 |
 |
 |
| |
VAST MAJORITY OF DETECTED NS5A RESISTANT VARIANTS ARE NOT AMPLIFIED IN HCV PATIENTS DURING 3-DAY MONOTHERAPY WITH THE OPTIMIZED NS5A INHIBITOR PPI-668
|
| |
| |
See attached poster pdf for a full view of the individual patient resistance/pk data.
Download the pdf here
Reported by Jules Levin
EASL 2013 Amsterdam April 28, 2013
PPI-383, gt3/Pan-genotypic Non
Nuc.......http://www.natap.org/2013/HCV/042513_01.htm
3-drug oral combination study: PPI668+ BI335 + BI127 - Presidio Pharmaceuticals Announces Collaboration with Boehringer
Ingelheim....http://www.natap.org/2013/HCV/031413_01.htm
Qi Huang1, Ningwu Huang1, Anja Huq1, Miriam Lau1, Eric Peng1, Jay Lalezari2, Geoff Farrell3, Purvee Shah4, Eric Lawitz5, Pamela Vig1, Nathaniel Brown1 and Richard Colonno1 1Presidio Pharmaceuticals, San Francisco, CA, USA; 2Quest Clinical Research and UCSF, San Francisco, CA, USA; 3The Canberra Hospital, Canberra, Australia; 4West Coast Clinical Trials, Costa Mesa, CA, USA; and 5Alamo Medical Research, San Antonio, TX, USA

ABSTRACT
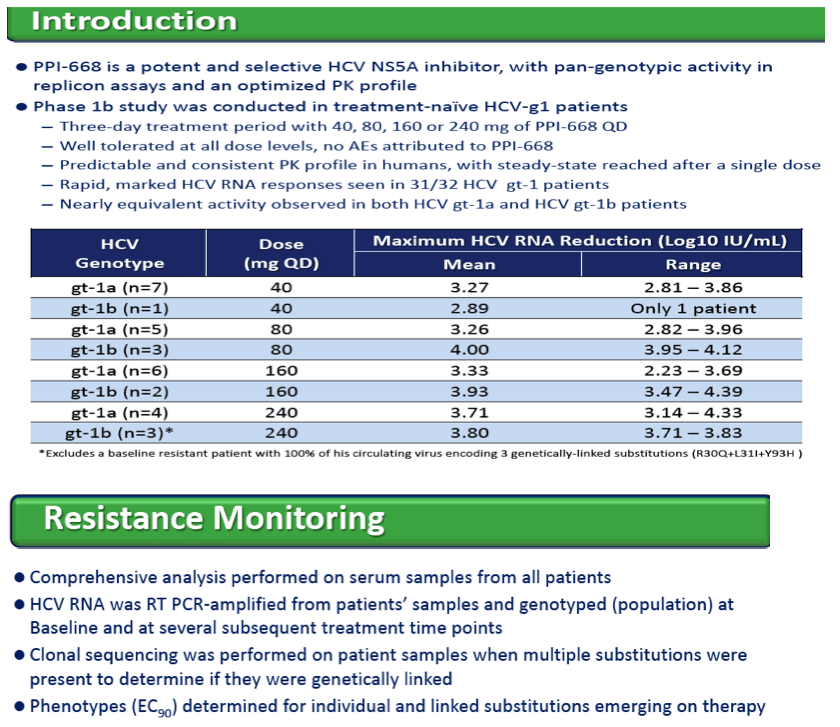
BACKGROUND: HCV NS5A inhibitor PPI-668 was assessed in a 3-day Phase 1b, placebo controlled, monotherapy trial in HCV gt-1 patients using QD oral doses of 40, 80, 160 and 240 mg, with 8/10 patients receiving PPI-668 per cohort. Mean maximal viral load reductions of 3.54-3.75 log10 IU/mL were observed with doses ≥80 mg QD.
