 |
 |
 |
| |
HEPATOCELLULAR CARCINOMA (HCC) RISK IN HBeAg-NEGATIVE CHRONIC HEPATITIS B (CHBE-) WITH OR WITHOUT CIRRHOSIS TREATED WITH ENTECAVIR: RESULTS OF THE NATIONWIDE HEPNET.GREECE COHORT STUDY
|
| |
| |
Reported by Jules Levin
EASL 2013 Amsterdam April 24-28
AUTHOR CONCLUSIONS
The risk of HCC in CHBe-
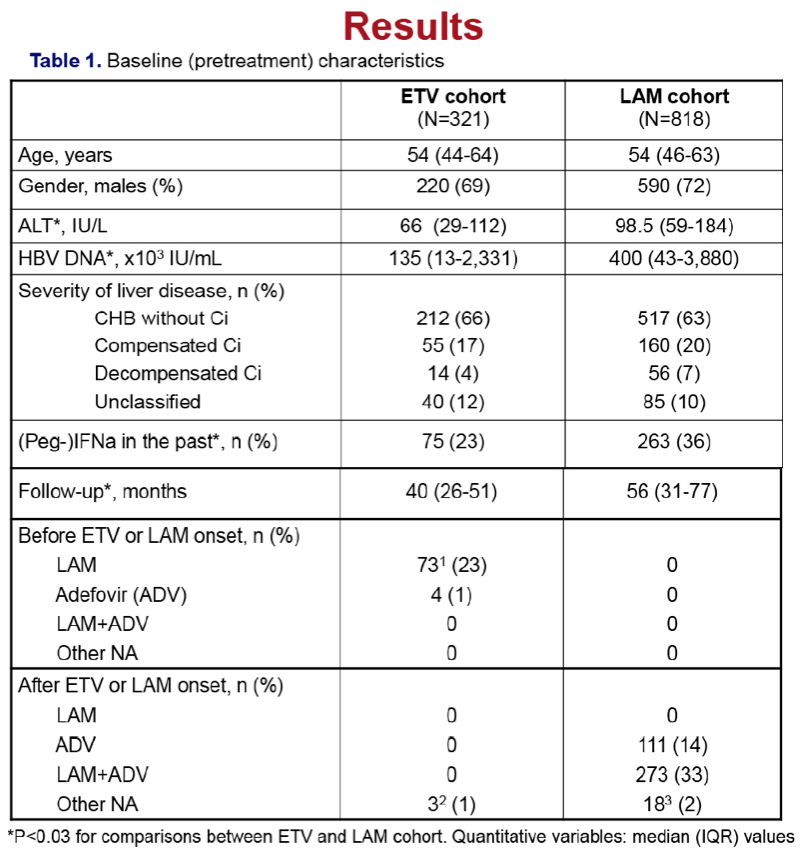
Remains increased in ETV treated patients with Ci (cirrhosis), particularly of older age, who should remain under surveillance
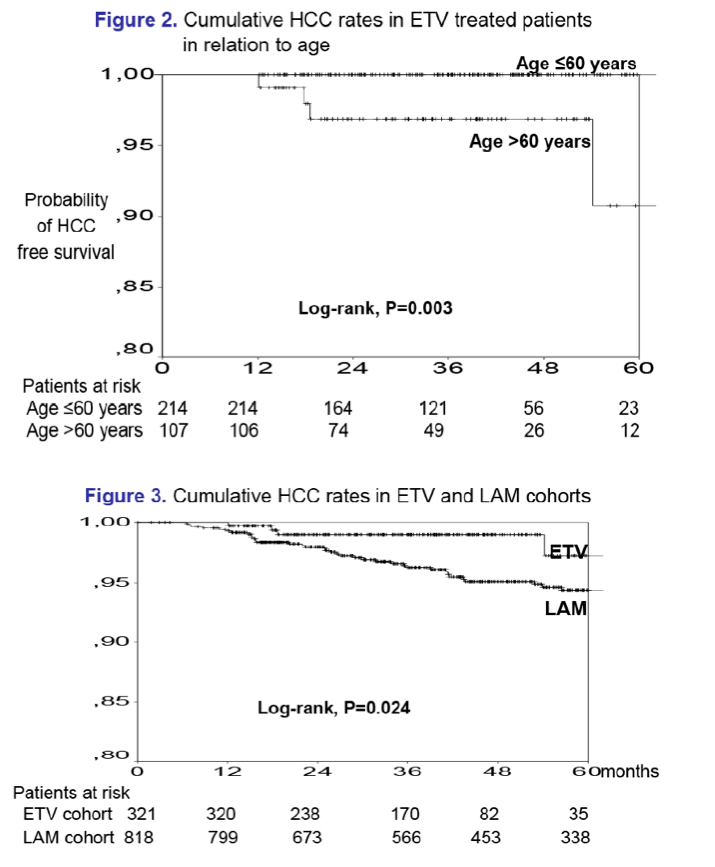
May be reduced with ETV compared to antiviral therapy starting with lamivudine, at least in patients with cirrhosis
More data in greater numbers of patients are required to evaluate the ETV effect on HCC incidence in cirrhosis
From Jules: as reported earlier in the tenofovir oral slide presentation, this risk is not confined to entecavir or lamivudeine but exists for tenofovir as well, the point is again as I mentioned in an earlier HCV-related report once cirrhosis develops the risk for HCC persists although much lower despite HCV SVR or successful HBV therapy, suggesting to me undue dely of therapy puts a patient at risk: HBV & HCV invade the liver and disease progression occurs over time, earlier therapy intervenes in that progression and the earlier the intervention the sooner progression is stopped. In HCV the difference is that HCV is short-term therapy & HCV is "curable", soon with IFN-free therapy of 12, 2 weeks.
