 |
 |
 |
| |
Stribild and Comparison Combinations
Have Similar Efficacy in Over-50s and Under-50s
|
| |
| |
ICAAC, September 10-13, 2013, Denver
Mark Mascolini
Stribild--the one-pill once-daily combination of elvitegravir, cobicistat, tenofovir, and emtricitabine--controlled HIV as well in people over 50 as in younger adults after 96 weeks in two phase 3 trials [1]. Virologic results with Stribild differed little from those with the comparison regimens--efavirenz or atazanavir/ritonavir (both with tenofovir/emtricitabine)--though certain safety results differed between regimens.
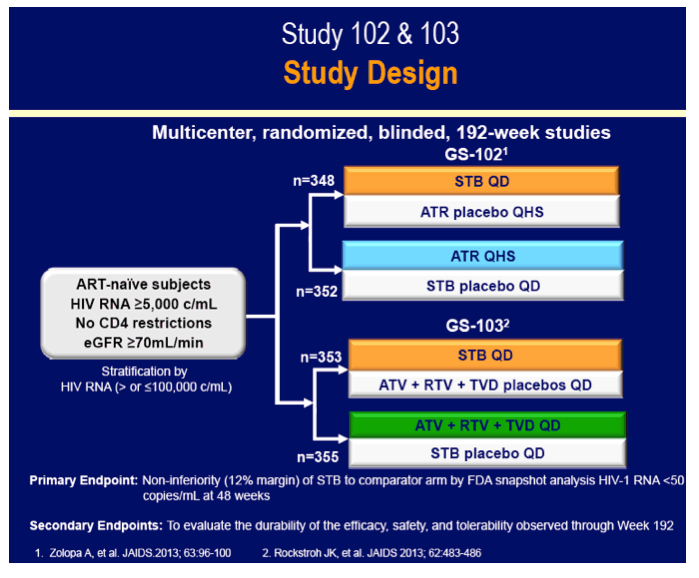
The GS-102 and GS-103 trials established the similar virologic efficacy of Stribild with an efavirenz (Atripla) regimen [2] or an atazanavir/ritonavir regimen [3]. Because HIV populations worldwide are aging, researchers working on these trials compared 96-week efficacy and safety results in people under 50 years old with those in people 50 or older for all three study regimens.
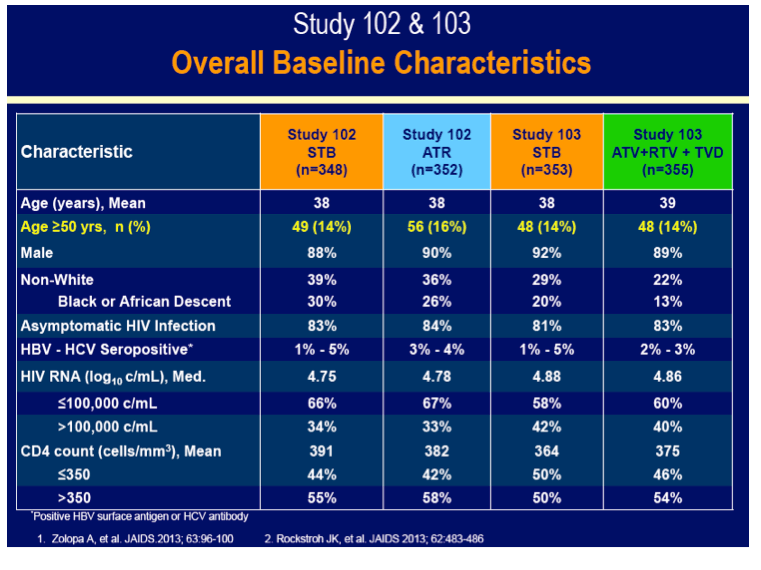
