 |
 |
 |
| |
Boosted PI (Kaletra) + NNRTIs Successful as 2nd-Line ART in Africa: EARNEST Study - "A pragmatic randomised controlled strategy trial of three second-line treatment options for use in public health rollout programme settings: the Europe-Africa Research Network for Evaluation of Second-line Therapy (EARNEST) Trial"
|
| |
| |
Reported by Jules Levin, IAS 2013 June 30-July 3 Kuala Lumpur
Presented by Nicholas Paton (United Kingdom).
N. Paton1,2, C. Kityo3, A. Hoppe1, J. Hakim4, J. van Oosterhout5, A. Siika6, P. Mwaba7, A. Kambugu8, P. Easterbrook8, J. Boles1, S. Walker1, P. Mugyenyi3, EARNEST Trial Group
1MRC Clinical Trials Unit, London, United Kingdom, 2National University of Singapore, Singapore, Singapore, 3Joint Clinical Research Centre, Kampala, Uganda, 4University of Zimbabwe Clinical Research Centre, Harare, Zimbabwe, 5College of Medicine, University of Malawi, Blantyre, Malawi, 6Moi University School of Medicine, Eldoret, Kenya, 7University Teaching Hospital, Lusaka, Zambia, 8Infectious Diseases Institute, Kampala, Uganda
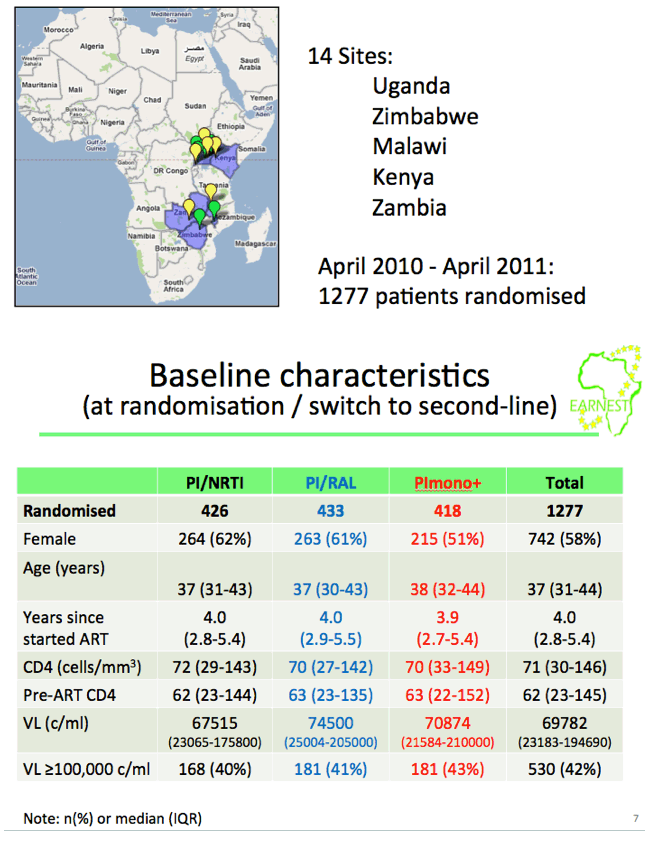
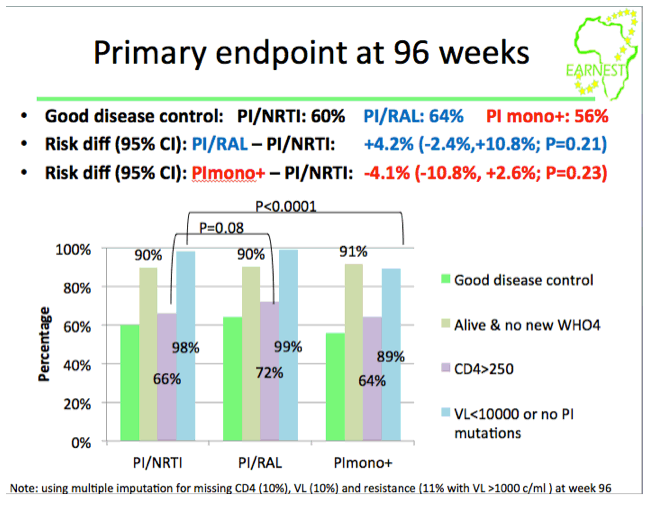
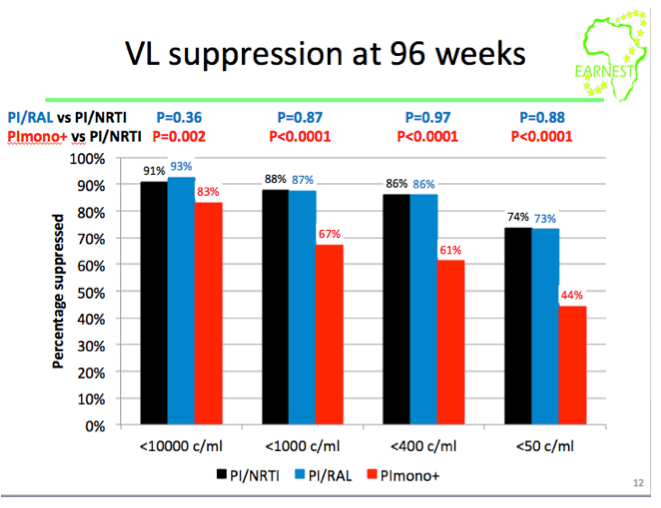
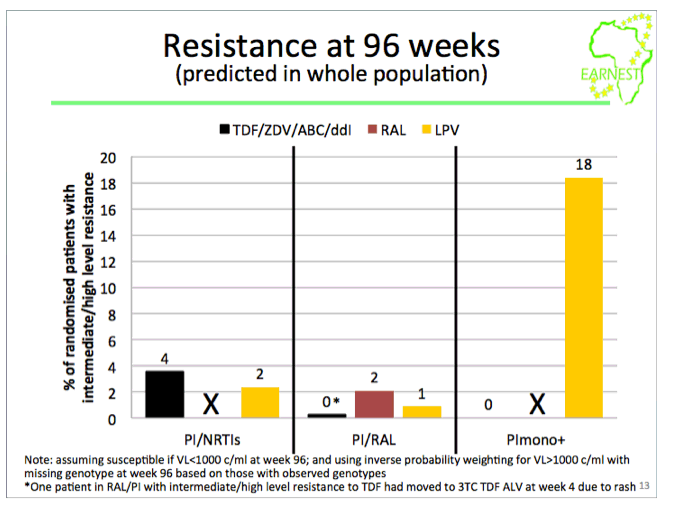
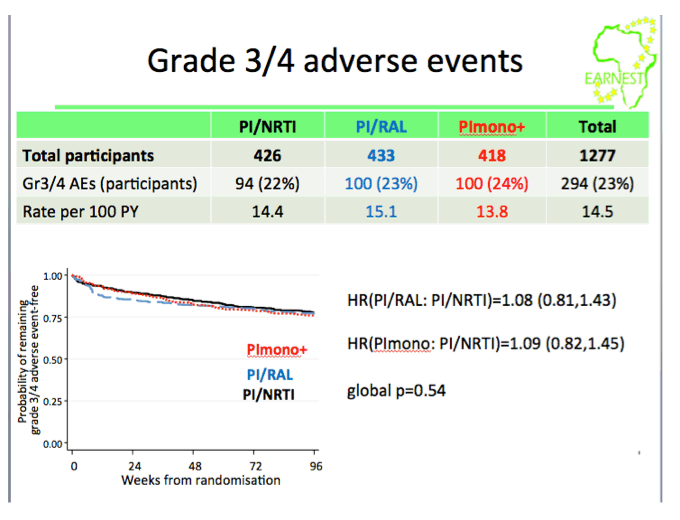
from Jules: this study was conducted in 14 sites in 6 countries in Africa where viral load & resistance testing monitoring are not used and where patients receive standardized first & second line therapy. WHO second-line ART guidelines