 |
 |
 |
| |
15% of HIV+ Adults in NY Clinics Still Seronegative for Measles, Mumps, Rubella
|
| |
| |
15% of HIV+ Adults in NY Clinics Still Seronegative for Measles, Mumps, Rubella
IDWeek, October 2-6, 2013, San Francisco
Mark Mascolini
About 15% of 1481 HIV-positive adults tested in two New York City clinics remained seronegative for measles, mumps, and rubella (MMR) and thus could benefit from vaccination [1]. Among vaccinated adults, seroconversion rates were above 50% for each of the infections.
Measles, mumps, and rubella, which can be prevented by vaccination, often have a more severe course in HIV-positive people. To get a better understanding of MMR serostatus and vaccination rates in HIV-positive people, researchers at Weill Cornell Medical College conducted a retrospective chart review of HIV-positive adults in their clinic from 1998 through April 2011 who had serologic MMR assessment and at least two CD4 count 6 months apart. The researchers used electronic medical records to identify patients who had MMR serology, their serology dates, CD4 counts and viral loads performed within 2 months of serology, MMR vaccination dates, age at first serology, and gender. For vaccinated people, the Cornell team measured CD4 count and viral load 3, 6, and 12 months after vaccination. Because viral load assays changed over time, an undetectable load in this study meant below 20 to below 400 copies.
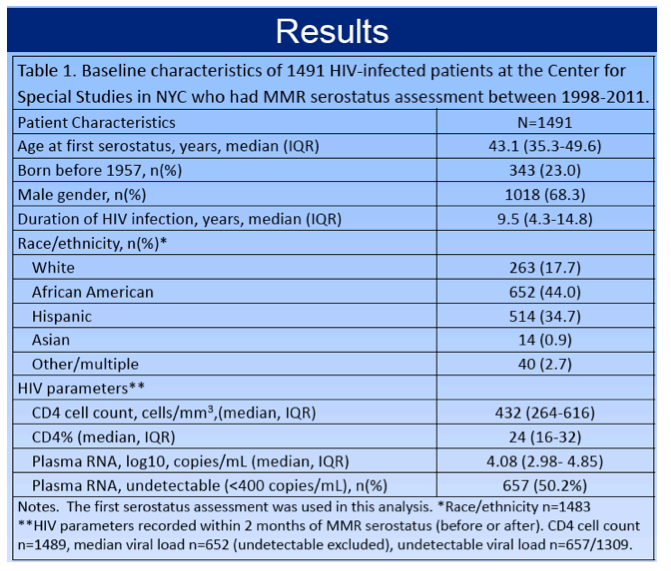
The 1491 people who had MMR serostatus assessment had a median age of 43.1 (interquartile range [IQR] 35.3 to 49.6) at first assessment. Two thirds of study participants (68.3%) were men, and the group had been infected with HIV for a median 9.5 years (IQR 4.3 to 14.8). While 44% of participants were African American, 35% were Hispanic, 18% white, and 1% Asian. Median CD4 count measured within 2 months of serostatus assessment was 432 (IQR 264 to 616) and median viral load 4.08 log10 copies (about 12,000 copies). Half of the study group had a viral load below 400 copies.
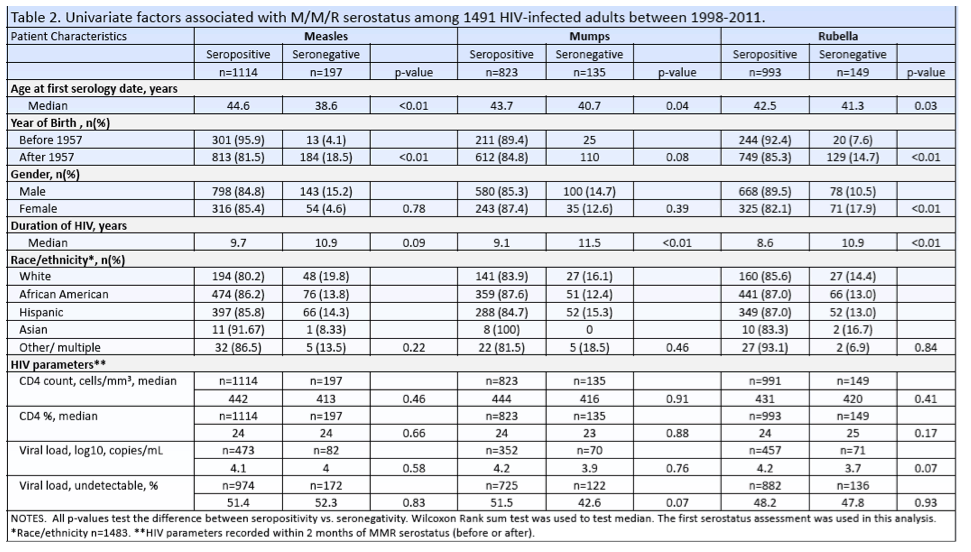
Seropositivity rates were 85.3% for measles, 85.6% for mumps, and 87.2% for rubella. For measles, seronegative people were younger than seropositives (median 38.6 versus 44.6, P < 0.01), more likely to be born after 1957 (18.5% versus 4.1%, P < 0.001), and had a trend toward longer HIV duration (10.9 versus 9.7 years, P = 0.09). People seronegative for mumps were also significantly younger than seropositives (40.7 versus 43.7, P = 0.04), tended to be born after 1957 (15.2% versus 10.6%, P = 0.08) and had significantly longer HIV duration (11.5 versus 9.1 years, P < 0.01).