G. Papatheodoridis1*, S. Manolakopoulos1, G. Touloumi2, G. Nikolopoulou3, M. Raptopoulou-Gigi4, C. Gogos5, I. Vafiadis-Zouboulis6, D. Karamanolis7, A. Chouta8, A. Ilias9, C. Drakoulis10, K.M. Mimidis11, I. Ketikoglou12, E. Manesis13, M. Mela14, G. Hatzis15, G. Dalekos16, HepNet.Greece Study Group
12nd Department of Internal Medicine, Athens University Medical School, Hippokration Hospital, 2Department of Hygiene, Epidemiology & Medical Statistics, Athens University School of Medicine, 3Viral Hepatitis Committee, KEELPNO, Athens, 42nd Medical Department, Aristotle University of Thessaloniki, Thessaloníki, 5Department of Internal Medicine, University of Patra, Patras, 61st Department of Propedeutic Medicine, Athens University Medical School, 72nd Gastroenterology Department, Evangelismos General Hospital of Athens, 84th Department of Internal Medicine, Athens University Medical School, Athens, 9Gastroenterology Department, Papanikolaou General Hospital of Thessaloniki, Thessaloníki, 102nd Department of Internal Medicine, General Hospital of Piraeus, Athens, 111st Department of Internal Medicine, Democritus University of Thrace, Alexandroupolis, 12Department of Internal Medicine, Hippokration General Hospital of Athens, 13Division of Internal Medicine, Athens University Medical School, 14Gastroenterology Department, Polykliniki Hospital of Athens, 15Department of Pathophysiology, Athens University Medical School, Athens, 16Department of Medicine & Research Laboratory of Internal Medicine, Thessaly University Medical School, Larissa, Greece.
ABSTRACT
Background and aims: HCC may still develop in patients starting with lamivudine, while the HCC rates in patients under newer antivirals are unclear. We estimated the incidence and evaluated predictors of HCC in a large nationwide prospective cohort of CHBe- patients treated with entecavir. The CHBe- patients from the same cohort (HepNet.Greece) who started lamivudine were used for comparison.
Methods: We included 321 adult CHBe- patients (age:53±14 years, males:69%) who were treated with entecavir for ≥12 months [antiviral(s)-naive:86%, lamivudine/adefovir exposure without resistance:23%/1%] and had no HCC during the first 6 months. Mean follow-up was 30±18 months. CHBe- without cirrhosis, compensated and decompensated cirrhosis were diagnosed in 75%, 20% and 5% of 281 patients (disease severity unclassified: 40). Lamivudine group included 818 CHBe- patients selected by the same criteria (therapy ≥12 months and no HCC within first 6 months).