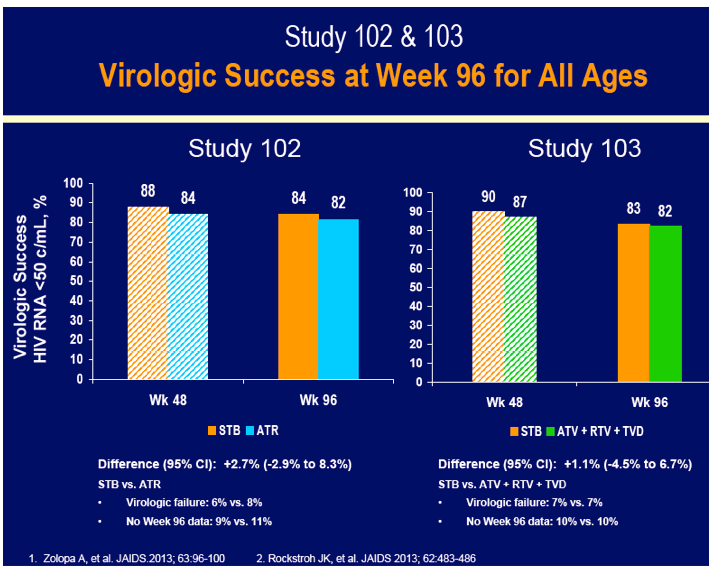
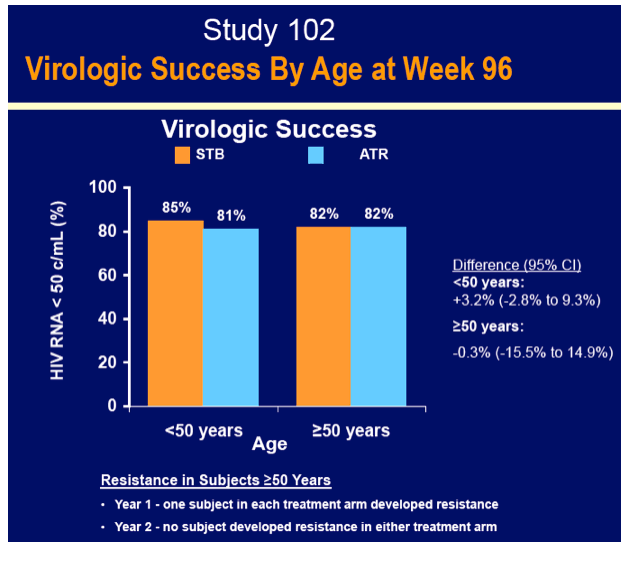
GS-102 included 49 and 299 people in the older and younger groups assigned to Stribild and 56 and 296 in the younger and older groups assigned to efavirenz. An FDA-favored snapshot analysis determined that slightly more than 80% in each age group had a viral load below 50 copies at week 96 regardless of regimen (50 or older, 82% below 50 copies in each arm; younger than 50, 85% on Stribild and 81% on efavirenz below 50 copies). In the first year of treatment among people 50 or older, 1 person in each treatment arm had resistant virus emerge upon virologic failure.
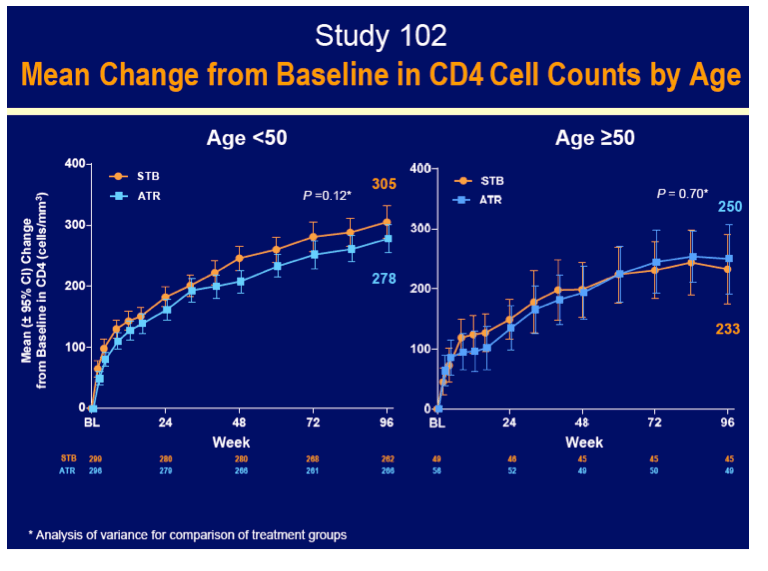
In the Stribild arm, older people gained fewer CD4 cells through 96 weeks (average 233 versus 305 in the younger group), though that difference may not be clinically meaningful. In the efavirenz arm, average CD4 gains at week 96 were 250 in younger participants and 278 in older participants. Within age groups, CD4 gains did not differ significantly by regimen through 96 weeks.
