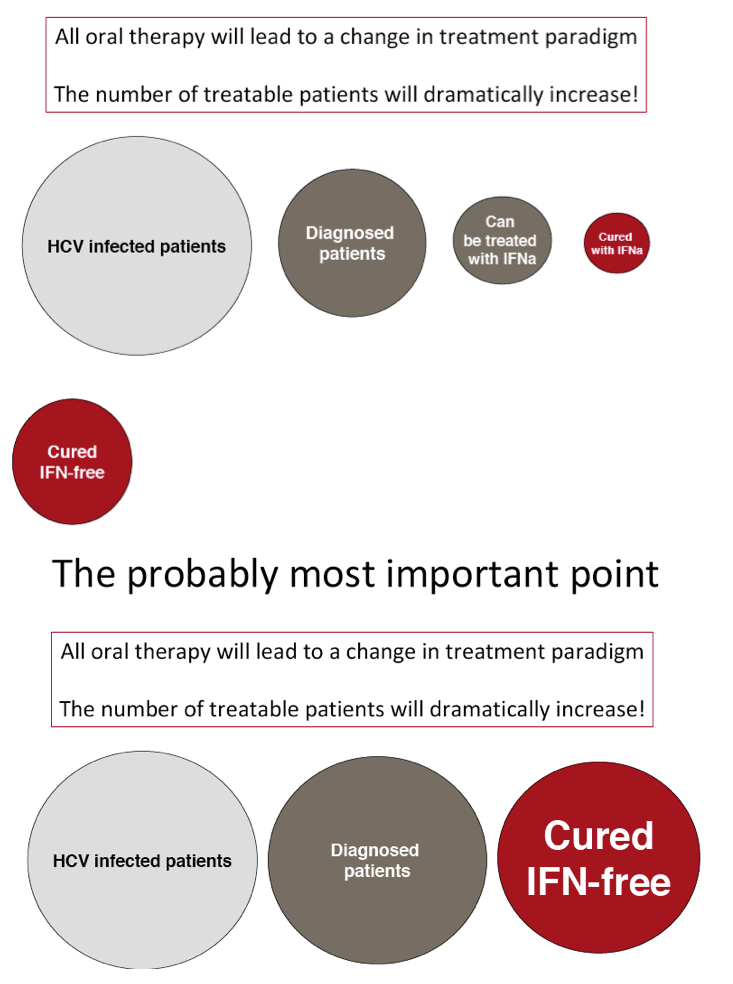

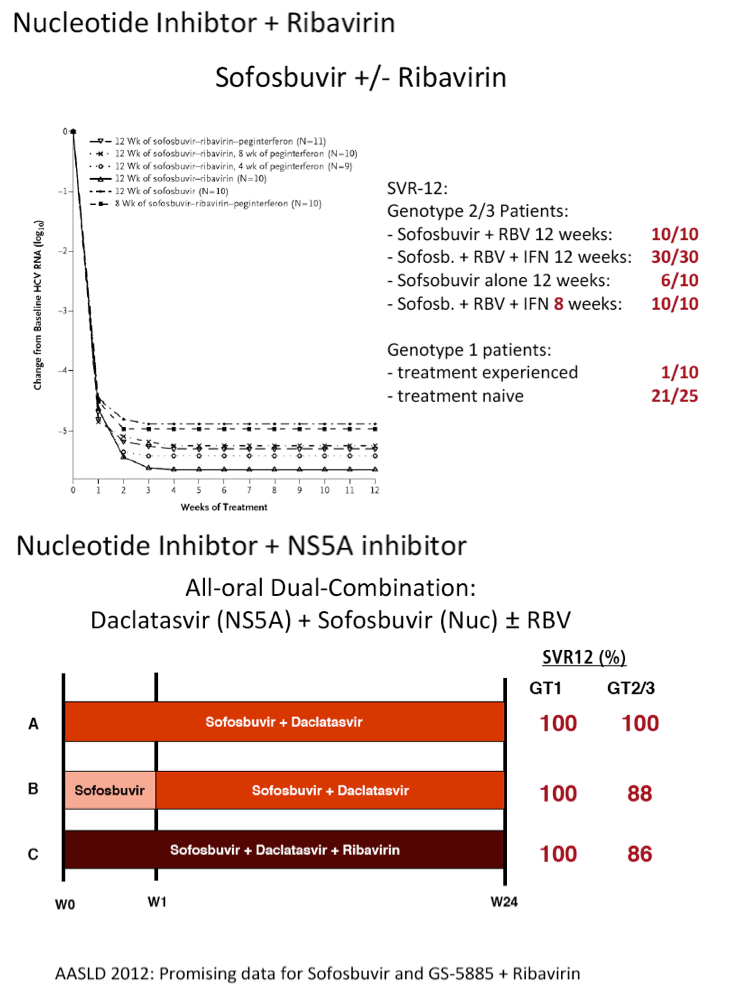
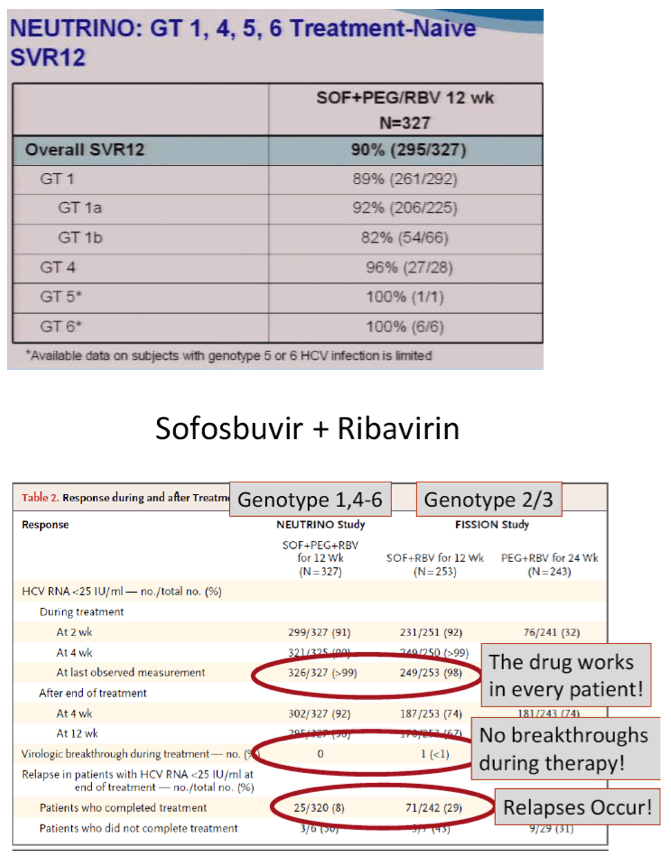
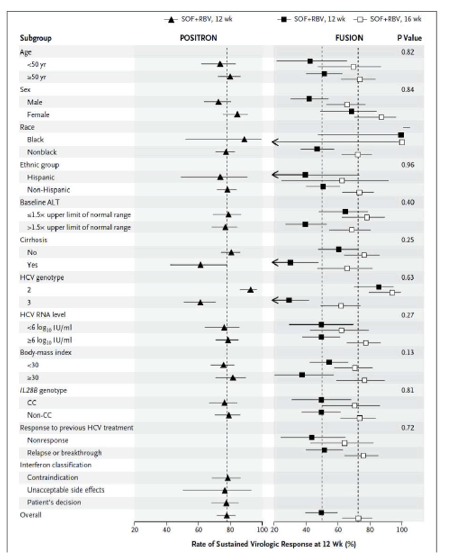
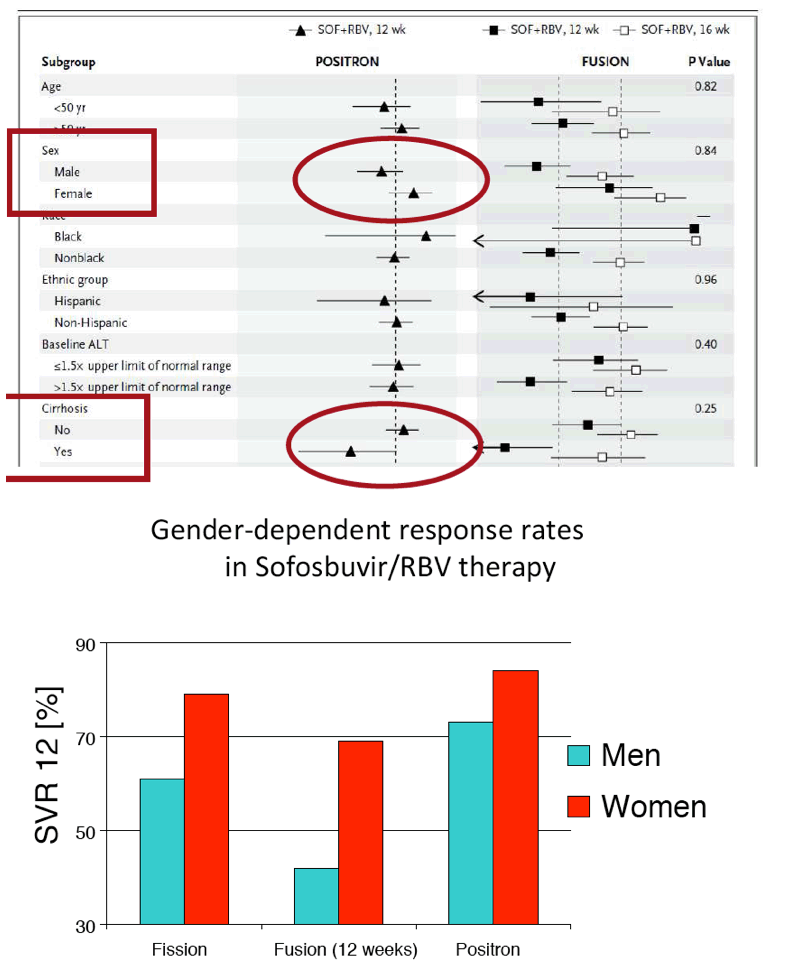
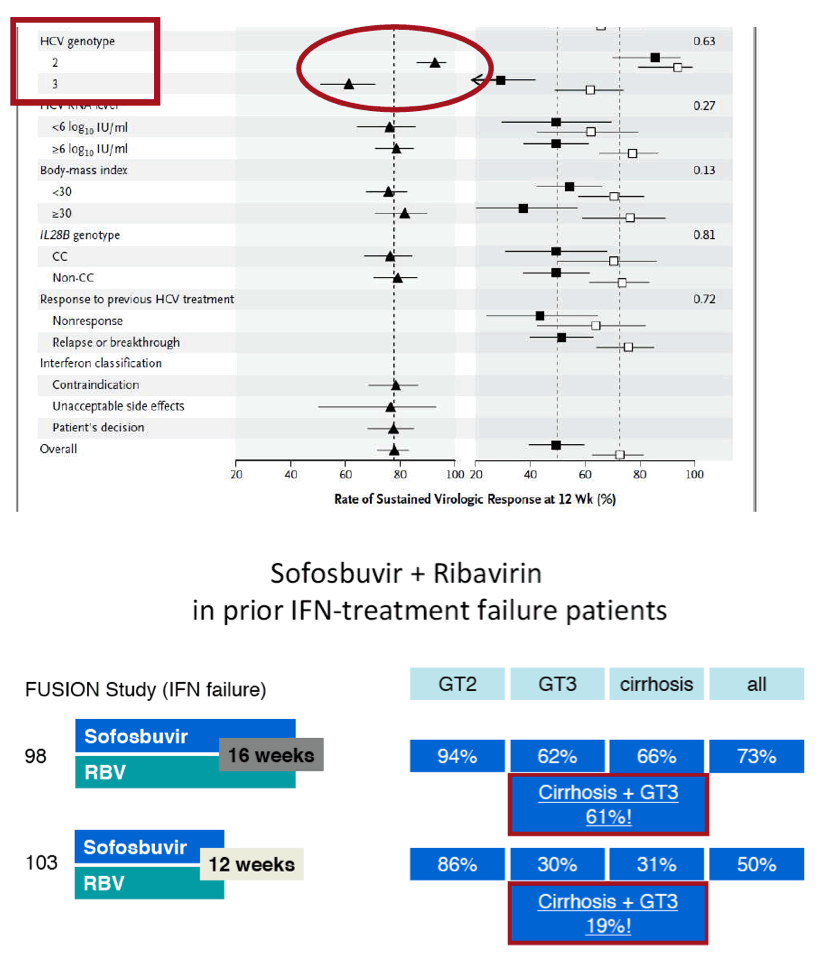
The FDA hearings on Oct 24/25 reviewed Simeprevir+PR for 24 weeks and Sofosbuvir +pR for 12 weeks, and as expected the panels voted unanimously to approve both 19-0 & 15-0. This is just the prelude to next year when the first 2 IFN-free regimens will be considered for approval by the FDA in the early part of 2014 & of course approval is expected, they are Gilead's Sofosbuvir+Ledipasvir & Abbvie's ABT450+ABT267+ABT333 both with and/or without RBV, phase 3 studies are ongoing, fully enrolled, phase 2 studied reported 95-100% SVR rates with 12 weeks therapy. FDA approval is expected around end of November & beginning of Decmber 2013. Sofosbuvir+Simprevir has yielded 96% SVR rates in hard-to-treat null responders & patients with advanced disease. Sofosbuvir+Daclatasvir has yielded 100% SVR rates in naives & patients who previously failed telaprevir or boceprevir.








|
| |
|
 |
 |
|
|