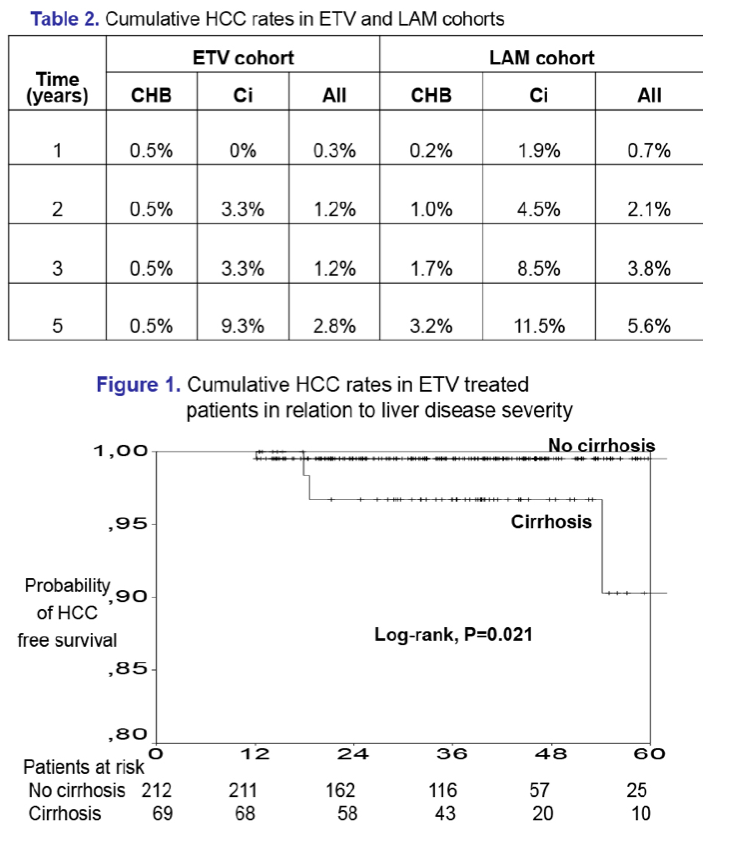
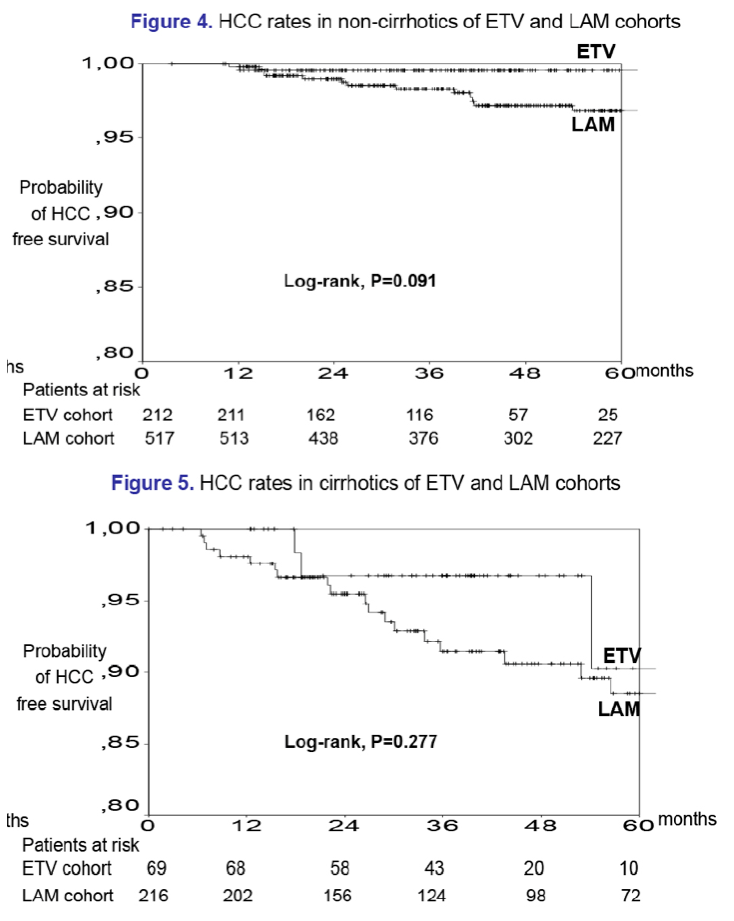
Results: In the entecavir group, HCC developed in 4/321 (1.2%) patients at a median of 1.5 (range:1.0-4.5) years, while the cumulative HCC incidence was significantly higher in cirrhotic than non-cirrhotic patients (1-, 3-, 5-year: 0%, 3%, 17% vs 1%, 1%, 1%; P=0.024, log-rank). The probability of HCC under entecavir was also higher in older patients [HR per year:1.21 (95% CI:1.02-1.42), P=0.026], but it was not significantly associated with gender or baseline ALT and HBV-DNA. Entecavir compared to lamivudine group patients had similar age, gender and cirrhosis distribution, but significantly lower baseline ALT and HBV-DNA levels and shorter follow-up, as well as a trend for lower HCC incidence (1-, 3-, 5-year: 0.7%, 3.8%, 5.6% vs 0.3%, 1.1%, 4.8%; P=0.096, log-rank). However, in multivariable Cox regression analysis including all 1139 patients, the HCC risk was independently associated with older age [HR per year:1.10 (1.06-1.13), P=0.001], male gender [HR:2.88 (1.28-6.50), P=0.011] and cirrhosis [HR:2.00 (1.10-3.66), P=0.024), but not with type of initial therapy.
Conclusions: Data from this large nationwide study indicate that the HCC risk remains increased in entecavir treated CHBe- patients with cirrhosis, particularly of older age. The HCC risk does not seem to be significantly reduced with entecavir compared to antiviral therapy starting with lamivudine, at least during the first 3 years of treatment.




|
| |
|
 |
 |
|
|