METHODS: Comprehensive monitoring of HCV resistant variants was performed during dosing and post-dosing periods. HCV RNA was extracted from patient serum samples, RT-PCR amplified and the resulting DNA subjected to population sequencing. Clonal sequencing was performed to determine if detected substitutions were genetically linked. PPI-668 susceptibility was assessed in transient transfection assays using HCV replicons encoding specific substitutions or population NS5A gene inserts from clinical samples.
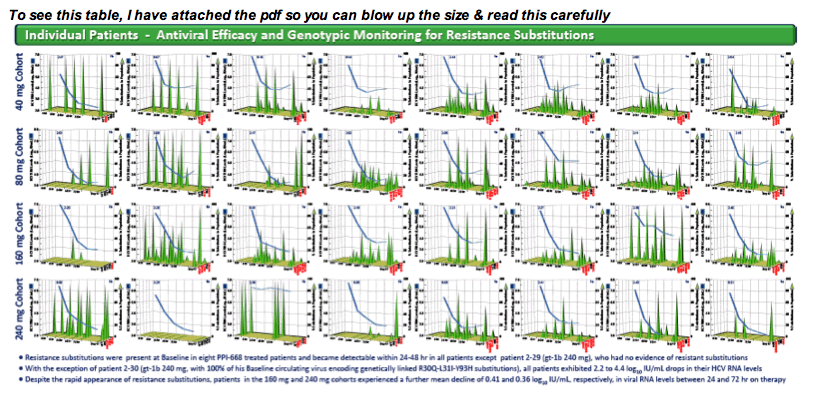
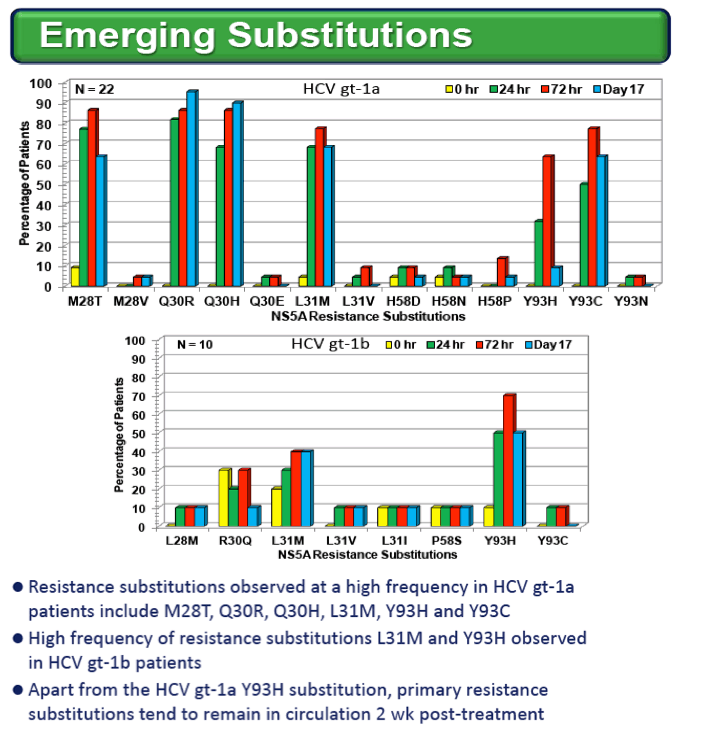
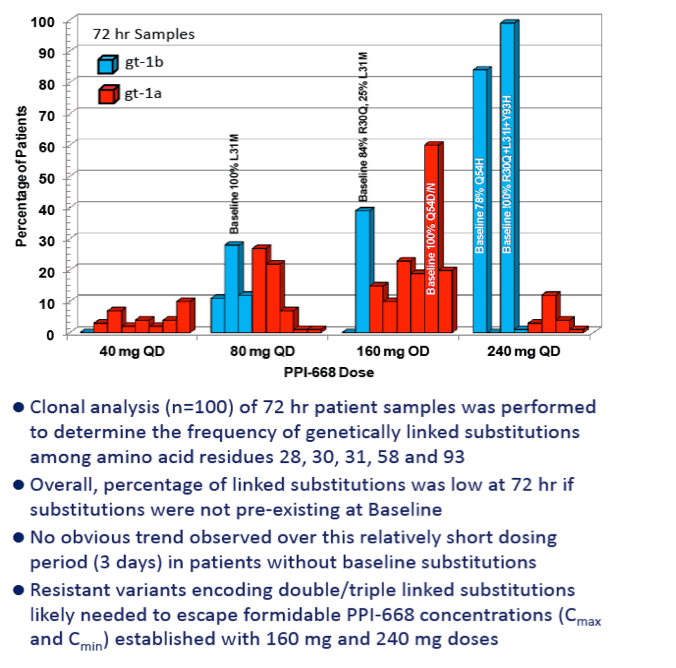
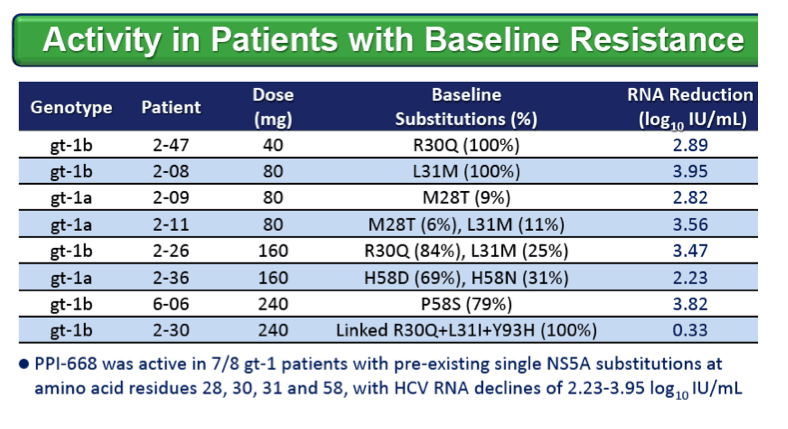
RESULTS: Known NS5A resistance substitutions at residues 28, 30, 31, 58 and 93 were detected early during PPI-668 monotherapy. Among