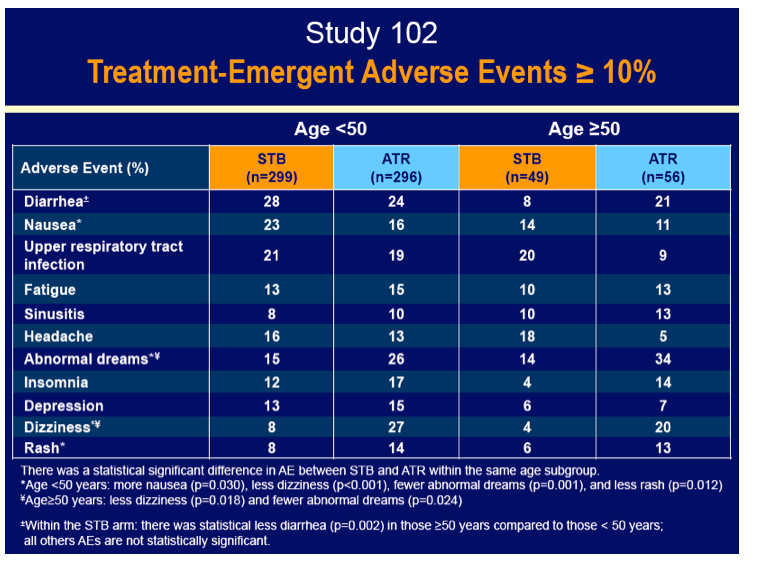
A larger proportion of older than younger people stopped treatment because of an adverse event in both the Stribild group (10% versus 4%) and the efavirenz group (11% versus 6%). But serious adverse event rates did not vary much between the older and younger groups in either the Stribild arm (18% versus 16%) or the efavirenz arm (13% versus 9%). Lower proportions of older and younger people randomized to Stribild than to efavirenz had neuropsychiatric adverse events (33% older and 49% younger versus 63% older and 67% younger) or rash (14% and 22% versus 29% and 31%). Among people 50 and older, dizziness and abnormal dreams were significantly less frequent with Stribild than with efavirenz (P = 0.018 and P = 0.024).
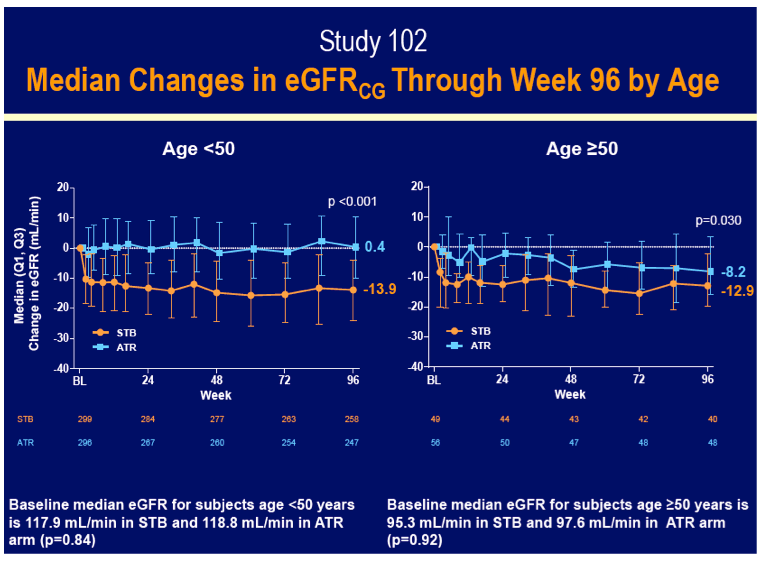
Median estimated glomerular filtration rate (eGFR) fell (worsened) in older people on Stribild (-12.9 mL/min), younger people on Stribild (-13.9 mL/min), and older people on efavirenz (-8.2 mL/min), while rising in younger people on efavirenz (+0.4 mL/min). The eGFR difference between the Stribild and efavirenz arms was significant in both older people (P = 0.03) and younger people (P < 0.001).
