 |
 |
 |
| |
WHO issues new ART HIV recommendations calling for earlier treatment - More Conservative Than HHS ART Guidelines
|
| |
| |
Download the PDF here
Download the PDF here
WHO & HHS Guidelines pdf attached (BUT HHS Guidelines recommend: "ART is recommended for all HIV-infected individuals to reduce the risk of disease progression....and prevention of transmission of HIV" see below for explanation Guidelines pdf attached
Earlier, safer and simpler antiretroviral therapy can push the HIV epidemic into irreversible decline
"The recommendation for initiating ART at CD4 counts between 350 and 500 cells/mm3 is based on a systematic review with GRADE evidence profiles (Web Annex www.who.int/hiv/pub/guidelines/arv2013/annexes) that assessed the quality and strength of the evidence from 21 observational studies (8,19-39) and three randomized controlled trials (3,18,40) reporting morbidity, mortality and immunological and virological outcomes.
They showed that initiating ART at a CD4 count >350 cells/mm3 compared with treatment at a CD4 count ≤350 cells/mm3 reduced the risk of progression to AIDS and/or death, TB, development of a non-AIDS-defining illness and increased the likelihood of immune recovery. Although no studies suggest that earlier ART causes individual harm, these
studies were of limited duration." see below
News release
30 June 2013 | Geneva - New HIV treatment guidelines by WHO recommend offering antiretroviral therapy (ART) earlier. Recent evidence indicates that earlier ART will help people with HIV to live longer, healthier lives, and substantially reduce the risk of transmitting HIV to others. The move could avert an additional 3 million deaths and prevent 3.5 million more new HIV infections between now and 2025.
The new recommendations are presented in WHO's "Consolidated guidelines on the use of antiretroviral drugs for treating and preventing HIV infection", as new data reveal a total of 9.7 million people were taking these lifesaving drugs at the end of 2012.
"These guidelines represent another leap ahead in a trend of ever-higher goals and ever-greater achievements," says WHO Director-General Dr Margaret Chan. "With nearly 10 million people now on antiretroviral therapy, we see that such prospects - unthinkable just a few years ago - can now fuel the momentum needed to push the HIV epidemic into irreversible decline."
Call to initiate treatment at 500 CD4 cells/mm3 or less
The new recommendations encourage all countries to initiate treatment in adults living with HIV when their CD4 cell count falls to 500 cells/mm3 or less - when their immune systems are still strong. The previous WHO recommendation, set in 2010, was to offer treatment at 350 CD4 cells/mm3 or less. 90% of all countries have adopted the 2010 recommendation. A few, such as Algeria, Argentina and Brazil, are already offering treatment at 500 cells/mm3.
WHO has based its recommendation on evidence that treating people with HIV earlier, with safe, affordable, and easier-to-manage medicines can both keep them healthy and lower the amount of virus in the blood, which reduces the risk of passing it to someone else. If countries can integrate these changes within their national HIV policies, and back them up with the necessary resources, they will see significant health benefits at the public health and individual level, the report notes.
Further recommendations
The new recommendations also include providing antiretroviral therapy - irrespective of their CD4 count - to all children with HIV under 5 years of age, all pregnant and breastfeeding women with HIV, and to all HIV-positive partners where one partner in the relationship is uninfected. The Organization continues to recommend that all people with HIV with active tuberculosis or with hepatitis B disease receive antiretroviral therapy.
Another new recommendation is to offer all adults starting to take ART the same daily single fixed-dose combination pill. This combination is easier to take and safer than alternative combinations previously recommended and can be used in adults, pregnant women, adolescents and older children.
"Advances like these allow children and pregnant women to access treatment earlier and more safely, and move us closer to our goal of an AIDS-free generation," said UNICEF Executive Director, Anthony Lake. "Now, we must accelerate our efforts, investing in innovations that allow us to test new born babies faster and giving them the appropriate treatment so that they enjoy the best possible start in life."
The Organization is further encouraging countries to enhance the ways they deliver HIV services, for example by linking them more closely with other health services, such as those for tuberculosis, maternal and child health, sexual and reproductive health, and treatment for drug dependence.