recommend a boosted PI + 2 NNRTIs but the question is the efficacy of the nukes in 2nd-line therapy since patients failed their 1st-line therapy & may have NNRTI resistance. This EARNEST Study therefore compared a boosted PI, Kaletra, plus NRTIs to 2 other regimens Kaletra mono therapy following a raltegravir induction and Kaletra + raltegravir. Here are the study author's conclusions and if you scroll down to the graph/table showing the viral suppression results you will see 74% receiving the bPI+NRTIs achieved <50 copies/ml, 73% achieved <50 c/ml with the bPI+RAL, and 44% achieved <50 c/ml using Kaletra monotherapy. So the study evaluated if the use of nukes in 2nd-line therapy was compromised because of potential NRTI resistance from using them in the 1st-line regimen & the study results were their use was NOT compromised. Second, the study evaluated if using raltegravir might have a superior efficacy and as you can see the two regimens had similar efficacy. The study aim was also to evaluate boosted PI mono therapy & in this study they used Kaletra which they found did not perform as well. The study aim also was to see if they could use raltegravir instead of nukes to avoid complexity, NRTI side effects/toxicity & cost of using nukes & to evaluate boosted PI monotherapy also to see if they could avoid the nukes for the same reasons just mentioned. In the adverse events slide seen below there was no difference in the total grade 3/4 events between the different regimens, the