the 40 enrolled gt-1 patients, eight had detectable NS5A resistance substitutions at baseline. One patient (gt-1b 240 mg) was a non-responder exhibiting high level resistance at baseline, with 100% of his circulating virus encoding genetically linked R30Q+L31I+Y93H substitutions. The other seven PPI-668 treated patients with baseline resistance substitutions responded well (RNA reductions of 2.23 to 3.95 log10 IU/mL).
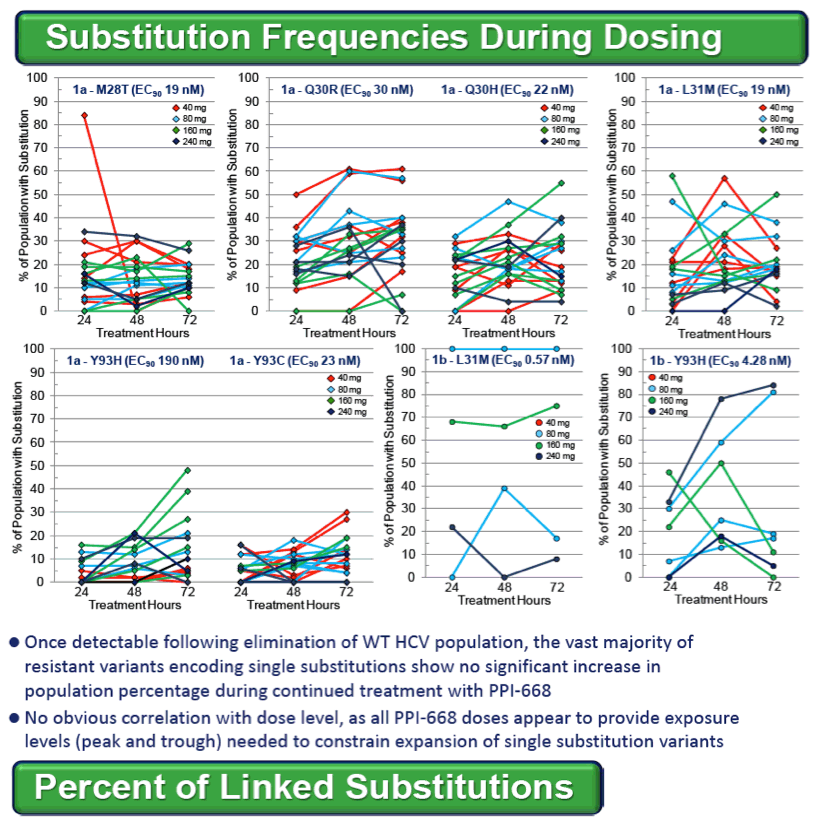
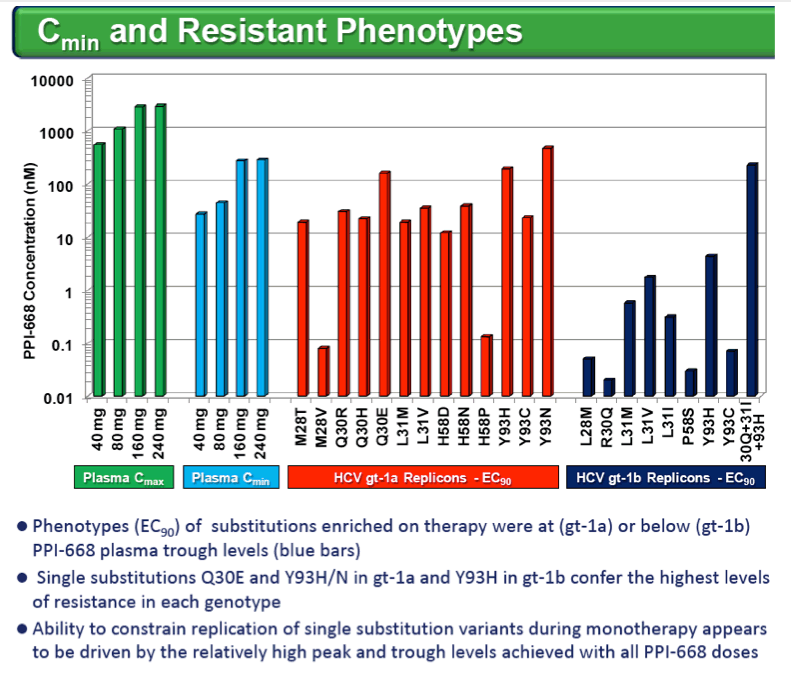
Resistance substitutions became detectable in all but one PPI-668 treated patient within 24-48 hr, as WT virus was rapidly eliminated. Importantly, these observed substitutions were not further amplified with continued monotherapy, suggesting that PPI-668 concentrations were sufficient to suppress these single-substitution variants. Susceptibility (EC90) of replicons encoding resistance substitutions detected in PPI-668 treated patient samples were generally at or below Cmin levels, confirming the advantageous PK profile of PPI-668 and its ability to cover single-substitution resistant variants. No significant differences were observed in the overall resistance patterns across the four PPI-668 treated gt-1 cohorts.
CONCLUSIONS: NS5A resistance variants frequently pre-exist among HCV patients, emphasizing the need for combination therapy and use of optimized NS5A inhibitors, such as PPI-668, that achieve plasma/liver levels high enough to suppress single substitution HCV variants. Further studies of PPI-668 in combination with other DAAs are warranted.
|
| |
|
 |
 |
|
|