"The new WHO guidelines are very timely in view of the rapid progress we have made in expanding programmes for prevention and treatment," says Dr Mark Dybul, Executive Director of the Global Fund to Fight AIDS, Tuberculosis and Malaria. "This is an example of how the Global Fund and the WHO work together to support countries as we move towards removing HIV as a threat to public health." Since its creation in 2002, the Global Fund has supported more than 1,000 programmes in 151 countries, providing HIV treatment for 4.2 million people.
Challenges remain
Challenges still remain. Alongside the new treatment guidelines, a treatment progress update by WHO, UNAIDS, UNICEF identified areas in need of attention.
While the number of all eligible children on ART has increased by 10% between 2011 and 2012, this is still too slow compared to the 20% increase in adults. A further complication is that many key populations such as people who inject drugs, men who have sex with men, transgender people and sex workers, continue to face legal and cultural barriers that prevent them getting treatment that otherwise would be more easily available. Another factor that needs to be addressed is the significant proportion of people who, for many reasons, 'drop out' of treatment.
Data reinforces feasibility of recommendations
Despite this, the Global update on HIV treatment: results, impact and opportunities contains encouraging data that reinforces the feasibility of the new WHO recommendation on earlier ART, which would expand the global number of people eligible for antiretroviral therapy to 26 million.
Between 2011 and 2012, the largest acceleration ever of people enrolled on ART was achieved, with an extra 1.6 million people benefitting from antiretroviral therapy, increasing the total to 9.7 million people. Furthermore, increased coverage of treatment occurred in every region of the world, with Africa leading. Four out of 5 people who started treatment in 2012 were living in sub-Saharan Africa.
"Today nearly 10 million people have access to lifesaving treatment. This is a true development triumph," says Michel Sidibe, Executive Director of the Joint United Nations Programme on HIV/AIDS (UNAIDS). "But we now have a new challenge - ensuring that all 26 million people eligible for treatment have access - not one person less. Any new HIV infection or AIDS-related death due to lack of access to antiretroviral therapy is unacceptable."
Today's recommendations were released by WHO on the opening day of the International AIDS Society 2013 conference in Kuala Lumpur. Among those endorsing the recommendations at the launch were representatives from countries, where such earlier ART intervention is already national policy, along with development agencies who are providing technical and financial support.
The International AIDS Society conference is held every two years and attracts leading scientists, clinicians, public health experts and community leaders to examine the latest developments in HIV-related research, and to explore how scientific advances can inform the global response to HIV/AIDS.
Note to editors:
The recommended treatment is now a combination of three antiretroviral drugs: tenofovir and lamivudine (or emtricitabine) and efavirenz, as a single pill, given once daily.
HHS

a There is insufficient evidence and/or favourable risk-benefit profile to support initiating ART at a CD4 cell count
>500 cells/mm3 or regardless of CD4 cell count or WHO clinical stage in the following situations: individuals with HIV
older than 50 years, individuals with HIV-1 infected or coinfected with HIV-2, individuals with HIV coinfected with HCV and
key populations with HIV with a high risk of transmission (such as people who inject drugs, men who have sex with men,
transgender people and sex workers). ART initiation in these populations should therefore follow the same principles and
recommendations as for other adults with HIV.
b There is insufficient evidence and/or favourable risk-benefit profile to support initiating ART in everyone coinfected with HIV
and HBV with a CD4 count >500 cells/mm3 or regardless of CD4 cell count or WHO clinical stage. Initiating ART regardless
of CD4 count is therefore recommended among people with evidence of severe chronic liver disease, who are at greatest risk
of progression and mortality from liver disease. For people without evidence of severe chronic liver disease, ART initiation
should follow the same principles and recommendations as for other adults.