authors concluded therefore there was no difference but I would suggest there is in general a difference in the actual types of AEs between nukes vs RAL. In the resistance slide below you can see that use of the PI mono therapy arm in this study was associated with the emergence of a greater number of resistance mutations. There is a year more of followup where the study will continue to evaluate outcomes & cost effectiveness. But as you can see in their conclusions just below here they were impressed that they did not find the efficacy of NRTI usage was compromised in the 2nd-line regimen after these patients failed their 1st-line regimen which included NRTIs, and they recommend in the conclusions just below that PI+NRTIs should be made more widely available for 2nd-line therapy in these settings where viral load & resistance testing is not monitored.
AUTHOR CONCLUSIONS
·PI/NRTI had excellent clinical outcome (90% WHO4 event-free survival) and VL suppression (86% < 400c/ml) at 96 weeks, even in advanced 1st-line failure (and without using resistance testing or VL monitoring)
·PI/NRTI regimen should be made more widely available for 2nd line therapy in public health approach
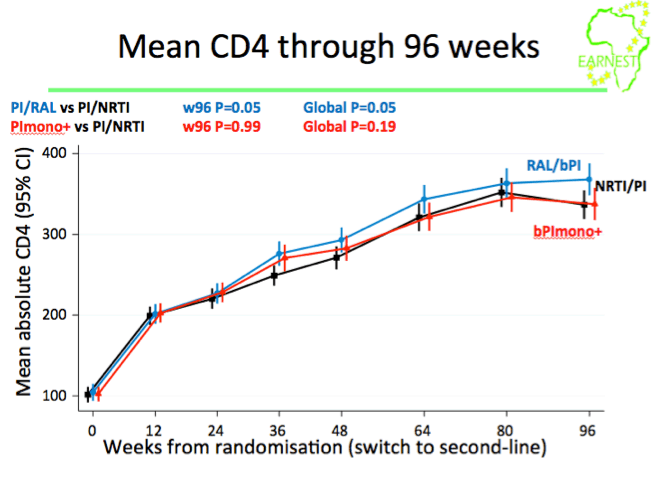
·PI/RAL was not superior to PI/NRTI; borderline CD4 advantage - 144w follow-up will assess clinical benefit and cost-effectiveness
·PI monotherapy is inferior to PI/NRTI: lower VL suppression, more resistance. PI mono unsuitable for public health approach.