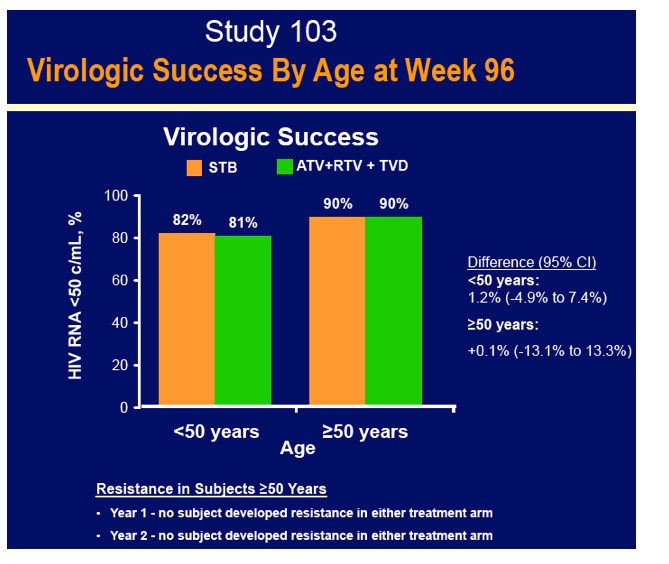
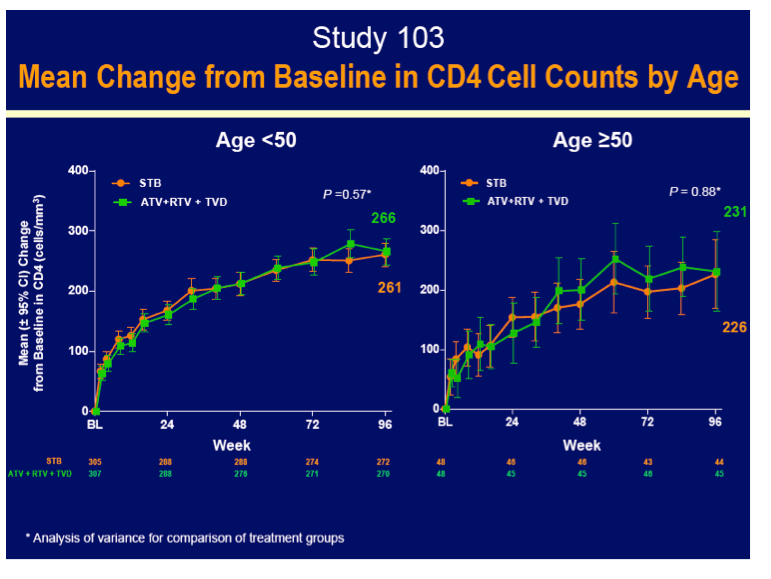
GS-103 included 48 people 50 or older and 305 under 50 randomized to Stribild and 48 people 50 or older and 307 under 50 randomized to atazanavir/ritonavir. Snapshot analysis determined that 90% of the older groups randomized to either regimen had a 96-week viral load under 50 copies, compared with 82% and 81% of younger people randomized to Stribild or atazanavir/ritonavir. No resistance mutations emerged in either treatment arm. On average, the older groups gained 35 fewer CD4 cells than younger people in both treatment arms through 96 weeks--probably a meaningless difference.
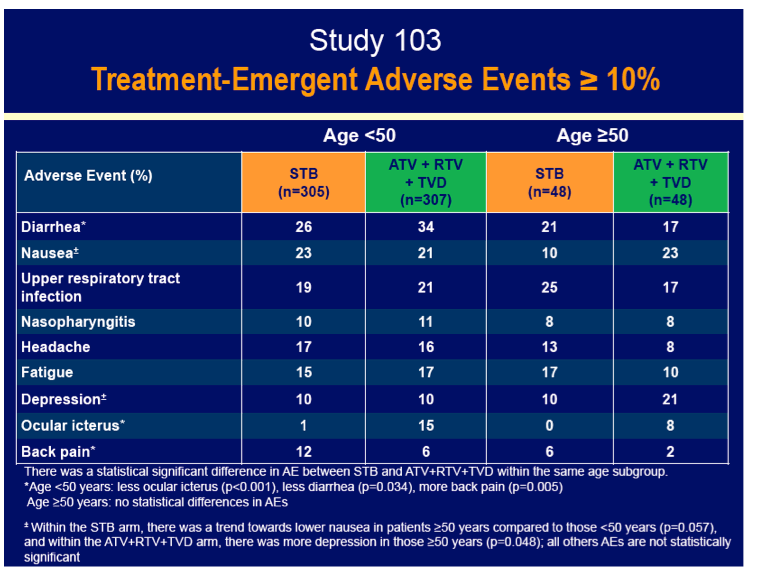
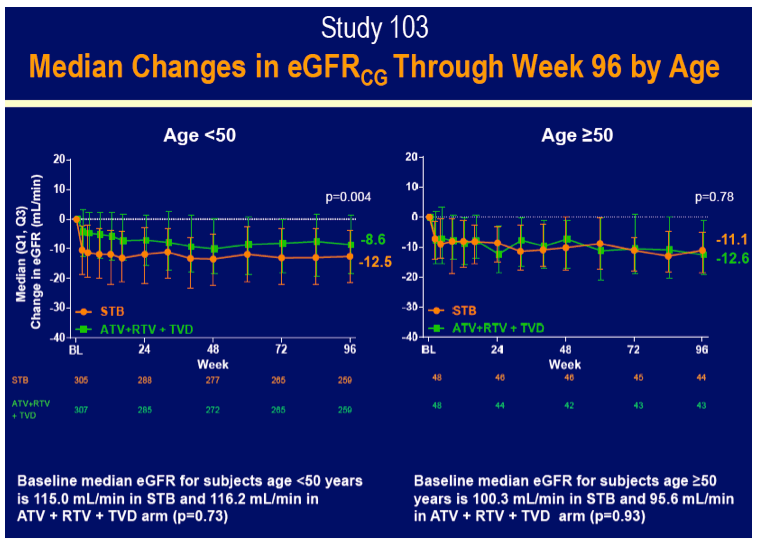
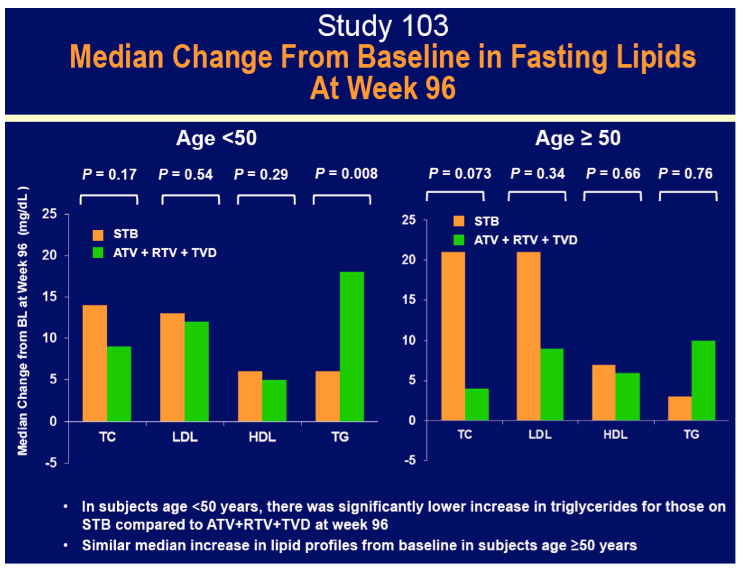
Rates of adverse events leading to discontinuation hardly differed by age or by treatment assignment, with all rates 8% or lower. Serious adverse events affected fewer older and younger people assigned to Stribild (8% and 10%) than assigned to atazanavir/ritonavir (23% and 13%). About one quarter of older and younger study participants in each treatment arm had rash. Among people 50 and older, rates of no treatment-emergent adverse event differed significantly between the Stribild group and the atazanavir group. Median eGFR fell more with Stribild than atazanavir/ritonavir in people younger than 50 (-12.5 versus -8.6 mL/min, P = 0.004), but eGFR declines were similar with Stribild and atazanavir/ritonavir in the older group (-11.1 and -12.6 mL/min).
These 96-week results suggest that being 50 or older does not imperil responses to any of these antiretroviral regimens. Older trial participants gained somewhat fewer CD4 cells, a finding seen in many studies and possibly reflecting decreasing thymic output with age. Because all of these regimens include tenofovir, and because cobicistat may also affect eGFR [4], this measure deserves close attention in an aging population taking these combinations.
References
1. Gallant JE, Hardy D, Bredeek F, et al. Elvitegravir/cobicistat/emtricitabine/tenofovir DF demonstrates comparable efficacy and favorable tolerability to efavirenz and to ritonavir-boosted atazanavir plus FTC/TDF in subjects 50 years and older at week 96. ICAAC. September 10-13, 2013. Denver. Abstract H-1459.