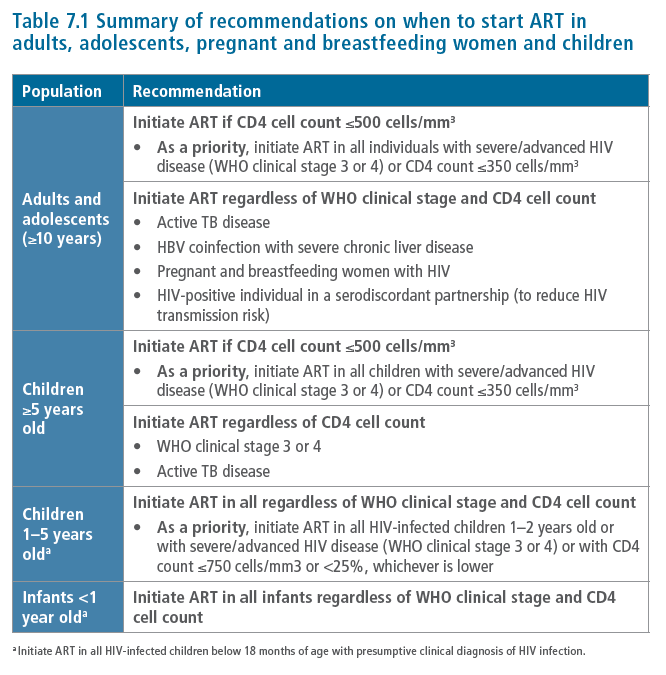
Background
Since 2002, WHO guidelines on ART have evolved as the body of evidence to support
the earlier initiation of ART has progressively increased (1). The 2010 WHO guidelines
for adults and adolescents (2) recommended initiating ART for all individuals (including
pregnant women) with a CD4 count ≤350 cells/mm3 regardless of WHO clinical stage and
for those with severe or advanced HIV disease (WHO clinical stages 3 or 4) regardless of
CD4 count. This strong recommendation was based on moderate-quality evidence from
randomized controlled trials (3,4) and observational studies (5-8) showing that initiating
ART at or below this CD4 threshold reduced mortality, disease progression (including TB),
vertical HIV transmission and serious adverse events. Mathematical modelling simulations
also suggested that initiating ART earlier could impact on both sexual and vertical HIV
transmission if there is high treatment coverage and full adherence (9). For people with
active TB disease or HBV coinfection requiring HBV treatment, the 2010 guidelines (2)
recommended initiating ART regardless of CD4 cell count.
Global ART coverage for those eligible according to the 2010 recommendations (CD4
≤350 cells/mm3) had reached 54% - or more than 8 million people - by the end of 2011
(10) , but coverage varies across regions, ranging from 15% to 68% (11). Only 9 lowand
middle-income countries have reported coverage exceeding 80%, and 68 countries
have reported coverage of less than 50%. Nevertheless, policy changes in countries
have been significant. A recent survey in 92 countries (Web Annex www.who.int/hiv/pub/guidelines/arv2013/annexes) showed that more than 90% had adopted the CD4
threshold for initiating ART of 350 cells/mm3 or less, and several other countries have
moved their CD4 threshold above 350 cells/mm3. The median CD4 count at the time ART
is initiated, although increasing, has been far lower than 350 cells/mm3 in almost all
settings, including high-income countries (12,13) , and late presentation for treatment is
associated with high early mortality rates and poor retention in care (6,14). Increasing
knowledge of HIV status, strengthening links between testing and care and ensuring
optimal long-term retention and adherence remain significant challenges in many
settings.
Rationale and supporting evidence
Since 2010, evidence and programmatic experience have continued to shift the riskbenefit
ratio towards initiating ART earlier. Increasing evidence also indicates that
untreated HIV may be associated with the development of several non-AIDS-defining
conditions (including cardiovascular disease, kidney disease, liver disease, several
types of cancer and neurocognitive disorders) (15-17) and that initiating ART earlier
reduces such events and improves survival. Recent evidence (18) also show that ART
substantially reduces sexual transmission in HIV-serodiscordant couples, but not all
studies have reported survival benefits. At the same time, more convenient and less
toxic regimens have become more widely available, and ARV costs have continued to
fall. How early ART should be started is still debated, and the Guidelines Development
Group paid close attention to evaluating the potential benefits and harms to the
individual and community in developing these new recommendations.