People seronegative for rubella were significantly younger than seropositives (41.3 versus 42.5, P = 0.03), more likely to be born after 1957 (14.7% versus 7.6%, P < 0.01), and had significantly longer HIV duration (10.9 versus 8.6 years, P < 0.01). Women were more likely to be seronegative for rubella than men (17.9% versus 10.5%, P < 0.01). Race, CD4 count, and viral load did not differ significantly between people seronegative versus seropositive for measles, mumps, or rubella.
Multivariate logistic regression determined that measles seropositivity was more likely with older age (odds ratio [OR] 1.08 per year, P < 0.001) and shorter duration of HIV infection (OR 0.93 per year, P < 0.001). People seropositive for mumps were also older (OR 1.04 per year, P < 0.001) and had HIV infection for a shorter period (OR 0.94 per year, P < 0.001). Rubella seropositivity was also associated with older age (OR 1.04 per year, P < 0.001) and shorter HIV duration (0.94 per year, P < 0.001) and was also associated with male gender (OR 1.86, P < 0.001).
Among 173 people who had MMR vaccination, 86 had postvaccination serology results. Seroconversion rates after single MMR vaccination were 53% for measles, 61% for mumps, 83% for rubella, 40% for any two vaccine components, and 50% for all three components. No cases of measles, mumps, or rubella developed during the study period. CD4 count and viral load did not change significantly 3, 6, or 12 months after vaccination.
The investigators noted that medical records did not include full data on HIV risk factors, country of birth, immigration status, nadir CD4 count, and use of antiretroviral therapy. Because all serostatus tests were ordered at the discretion of clinicians (until measles serology was added to routine new patient testing in April 2011), serostatus assessment may have been biased.
The researchers proposed that younger HIV-positive patient, those born after 1957, and those with longer duration HIV infection should have MMR serostatus assessment as part of routine care. The Cornell team believes their findings support 2013 ACIP guidelines for MMR vaccination [2].
References
1. Singh H, Chiu YL, Wilkin T. Measles, mumps, and rubella serostatus among an HIV-infected cohort and response to MMR vaccination. IDWeek 2013. October 2-6, 2013. San Francisco. Abstract 335.
2. Centers for Disease Control and Prevention. Advisory Committee on Immunization Practices (ACIP) Recommended Immunization Schedule for Adults Aged 19 Years and Older-United States, 2013. Morbidity and Mortality Weekly Report (MMWR). February 1, 2013;62:9-19. http://www.cdc.gov/mmwr/preview/mmwrhtml/su6201a3.htm
-------------------------------
Reported by Jules Levin
Measles, Mumps, and Rubella (MMR) Serostatus and Response to MMR Vaccination among HIV-infected Adults
Harjot K. Singh, MD, ScM, Ya-Lin Chiu, MS, Timothy Wilkin, MD, MPH
Weill Cornell Medical College/New York Presbyterian Hospital
CONCLUSIONS
In a large diverse NYC adult HIV clinic, approximately 15% of persons were seronegative for measles, mumps, or rubella. Younger patients, patients born after 1957, and patients with longer duration of HIV infection are at higher risk of measles, mumps, and rubella seronegativity and should have serostatus assessment as part of routine care. MMR vaccination had no negative impact on HIV control and 53-83% of patients were able to achieve seroconversion after single MMR vaccination. Our study suggests that we should be assessing MMR serostatus in our patients and provides evidence to support the new 2013 ACIP guidelines. Prospective studies of MMR vaccination with more complete demographic and vaccination records among HIV-infected persons would further inform our understanding of these preventable diseases.
Study Limitations
·Retrospective nature of this study
·Lack of extractable data from the EMR for several important variables limited our ability to extract vaccination records, HIV risk factors, country of birth/immigration status, nadir CD4 cell count, use of HAART, and HAART risk factors
·All tests were ordered at the discretion of the care provider and may have introduced additional bias into serostatus assessment.
·We were not able to assess cellular response which plays a role in MMR immunity
·We did not have an HIV-uninfected group for comparison.




|
| |
|
 |
 |
|
|