 |
 |
 |
| |
Less Bone Loss With Raltegravir Than NRTIs in Second-Line Regimens
|
| |
| |
7th IAS Conference on HIV Pathogenesis, Treatment and Prevention, June 30-July 3, 2013, Kuala Lumpur
Mark Mascolini
People randomized to second-line regimens containing raltegravir and lopinavir/ritonavir lost less bone mineral density (BMD) through 48 weeks than those randomized to nucleosides or tenofovir (NRTIs) plus lopinavir/ritonavir in a multinational study [1]. But incidence of osteopenia or osteoporosis did not differ between the two treatment arms.
BMD declines during antiretroviral therapy, especially among people taking tenofovir. Because BMD changes with the integrase inhibitor raltegravir are poorly defined, SECONDLINE trial investigators conducted this 48-week comparison.
SECONDLINE is a 96-week trial at 37 sites in Asia, Africa, Australia, Latin America, and Europe. People taking a failing nonnucleoside-based combination were randomized to once- or twice-daily lopinavir/ritonavir plus two or three NRTIs or to lopinavir/ritonavir plus twice-daily raltegravir with no NRTIs. Participants must be at least 16 years old and have two consecutive viral loads above 500 copies while taking a first-line nonnucleoside regimen. No one can have taken protease inhibitors or integrase inhibitors.
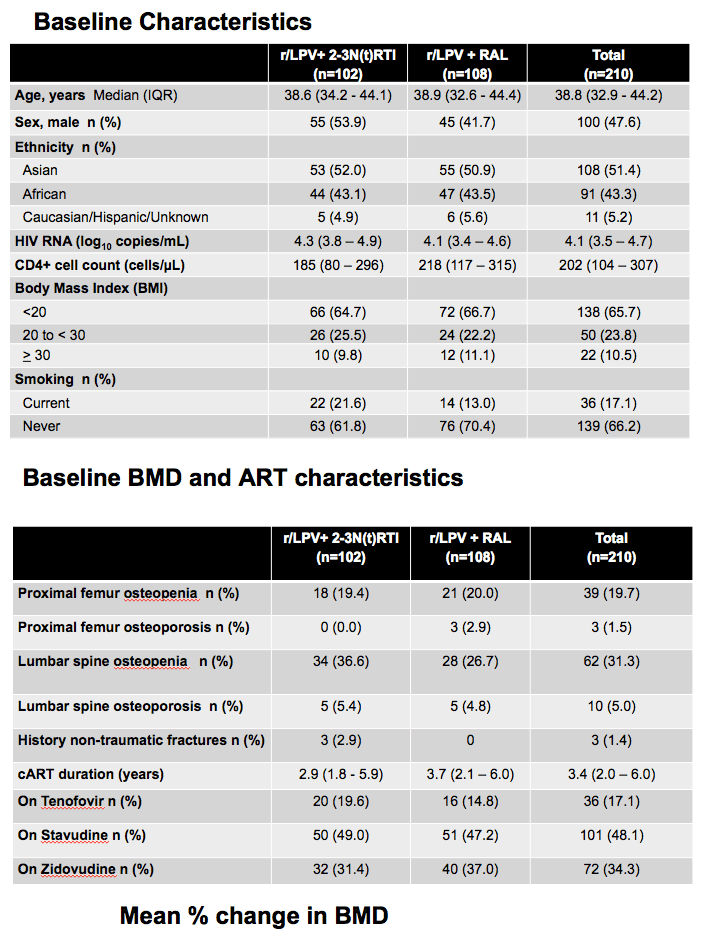
A bone substudy involved eight sites in South Africa, India, Malaysia, Thailand, and Argentina. Substudy participants had DEXA scans of the proximal femur and lumbar spine at week 0 and week 48. Among 236 people who consented to the bone substudy, 211 were randomized. Among 108 people who began lopinavir plus raltegravir, 107 reached week 48. Among 102 who began lopinavir plus NRTIs, 97 reached week 48.