Initiating ART in individuals with symptomatic and asymptomatic
HIV disease at a CD4 count ≤350 cells/mm3 as a priority
The benefits of initiating ART are greatest among individuals with symptomatic HIV
disease or those with lower CD4 counts. The 2013 Guidelines Development Group did
not change the strength and quality of evidence for this recommendation established in
the 2010 ART guidelines (2). Moderate-quality evidence from two randomized controlled
trials and several observational studies shows that initiating ART at CD4 ≤350 cells/mm3
significantly reduces mortality, disease progression and the incidence of opportunistic
diseases, especially TB and non-AIDS-defining conditions (2).
Initiating ART at a CD4 count between 350 and 500 cells/mm3
The risk-benefit analysis of the rationale for ART initiation between 350 and 500 CD4
cells/mm3 in these guidelines was debated. The Guidelines Development Group agreed
that impact on HIV transmission is strongly supported by the evidence. The quality
of evidence for clinical benefit of earlier ART initiation was rated as moderate using
the GRADE system, as it mostly relies on observational data mainly from high-income
countries. The Guidelines Development Group strongly recommended earlier ART as a
public health approach. In settings where feasibility of implementation is a concern,
the Guidelines Development Group suggested conducting operational research during
implementation to assess context-specific factors such as feasibility, linkage to and
retention in care, adherence and resource allocation.
The recommendation for initiating ART at CD4 counts between 350 and 500 cells/mm3
is based on a systematic review with GRADE evidence profiles (Web Annex www.who.
int/hiv/pub/guidelines/arv2013/annexes) that assessed the quality and strength of the
evidence from 21 observational studies (8,19-39) and three randomized controlled trials
(3,18,40) reporting morbidity, mortality and immunological and virological outcomes.
They showed that initiating ART at a CD4 count >350 cells/mm3 compared with treatment
at a CD4 count ≤350 cells/mm3 reduced the risk of progression to AIDS and/or death,
TB, development of a non-AIDS-defining illness and increased the likelihood of immune
recovery. Although no studies suggest that earlier ART causes individual harm, these
studies were of limited duration.
The pooled analysis of the observational studies found a consistent decreased risk of death
with earlier initiation of ART in 13 studies (21-23,26,29-31,34-39) and a decreased risk of
progression to AIDS or death in 9 studies (21,23,26,27,30,33,34,36,39) and 3 randomized
controlled trials (3,18,40) , with a low level of heterogeneity, supporting moderate-quality
evidence for earlier treatment. A further subgroup analysis showed a reduced risk of
mortality with a CD4 threshold for initiating ART of 500 cell/mm3. The impact on immune
recovery was inconsistent and rated as low- to very-low-quality evidence (20,24,28). Two
studies found no significant difference in the likelihood of viral suppression (<500 copies/
ml), risk of virological failure and viral rebound when treatment is initiated at higher or
lower CD4 cell counts (20,36).
In the pooled analysis of two randomized controlled trials (3,18) there was low-quality
evidence supporting ART initiation at higher CD4 thresholds for reducing mortality, disease
progression or the combined outcome of death and/or progression and, in one trial,
the risk of non-AIDS-defining illnesses. The risk of severe adverse events did not differ
significantly, but the risk of Grade 3 or 4 laboratory abnormalitiesii was increased in one
randomized controlled trial (40). Since treatment in the delayed arm of the SMART trial (3)
was initiated when the CD4 count fell below 250 cells/mm3 (rather than 350 cells/mm3),
the quality of the evidence for clinical benefit was graded as low because of imprecision
and indirectness.
A separate systematic review (41) identified one randomized clinical trial (18) and two
observational studies (42,43) reporting a decreased risk of TB when individuals initiated
ART with CD4 counts exceeding 350 cells/mm3. ART also reduces recurrent TB by about
50% (44). Dynamic models have suggested ART initiation above 350 cells/mm3 could lead
to a more substantial reduction in population tuberculosis incidence (45).
Finally, there is high-quality evidence from one randomized controlled trial (18) indicating
that earlier ART can markedly reduce the risk of sexual transmission to HIV-negative sexual
partners. This is supported by the secondary outcomes of a trial that also found a 92%
reduction in HIV sexual transmission from partners with HIV taking ART (46).