·New/recycled NRTIs retain substantial virological activity in 2nd line with little added toxicity
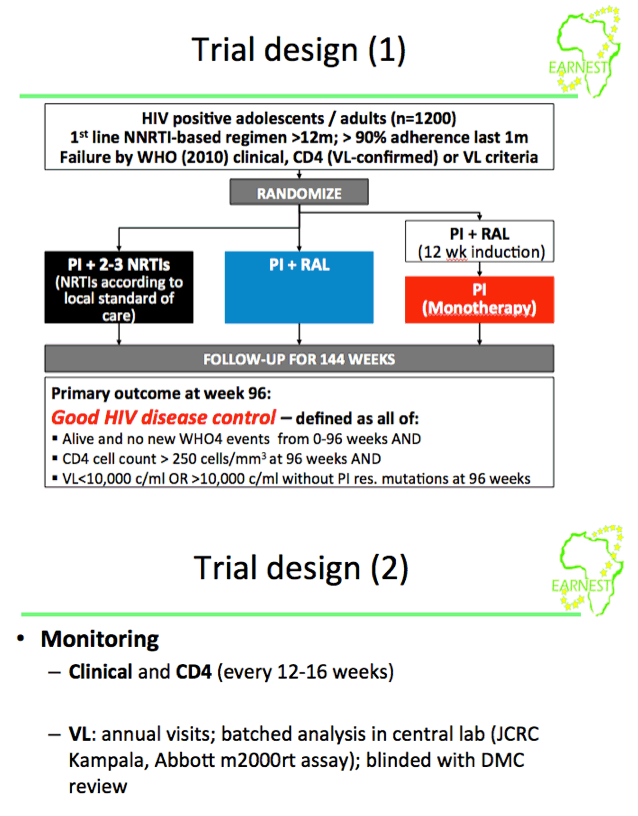
Background: The incremental benefits of new/recycled NRTIs or raltegravir (RAL) on a boosted PI (bPI) backbone for second-line therapy are uncertain, particularly in typical rollout programme settings where individualised therapy with resistance testing, regular viral load (VL) monitoring, and early treatment switches are infeasible.
Methods: 1277 patients aged ≥12y who met WHO-defined treatment failure criteria (confirmed by VL >400 copies/ml) after >12 months on NNRTI-based first-line treatment were randomised in an open-label trial in 14 sub-Saharan African sites.
The superiority of
(B) bPI plus RAL (400 mg b.d.) and the non-inferiority (10% margin) of
(C) bPI monotherapy (+RAL induction for first 12 weeks) were compared to
(A) bPI+2/3 physician-selected NRTIs.
bPI was standardised to lopinavir/ritonavir,400mg/100mg b.d.
Treatment was monitored clinically and by open CD4 count; VL and resistance testing were done annually blinded, reviewed by a data monitoring committee. The primary (composite) endpoint, good disease control, was defined as no new WHO stage 4 events (or death) after randomisation, and CD4 count >250 cells/mm3 and VL < 10,000 copies/ml (or >10,000 copies/ml without major/minor PI resistance mutations) at week 96.
Results: Patients were 58% female, median baseline CD4=71 cells/mm3, VL=69,782 copies/ml; 1% were withdrawn/lost to follow-up by week 96. Proportions with good disease control were (A) 60%, (B) 65% (absolute risk difference vs A: +5.7% (-0.9%,+12.3%; P=0.09)) and (C) 57% (difference vs A: -2.2% (-9.0%,+4.5%; P=0.52)).
There was no difference in grade 3/4 adverse events between groups (P=0.39). However, 61% (C) had VL< 400 copies/ml at 96 weeks vs 86% (A) (difference -25% (-31%,-19%; P< 0.0001)) and 86% (B) (difference vs A -0.2% (-5%,+ 4%; P=0.94). Based on samples analysed to date we estimate 3% (A), 1% (B) and 17% (C) (p=0.002) have intermediate/high level lopinavir resistance (Stanford).
Conclusion:
bPI+RAL did not show clear superiority to bPI+2NRTI at week 96; further follow up will determine whether it is advantageous over the longer term. bPI monotherapy was non-inferior on the primary endpoint, but markedly lower rates of VL suppression and more resistance indicate that this approach is unsuitable for typical rollout programme settings that lack regular/reliable VL monitoring. New/recycled NRTIs retain substantial virological activity.




|
| |
|
 |
 |
|
|