About half of study participants were men, 51% Asian, 43% African, and 5% Caucasian. Two thirds of study participants (66%) had a body mass index below 20 kg/m(2), 24% were between 20 and less than 30 kg/m(2), and the rest were 30 kg/m(2) or more. (Body mass index below 18.5 kg/m(2) is considered underweight.) Body mass index distributions were similar in the two study arms. The lopinavir/NRTI group had a higher proportion of current smokers (22%) than did the lopinavir/raltegravir arm (13%). Low body mass index and smoking are risk factors for osteopenia and osteoporosis.
Baseline femoral osteopenia rates were 19% in the NRTI group and 20% in the raltegravir group. Respective baseline spinal osteopenia rates were 37% and 27%, while 5% in each group had spinal osteoporosis going into the study. Antiretroviral duration was shorter in the NRTI group than the raltegravir group (2.9 versus 3.7 years), and similar proportions were taking tenofovir (20% and 15%), stavudine (49% and 47%), or zidovudine (31% and 37%).
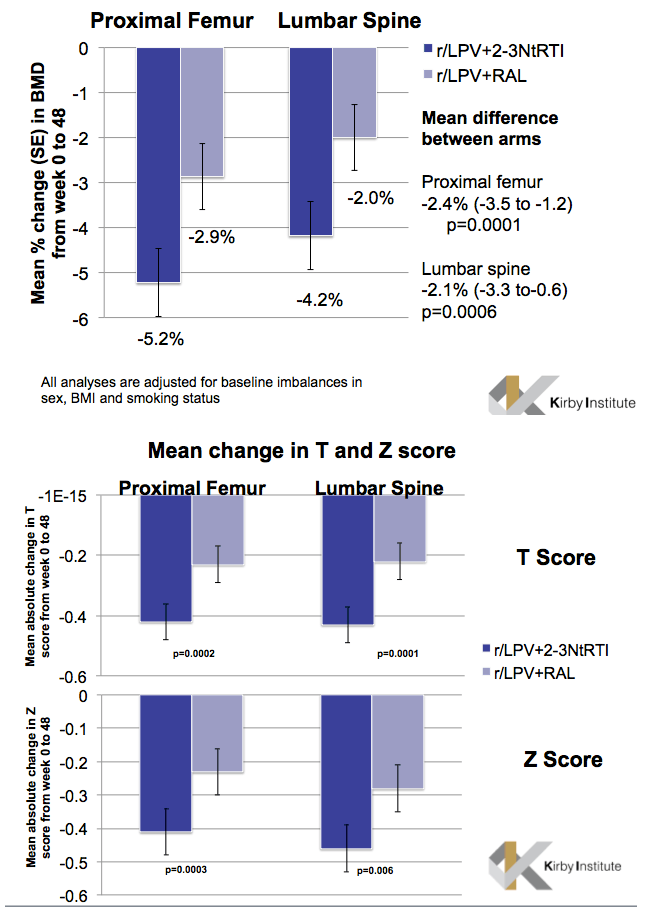
During 48 weeks of treatment, people randomized to lopinavir plus NRTIs lost significantly more BMD than those randomized to lopinavir plus raltegravir in both the proximal femur (-5.2% versus -2.9%, mean difference -2.4%, P = 0.0001) and the lumbar spine (-4.2% versus -2.0%, mean difference -2.1%, P = 0.0006). Changes in both T and Z scores for both the femur and spine were significantly worse in the NRTI group than in the raltegravir group.