HHS
Patients with CD4 counts >500 cells/mm3
The NA-ACCORD study also observed patients who started ART with CD4 counts >500 cells/mm3 or after their CD4 counts dropped below this threshold. The adjusted mortality rates were significantly higher in the 6,935 patients who deferred therapy until their CD4 counts fell to <500 cells/mm3 than in the 2,200 patients who started therapy with CD4 counts >500 cells/mm3 (risk ratio: 1.94, 95% CI: 1.37-2.79).16 Although large and generally representative of the HIV-infected patients in care in the United States, the study has several limitations, including the small number of deaths and the potential for unmeasured confounders that might have influenced outcomes independent of ART.
In contrast, results from 2 cohort studies did not identify a benefit of earlier initiation of therapy in reducing AIDS progression or death. In an analysis of the ART-CC cohort,11 the rate of progression to AIDS/death associated with deferral of therapy until CD4 count is in the 351 to 450 cells/mm3 range was similar to the rate with initiation of therapy with CD4 count in the 451 to 550 cells/mm3 range (HR: 0.99, 95% CI: 0.76-1.29). There was no significant difference in rate of death identified between the two groups (HR: 0.93, 95% CI: 0.60-1.44). This study also found that the proportion of patients with CD4 counts between 451 and 550 cells/mm3 who would progress to AIDS or death before having a CD4 count <450 cells/mm3 was low (1.6%; 95% CI: 1.1%-2.1%). In the CASCADE Collaboration,17 among the 5,162 patients with CD4 counts in the 500 to 799 cells/mm3 range, compared with patients who deferred therapy, those who started ART immediately did not experience a significant reduction in the composite outcome of progression to AIDS/death (HR: 1.10, 95% CI: 0.67-1.79) or death (HR: 1.02, 95% CI: 0.49-2.12).
While it was not a clinical endpoint study, a recent clinical trial (Setpoint Study) randomized patients within 6 months of HIV seroconversion to receive either immediate ART for 36 weeks or deferred treatment. More than 57% of the study participants had CD4 count >500 cells/mm3. The deferred treatment group had a statistically higher risk of meeting ART eligibility criteria than the immediate treatment group. The study was halted early and illustrated that the time from diagnosis of early infection to the need for initiation of ART was shorter than anticipated in the deferred therapy group.20
The expanded use of ART to treat individuals with CD4 counts >500 cells/mm3 has also demonstrated public health benefits. In 2010, a large, publicly-funded clinic in San Francisco adapted a universal ART approach to initiate ART in all HIV-infected persons and evaluated temporal trends in viral suppression. In 534 patients entering the clinic with CD4 counts >500 cells/mm3, the 1-year incidence of viral suppression increased from 9% to 14% before universal ART to >52% after the approach was adopted. After adjustment, this policy was associated with a six-fold increase in the probability of viral suppression six months after clinic entry.21 Because the risk of HIV transmission is associated with level of viremia, from a public health standpoint, this reduction in community viral load can potentially reduce new HIV infections at the community level.
With a better understanding of the pathogenesis of HIV infection, the growing awareness that untreated HIV infection increases the risk of many non-AIDS-defining diseases (as discussed below), and the benefit of ART in reducing transmission of HIV, the Panel recommends initiation of ART in patients with CD4 counts >500 cells/mm3 (BIII).
When discussing initiation of ART at high CD4 cell counts (i.e., >500 cells/mm3), clinicians should inform patients that data on the clinical benefit of starting treatment at such levels are not conclusive, especially for patients with very high CD4 counts. Clinicians should also inform patients that viral suppression from effective ART can reduce the risk of sexual transmission to others. Patients should also be told that untreated HIV infection will eventually lead to immunological deterioration and increased risk of clinical disease and death. Therefore, if therapy is not initiated, continued monitoring and close follow-up is necessary.
Further ongoing research (both randomized clinical trials and cohort studies) to assess the short- and long-term clinical and public health benefits and cost effectiveness of starting therapy at higher CD4 counts is needed. Findings from such research will provide the Panel with guidance to make future recommendations.
|
| |
|
 |
 |
|
|