2. Zolopa A, Sax PE, DeJesus E, et al. A randomized double-blind comparison of coformulated elvitegravir/cobicistat/emtricitabine/tenofovir disoproxil fumarate versus efavirenz/emtricitabine/tenofovir disoproxil fumarate for initial treatment of HIV-1 infection: analysis of week 96 results. J Acquir Immune Defic Syndr. 2013;63:96-100.
3. DeJesus E, Rockstroh JK, Henry K, et al. Co-formulated elvitegravir, cobicistat, emtricitabine, and tenofovir disoproxil fumarate versus ritonavir-boosted atazanavir plus co-formulated emtricitabine and tenofovir disoproxil fumarate for initial treatment of HIV-1 infection: a randomised, double-blind, phase 3, non-inferiority trial. Lancet. 2012;379:2429-2438. www.natap.org/2012/HIV/PIIS0140673612609180.pdf
4. German P, Liu HC, Szwarcberg J, et al. Effect of cobicistat on glomerular filtration rate in subjects with normal and impaired renal function. J Acquir Immune Defic Syndr. 2012;61:32-40. http://www.natap.org/2012/HIV/092012_06.htm
--------------------------
Elvitegravir/Cobicistat/Emtricitabine/Tenofovir DF Demonstrates Comparable Efficacy and Favorable
Tolerability to Efavirenz/Emtricitabine/Tenofovir DF and to Ritonavir-boosted Atazanavir Plus Emtricitabine/Tenofovir
DF in Patients ≥50 Years at Week 96
Studies 102 and 103 - Age Sub-analysis
J Gallant1, D Hardy2, F Bredeek3, K Workowski4, W Towner5, L Dau6, H Liu6, J Curley6, M Rhee6, D Piontkowsky6, J Szwarcberg6
1Southwest CARE Center, Santa Fe, NM, 2David Geffen School of Medicine-UCLA, Los Angeles, CA, 3Metropolis Med Group, San Francisco, CA, 4Emory Univ, Atlanta, GA, 5Kaiser Permanente, Los Angeles, CA, 6Gilead Sciences, Foster City, CA
Conclusions
Subjects age ≥50 years on STB through Week 96
· Had high and similar rates of virologic suppression as subjects age <50 years
· Had low rate of treatment emergent drug resistance (n=1)
· Had significantly less dizziness and abnormal dreams compared to ATR
· Experienced an early decline in eGFR (CG) that stabilized through Week 96
- greater decline than with ATR
- similar to ATV+RTV + TVD
· Had similar median CD4 increases to ATR and ATV+RTV + TVD
- Age ≥50 compared to <50 years for all treatment groups had lower increases in CD4 cell count
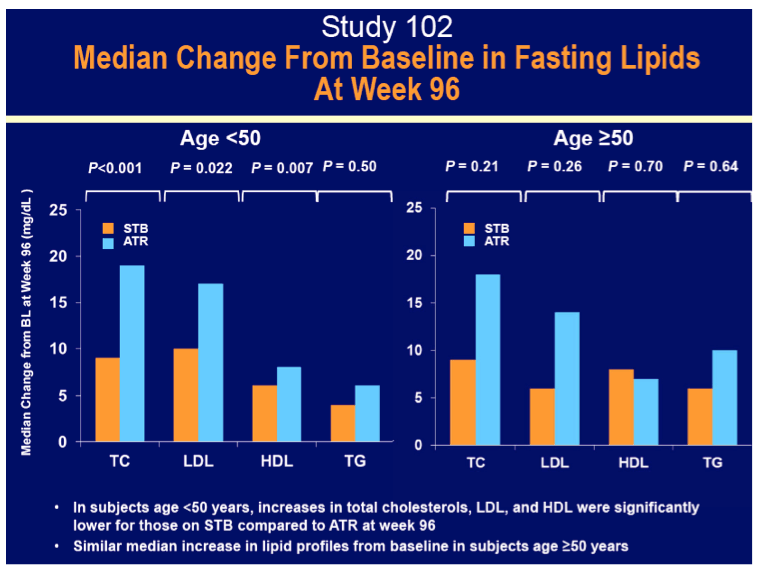
· Had similar median increases in lipid parameters to ATR and ATV+RTV + TVD


|
| |
|
 |
 |
|
|