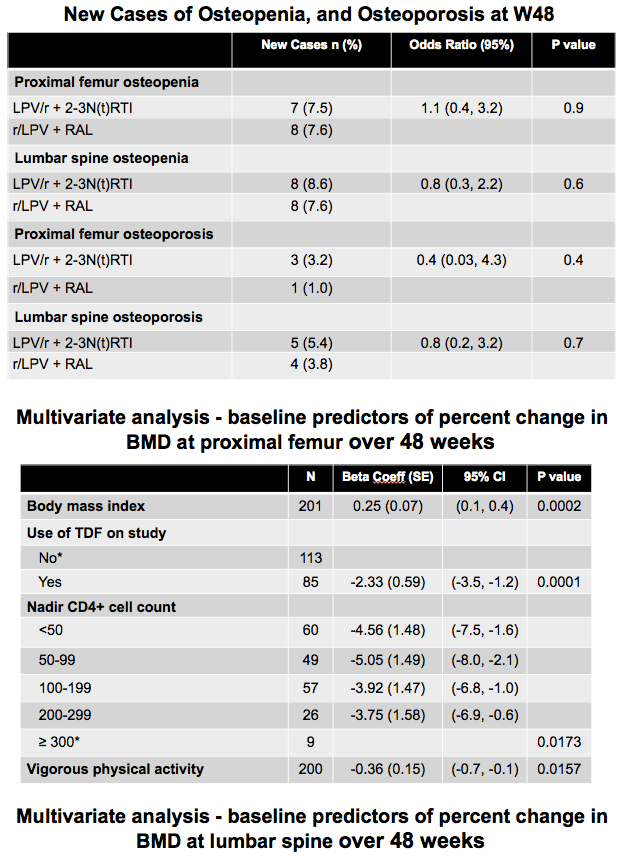
New diagnoses of osteopenia and osteoporosis did not differ significantly between treatment arms. For NRTIs versus raltegravir, new case rates were 7.5% versus 7.6% for femoral osteopenia, 8.6% versus 7.6% for spinal osteopenia, 3.2% versus 1.0% for femoral osteoporosis, and 5.4% versus 3.8% for spinal osteoporosis.
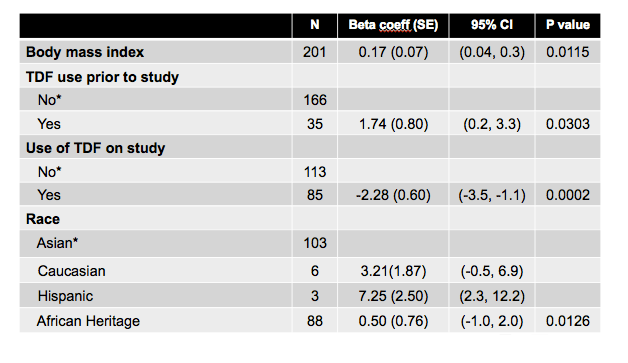
Multivariate analysis to identify predictors of BMD change over 48 weeks considered age, sex, ethnicity, body mass index, smoking, blood pressure, prior and on-study tenofovir use, and other HIV-specific and classic bone risk factors. Significant risk factors for femoral BMD change were body mass index (beta coefficient 0.25, P = 0.0002), on-study tenofovir (beta -2.33, P = 0.0001), lower nadir CD4 count (P = 0.0173), and vigorous physical activity (beta -0.36, P = 0.0157). Significant predictors for spinal BMD change were body mass index (beta 0.17, P = 0.0115), tenofovir use before study (beta 1.74, P = 0.0303), on-study tenofovir (beta -2.28, P = 0.0002), and non-Asian ethnicity (P = 0.0126).
The researchers noted that the magnitude of BMD loss in this study was similar to that seen after antiretroviral-naive people start therapy. They believe these findings "confirmed that reduction in BMD secondary to antiretroviral therapy remains a significant comorbidity in the long-term management of HIV."
Reference
1. Hoy J, Martin A, Moore C, et al. Changes in bone mineral density over 48 weeks among participants randomised to either lopinavir/ritonavir (LPV/r) + 2-3N(t)RTI or LPV/r + raltegravir as second-line therapy: a sub-study of the SECONDLINE trial. 7th IAS Conference on HIV Pathogenesis, Treatment and Prevention, June 30-July 3, 2013, Kuala Lumpur. Abstract WELBB05.

|
| |
|
 |
 |
|
|